Influence of tungsten and titanium on the structure of chromium cast iron
Texto
Imagem
![Fig. 1. Microstructure of high-chromium cast iron – hypoeutectic – a), near-eutectic – b) and hypereutectic –c) [7,own studies]](https://thumb-eu.123doks.com/thumbv2/123dok_br/18172721.330125/2.871.96.389.173.868/fig-microstructure-high-chromium-hypoeutectic-eutectic-hypereutectic-studies.webp)

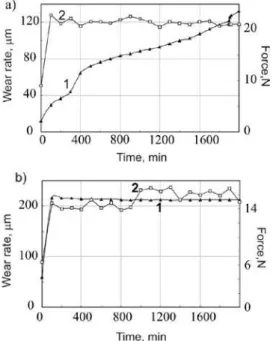
Documentos relacionados
he alloying elements of maraging steel, such as molybdenum, titanium, nickel, cobalt, aluminum, and others, have a great influence on the mechanical properties of this alloy, in
Remelted zone of the probes after treatment with current intensity of arc plasma – 100 A, lower bainite, retained austenite and secondary cementite.. Because the secondary
We also determined the critical strain rate (CSR), understood as the tangent of the inclination angle between the tangent to the crack development curve and the crack development
This log must identify the roles of any sub-investigator and the person(s) who will be delegated other study- related tasks; such as CRF/EDC entry. Any changes to
Além disso, o Facebook também disponibiliza várias ferramentas exclusivas como a criação de eventos, de publici- dade, fornece aos seus utilizadores milhares de jogos que podem
The probability of attending school four our group of interest in this region increased by 6.5 percentage points after the expansion of the Bolsa Família program in 2007 and
Na hepatite B, as enzimas hepáticas têm valores menores tanto para quem toma quanto para os que não tomam café comparados ao vírus C, porém os dados foram estatisticamente
É nesta mudança, abruptamente solicitada e muitas das vezes legislada, que nos vão impondo, neste contexto de sociedades sem emprego; a ordem para a flexibilização como