METHOD DEVELOPMENT AND VALIDATION OF METFORMIN, GLIMEPIRIDE AND PIOGLITAZONE IN TABLET DOSAGE FORM BY RP-HPLC
Texto
Imagem
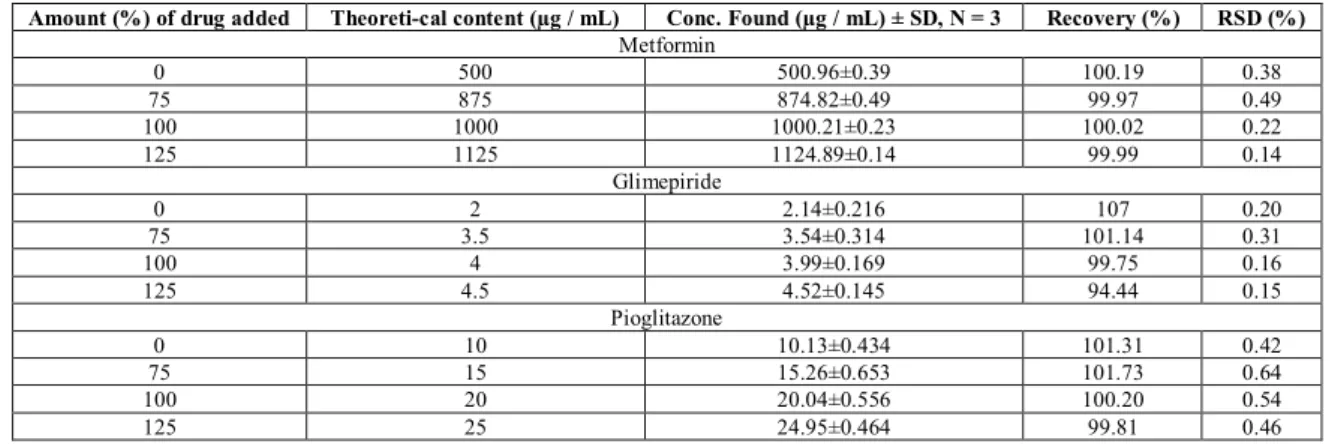
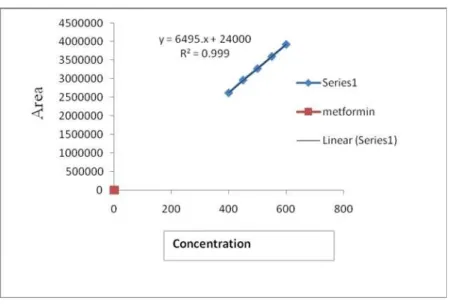
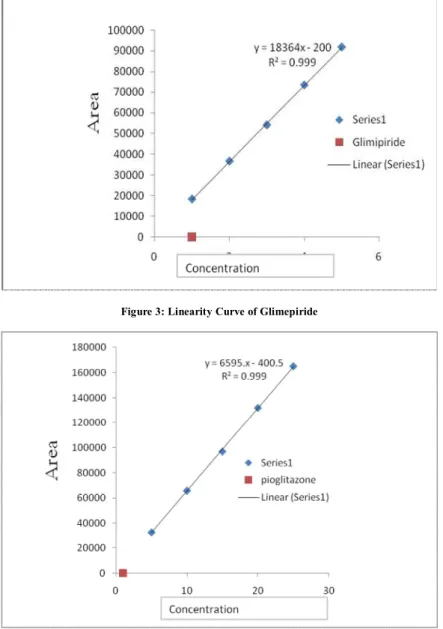
Documentos relacionados
Commercial capsule formulation was successfully analyzed using the developed method and the proposed method is applicable to routine analysis of determination of
The two points considered at the alternate sides, of the tangents through the diameter of the circle, and then the line joining these points divides the circle
At the first stage of the measurements results analysis, the gear wheel cast surface image was compared with the casting mould 3D-CAD model (fig.. Next, the measurements results
The structure of the remelting zone of the steel C90 steel be- fore conventional tempering consitute cells, dendritic cells, sur- rounded with the cementite, inside of
Development and Validation of a HPLC Method for the Determination of Aldicarb, Aldicarb Sulfoxide and Aldicarb Sulfone in Liquid Samples from Anaerobic Reactors..
A new simple high performance thin layer chromatographic method (HPTLC) for simultaneous determination of nabumetone and paracetamol in combined tablet dosage form was developed
In the present work, metformin HCl and glimepiride, the two well-known anti- diabetic drugs, have been evaluated directly by transmission FTIR in dosage form by preparing
Validation parameters of the developed HPLC method for simultaneous determination of sesquiterpene lactones in the dichloromethane fraction of the hydroalcoholic extract obtained