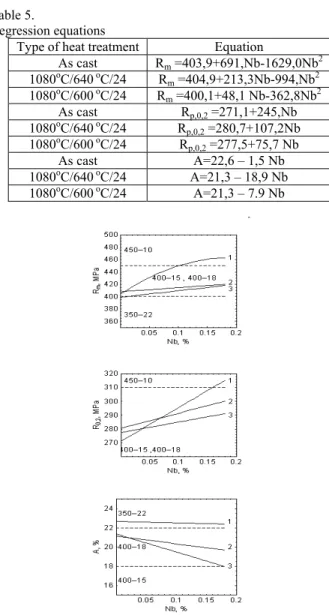
Synthesis of nanoparticeles in ductile iron with small additions of vanadium and niobium and its mechanical properties
Texto
Imagem

Documentos relacionados
[r]
Este artigo discute o filme Voar é com os pássaros (1971) do diretor norte-americano Robert Altman fazendo uma reflexão sobre as confluências entre as inovações da geração de
4.1. Ratificam-se os termos das Condições Gerais que não foram expressamente alterados pela presente Cobertura Adicional... Garantir ao Segurado, até o limite máximo de
Portanto, é importante que os funcionários das farmácias e drogarias, os médicos, enfermeiros e farmacêuticos das unidades básicas de saúde, assim como
Figura 8.4: Evolução da energia absorvida pela estrutura do automóvel ao longo do tempo, para as simulações de impacto lateral com (c/ Mac) e sem (s/ Mac) padding de
This paper uses a variation in access to a targeted loan program from the Brazilian Development Bank (BNDES) to assess the causal effect of better credit conditions on investment
There are several causes contributing to PPI refractoriness, such as inappropriate use of the drug (lack of patient adherence to PPI therapy, inadequate dosage of PPI), residual
[r]