DEVELOPMENT AND VALIDATION OF A RP- HPLC METHOD FOR SIMULTANEOUS ESTIMATION OF OMEPRAZOLE AND CINITAPRIDE IN BULK AND CAPSULE DOSAGE FORM
Texto
Imagem

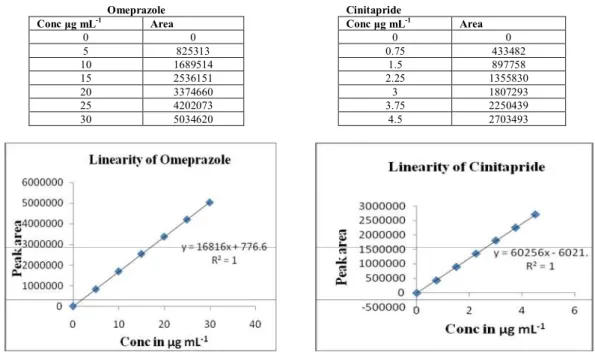


Documentos relacionados
Simultaneous estimation and validation of developed method of Nifedipine (NIF) and Atenolol (ATN) in combined dosage form as well as in laboratory mixture
Yadav .Method development and its validation for simultaneous estimation of Atorvastatin and Amlodipine in combination in tablet dosage form by UV spectroscopy,
Development and validation of a stability-indicating HPTLC method for analysis of Nebivolol hydrochloride and Hydrochlorothiazide in the bulk material and in
Then pipette out 10ml from the standard stock solution in other 100ml volumetric flask and diluted up to mark with Methanol to give a working standard solution having
Preparation of stock solution: The Stock Solution has been prepared by weighing accurately and transferred 10 mg of Prasugral Working standard into a 10 mL volumetric
A new simple high performance thin layer chromatographic method (HPTLC) for simultaneous determination of nabumetone and paracetamol in combined tablet dosage form was developed
The proposed HPLC method is simple, isocratic, rapid, speciic, accurate and precise for determination of GMFX in bulk, pharmaceutical dosage formulation and human serum
The CPE-AaCNT electrode was successfully employed in the development of an electroanalytical method for the simultaneous determination of copper and lead in commercial samples