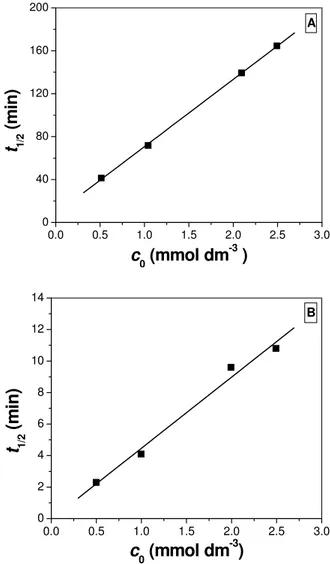
Kinetics of photocatalytic removal of 2-amino-5-chloropyridine from water
Texto
Imagem

Documentos relacionados
photocatalytic process, phenol is used as another colorless target pollutant to test the photocatalytic activity of BiFeO 3 microspheres and 16CN/BFO sample under simulated
The degradation experiments of this study have shown that the solar photocatalytic reactor, using immobilized TiO 2 photocatalyst is more efficient than the
The photocatalytic activity of prepared photocatalysts was investigated by degradation of rhodamine B (RhB) in aqueous solution under visible light irradiation and natural sunlight..
Recently, [7] suggested a Bayesian approach in order to detect additive outliers in Poisson first-order INteger-valued AutoRegressive, INAR(1), modeis. The clnss of INAR modeis for
The α -amino acid ( S )- 5 was synthesized using in the key step a chemoselective nucleophilic substitution between a diester derived from L-aspartic acid and 2-lithium
The photocatalytic degradation and decolorization of MO was also investigated at a fixed concentration of MO (10.0 mg L -1 ) in pH 7.0 aqueous solution in the presence. of
This paper describes the photocatalytic degradation of EDTA in the aqueous solution using phosphate modified nano titania with the final aim of establishing the best
2-Amino-4-aryl-6-mercaptopyridine-3,5-dicarbonitriles as starting materials have been prepared by reducing of 2-amino-4-aryl-6-(phenylthio)pyridine-3,5-dicarbonitrile derivatives