Brazilian
Journal
of
OTORHINOLARYNGOLOGY
www.bjorl.org
ORIGINAL
ARTICLE
Brainstem
auditory
evoked
potentials
in
children
with
lead
exposure
夽
Katia
de
Freitas
Alvarenga
a,
Thais
Catalani
Morata
b,
Andrea
Cintra
Lopes
a,
Mariza
Ribeiro
Feniman
a,
Lilian
Cassia
Bornia
Jacob
Corteletti
a,∗aDepartmentofSpeechTherapyandAudiology,FaculdadedeOdontologiadeBauru,UniversidadedeSãoPaulo(FOB-USP),
Bauru,SP,Brazil
bNationalInstituteforOccupationalSafetyandHealth(NIOSH),DivisionofAppliedResearchandTechnology,Cincinnati,OH,
UnitedStates
Received11September2013;accepted23December2013 Availableonline18October2014
KEYWORDS
Toxicity;
Leadpoisoning;
Brainstemauditory
evokedpotentials;
Pure-toneaudiometry
Abstract
Introduction:Earlierstudieshavedemonstratedanauditoryeffectofleadexposureinchildren, butinformationontheeffectsoflowchronicexposuresneedstobefurtherelucidated. Objective: Toinvestigatetheeffectoflowchronicexposuresoftheauditorysysteminchildren withahistoryoflowbloodleadlevels,usinganauditoryelectrophysiologicaltest.
Methods:Contemporarycross-sectionalcohort.Studyparticipantsunderwenttympanometry, puretoneandspeechaudiometry,transientevokedotoacousticemissions,andbrainstem audi-toryevoked potentials,withbloodlead monitoringoveraperiodof35.5months.Thestudy included130children,withagesrangingfrom18monthsto14years,5months(meanage6 years,8months±3years,2months).
Results:Themeantime-integratedcumulativebloodleadindexwas12g/dL(SD±5.7,range:
2.433).Allparticipantshadhearingthresholdsequaltoorbelow20dBHLandnormalamplitudes of transient evoked otoacoustic emissions.No associationwas found between the absolute latenciesofwavesI,III,andV,theinterpeaklatenciesI---III,III---V,andI---V,andthecumulative leadvalues.
Conclusion: Noevidenceoftoxiceffectsfromchroniclowleadexposureswasobservedonthe auditoryfunctionofchildrenlivinginaleadcontaminatedarea.
© 2014Associac¸ãoBrasileira de Otorrinolaringologiae CirurgiaCérvico-Facial. Publishedby ElsevierEditoraLtda.Allrightsreserved.
夽
Pleasecitethisarticleas:AlvarengaKF,MorataTC, LopesAC,FenimanMR,CortelettiLC.Brainstemauditoryevokedpotentialsin childrenwithleadexposure.BrazJOtorhinolaryngol.2015;81:37---43.
∗Correspondingauthor.
E-mail:lilianjacob@fob.usp.br(L.C.B.J.Corteletti).
http://dx.doi.org/10.1016/j.bjorl.2013.12.001
PALAVRAS-CHAVE
Toxicidade;
Intoxicac¸ãopor
chumbo;
Potenciaisevocados
auditivosdotronco
encefálico;
Audiometriadetons
puros
Potencialevocadoauditivodetroncoencefálicoemcrianc¸ascomexposic¸ãoao chumbo
Resumo
Introduc¸ão:Estudosanteriorestêm demonstradoefeitosdaexposic¸ãoaochumbonosistema auditivoemcrianc¸as,porémaexposic¸ãodestemetalemníveisbaixosaindaprecisaser inves-tigada.
Objetivo:Investigarosefeitosdaexposic¸ãocrônicaaochumbonosistemaauditivodecrianc¸as comhistóricodebaixonívelsanguíneodechumbopormeiodeumtesteeletrofisiológico. Método: Estudo de coorte transversal contemporânea. Foram realizados imitanciometria, audiometria tonal liminare vocal, emissões otoacústicas evocadastransientes e potenciais evocadosauditivosdetroncoencefálico,comomonitoramentosanguíneodechumbodurante umperíodode35,5meses.Participaram130crianc¸asnafaixaetáriade18mesesa14anose 5meses(6a8m±3a2m).
Resultados: Amédiaestimadadoíndicedepumblemiafoi12g/dL(DP±5,7).Todosos
partic-ipantesapresentaramlimiaresauditivosiguaisouinferioresa20dBNAeamplitudenormaldas emissõesotoacústicasevocadastransientes.Nãofoiencontradaassociac¸ãoentreaslatências absolutasdasondasI,IIIeVeinterpicosI-III,III-VeI-Veosvalorescumulativosdechumbo. Conclusão:não foiobservadaevidência deefeitostóxicosem baixasexposic¸ões crônicasao chumbosobreafunc¸ão auditivadecrianc¸as quevivememuma áreacontaminada poreste metal.
©2014Associac¸ãoBrasileira deOtorrinolaringologiaeCirurgiaCérvico-Facial.Publicadopor ElsevierEditoraLtda.Todososdireitosreservados.
Introduction
Leadis a known neurotoxic agent that can cause serious
damagetonervoustissue,particularlyduringdevelopment
ofthe centralnervoussystem,resultingin neurocognitive
andneurophysiologicaldisordersinchildrenandadults.Both
occupational and environmental sourcesof lead exposure
constituteapublichealthconcern.
Adversehealtheffects(especiallyincognitivefunction,
attentionand learning) have been linkedto low levelsof
leadinblood(<10---20g/dL).1---5Inareviewoftheliterature
ontheneurotoxicity associatedwith exposureof children
tolowlevelsof lead,theauthors concludedthatthereis
nosafelevelwithout neurologicaleffects inthe body(no
adverse effectlevel), i.e., any exposure toleadis
harm-ful to the central nervous system.6 One of the goals of
Healthy People Program 2020 of the U.S. Department of
Health and Human Services (DHHS) included the
elimina-tionof leadlevelsin blood≥10g/dLinchildrenyounger
than5 years ofage by the year 2020.7 In Brazil, thereis
nopolicy directedtoward the prevention of poisoning by
environmentalexposuretoheavymetalsandcurrent
Brazil-ianstandards stillconsider40g/dLastherecommended
biologicalindex.8
Differenttypesofevokedpotentialsandseveral
neurobe-havioraltestshavebeenusedtodetectsubclinicalchanges
in individuals exposed toa range of lead levels, in order
toprevent acuteand/or persistent neurological disorders
inexposedindividuals.9SchwartzandOtto10suggestedthat
theevoked potentialmay bethe most sensitive indicator
ofcentralnervoussystemdysfunctioninchildren.Evidence
from studies on the effects of occupational exposure to
leadonthehumanauditorysystemhasbeenreportedover
the pastthreedecades.Effects onlatencyand amplitude
werereportedinsomatosensory,visual,andauditoryevoked
potentials, includingcognitiveevokedpotential.11---23
How-ever, thereisnoconsensus:(1)onthelimitandthe level
ofleadpoisoningneededtoinduceeffectsontheauditory
systemofchildren;(2)whichstructuresorfunctionsofthe
auditorysystemaresusceptible;and(3)themostsensitive
testsfortheassessmentofleadpoisoningeffects.
The first study that used brainstem auditory evoked
potential(BAEP)toinvestigatetheeffectson
electrophysio-logicalrecordingsinchildrenexposedtoleadwasperformed
by Otto et al.24 The results showed a significant
associa-tionbetweenthelevelsofbloodlead(mean28g/dL)and
theabsolutelatenciesof wavesIIIandV,withan increase
in latency correlatingwiththeincreased levelsoflead in
blood.Thisfindingsuggestedtheeffectofthismetaloccurs
atthelevelofthelowerbrainsteminthecochlearnucleus
region.However,thepresenceofcochleardamagewasnot
ruledout.
Subsequent studies in children with higher exposure
levels (43---72g/dL) also described changes in BAEPs,
which supported auditory system impairment, but with
no consensus on which auditory system structures were
affected. Some of these studies suggested peripheral
lesions,13,25whileotherssuggestedbothcentraland
periph-eral dysfunctions.26 However, these findings were not
confirmed in other studies,27,28 which found nosignificant
association between leadexposure andauditory function.
An exception was the paper of Holdstein et al.,13 that
assessedthe effectsofleadlevelsinblood obtainedfrom
levelsinbloodobtainedatthetimeoftheinvestigationas
abiomarkerofleadexposure.
Withthis inperspective, thepurposeofthis studywas
toinvestigatetheeffectofchronicexposuretoleadonthe
auditorysystemofchildrenwithahistoryoflowbloodlead
levels,usinganelectrophysiologicaltest.
Methods
This was a contemporary cross-sectional cohort study,
approvedbytheEthicsCommitteeoftheInstitution,under
No. 098/2009. Childrenliving near a battery factory that
caused lead contamination in the soil and river were
recruitedtoparticipateinthestudy,duetotheirhighriskof
leadexposure.Thosewhohadbloodleadlevels≥10g/dL
wereeligiblefor the study,acriterion basedonthe1991
recommendationoftheCentersforDiseaseControland
Pre-vention,whichidentifiedthebloodleadlevelof10g/dLas
‘‘levelofconcern’’.29
The study included 130 children (80 males and 50
females), aged 18 months to 14 years (mean: 6 years, 8
months±2years3months).Levelsofleadinthebloodwere
evaluatedlongitudinally,withallparticipantssubmittedto
audiological assessment. Study participants underwentan
extensiveclinicalevaluationbyateamconsistingofa
pedi-atrician, aneurologist, a dentist,and aspeechtherapist,
andwerefreeofanysymptomsordiagnoseddisease.
Monitoringofleadlevelsinblood
Themonitoringofleadlevelsinblood wasconducted
dur-ingaperiodlasting35.5months.Participantsprovidedtwo
to four blood samples, collected in heparinized tubes by
thelaboratoryinchargefollowingstandardprocedures.The
analyseswereperformedunderthecontroloftheMunicipal
HealthSecretariatandthecity’sRegionalHealthDivision,
andwereallsenttothesamelaboratoryforanalysis.
Sam-plesweretransportedat4◦Candkeptatthistemperature
priortoanalysis.The blood leadlevel wasobtained using
atomicabsorptionspectrometrywithgraphitefurnace.
Thedataoftheoriginalsamplewereusedtoassess
eligi-bility,andtheeligiblecases,thatis,thechildrenwithblood
leadresults≥10g/dLandwithoutanyassociateddisease,
werefollowedforaperiodof35.5months.Duringthisstudy,
therewerefoursessionsforbloodcollection;allparticipants
wereinvitedtoparticipateineachofthem,butnotalldid.
Audiometricscreening
Initially,anotoscopicassessmentwasperformedtoexclude
the presence of perforated tympanic membrane or otitis
externa.
The audiometric test was performed to rule out any
conductiveorsensorineuralhearinglossinthefrequencies
correlatedwiththoseofthe clickstimulus(500---4000Hz),
withthe aimof controlling interference onthe resultsof
the electrophysiological testing. The results of pure-tone
audiometrywereclassifiedasnormalifthehearing
thresh-oldswere≤20dBHL,obtainedwithaMIDIMATE622clinical
audiometermodelandTDH-39Pphones.Tympanometrywas
performed using GSI TympSTAR equipment, and
tympa-nometry curves were considered normal when the static
compliancewas0.3---1.3mLandthe maximumcompliance
peakpressurewas90---100daPa,according totheprotocol
usedintheservice.
Thetestswereperformedinaboothwithacoustic
treat-mentinaccordancewiththeANSIstandard.30
Therecordingoftransientevokedotoacousticemissions
(TEOAEs)wasobtainedinchildrenaged2years,6months
andolder,usingtheOtodynamicsILO292DPECHOResearch
OAESystem. Theprobestabilitywasalways >80%andthe
stimuluswascalibratedbeforeeachdayofdatacollection.
CriteriawerethepresenceofTEOAEresponse,
reproducibil-ityof70%orhigher,andaresponseamplitude<3dBinthe
frequencybandof 1500---5000Hz. Theresultsofpure tone
audiometry,TEOAE,andtympanometrywerewithinnormal
clinicalvalues,confirmingtheabsenceofsensorineural or
conductivehearinglossinallparticipants.
BAEPs
ThetestwasperformedusingaHortmannauditoryevoked
potentials system in an electrically shielded sound-proof
booth,withthechildsittingcomfortably, sleepingor with
eyesclosed,in ordertoeliminatethe artifactscaused by
eyemovement.DisposableKedallelectrodes,model
Medi-trace200,werepositionedasfollows:activeelectrodeFz,
reference electrode/earth in M1 and M2 (right and left
mastoids), to record the ipsilateral BAEP. The individual
electrodeimpedancewaslessthan5k,andbetween
elec-trodes, less than 2k. The click stimulus was presented
throughaTDH-39earphoneatanintensitylevelof80dBHL,
withalternatingpolaritytoreduceelectricalartifacts,and
apresentationrateof21.1clicks/s,averaging1000stimuli
ineachcollection, withbandpassfilterof 30and3000Hz.
Wavereproducibility wasusedtoidentifythe presenceof
responses.Thefirsteartobetestedwasrandomlychosen.
Theabsolutelatenciesof wavesI,III,V,andthevaluesof
interpeakintervals I---III, III---V, and I---V were measured in
milliseconds(ms).
Statisticalanalysis
Descriptivestatisticalanalysiswasperformed by
consider-ingthecomponentsoftheBAEPrecordedseparatelyineach
ear,leadlevelsinblood,andtheestimatedbloodlead
val-ues.Consideringthelackofcertaintyaboutthecumulative
effectofleadontheauditorysystem,i.e.,itsinfluencein
the long-term, the authors decided tostudy the possible
influenceofthedurationofleadpoisoningontheresultsof
theBAEPs.Forthatpurpose,thebloodleadvalueestimated
ontheday of the audiological assessment wascalculated
havingasreferencethe datewhen thefirst blood sample
wascollectedfromthepopulation.Theaudiological
assess-mentwasperformedatdifferenttimesforeachparticipant;
however,withintheperiodofbloodleadlevelmonitoring.
Duringthestudyperiod,severalmeasuresweretakento
preventthechildren’sexposuretolead,andthedata
con-firmedthatthelevelsofleadinblooddecreasedasaresult
ofsuchinterventions.Pearson’scorrelationcoefficientwas
waves I,III, and V andvalues of interpeakintervals I---III,
III---V, I---V) and the independent variables (age, values of
leadinbloodobtainedfromcollectionsmade,andestimated
bloodleadvalues).
Subsequently,alinearregressionmodelwasusedforthe
rightandleftears,usingtheabsolutelatencyandinterpeak
values. Linear regression was the method used to select
variablesin themodel, andthe significancelevel wasset
atp≤0.05.
Results
Inthepresentstudy,bloodcollectionforthemeasurement
ofleadlevelswasperformedfourtimesthroughoutaperiod
of 35 months; the results for each blood lead level (1st
to4th) are shown for each date of collection. The
num-berofparticipantsineachcollectionvaried,despiteefforts
toincludeallparticipantsoneachoccasion.Theestimated
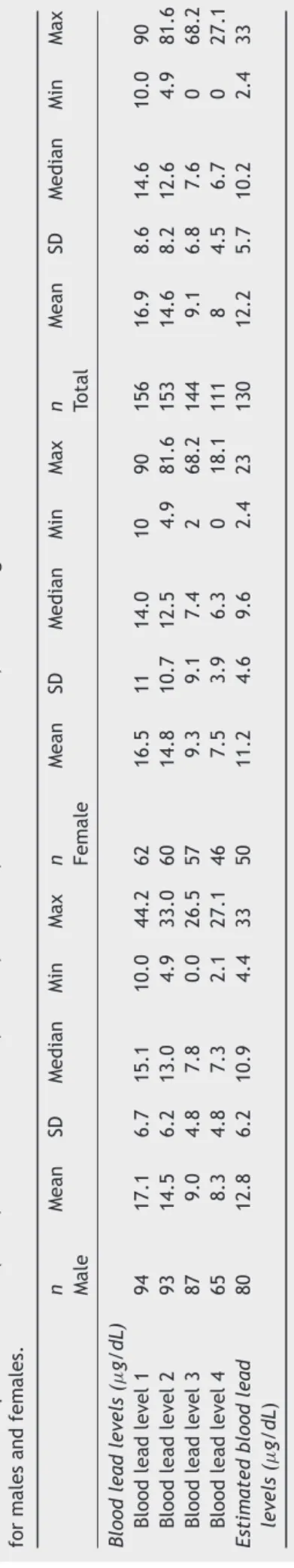
blood lead level was 12.2g/dL (±5.7g/dL SD, ranging
from2.4to33g/dLSD).Table1showstheresultsofthe
descriptivestatistical analysis (mean, standard deviation,
median,minimum,andmaximum values)ofleadlevelsin
bloodobtained duringthe periodofleadlevel monitoring
andestimatedbloodleadlevels.
Astronglinearcorrelationwasfoundbetweentheresults
ofthe initial leadlevel samples in blood andthe timeof
audiologicalassessment (Pearson’s r=0.78).The following
variableswereincludedineachsubsequentmodel:latency
of waves I, III, or V, age, gender, time of audiological
assessment,andestimatedbloodleadlevelsonthedateof
theaudiologicalassessment.Toinvestigatetheassociation
betweenlevelsofleadinbloodwiththeresultsofBAEP,the
absolutelatenciesofwavesI,III,andVwereincludedinthe
model(withwaveIIIlatencyadjustedforwaveIlatency,and
waveVlatencyadjustedforwaveIIIlatency).Theresultsof
theinitialregressionoftheabsolutelatencyofwaveIbyear
inrelationtoage,gender,cumulativebloodleadlevels,and
dateoftheaudiologicalassessmentwerenotsignificant.
Table2showsthefinalregressionmodelfortheabsolute
latencyofwavesIIIandV(variableswithnosignificant
asso-ciations were not included in the table). The association
betweenabsolute latenciesand gender wassignificant. In
thisstudy,malechildrenhadhigherlatenciesandinterpeak
intervals,with asignificant difference whencompared to
femalechildren.
Discussion
Concern about the effects of lead on health has led to
several experimental and clinical studies conducted with
industrialworkers,adults,andchildrenwithahistoryoflead
exposure.Leadlevelsinbloodreflectthedynamicbalance
betweenabsorption,retention,release,andeliminationof
thesubstance.Inlong-termexposure,thismarkerprovides
areliable indicator of current exposure,unless the
expo-suresvarywidely,inwhichcasethepreviousexposureswill
notbeaccuratelyreflected.
In the present study, the reduction in the blood lead
levelswas observed over time, and the last twosamples
showed that the mean level of lead in the blood of the
participantswaslowerthan10g/dL.Thedecreaseinthe
Table2 Resultsofthefinallinearregressionmodelmeasuringthebrainstemauditoryevokedpotential(absolutelatencyof wavesIIIandV),rightandleftears,formalescomparedtofemales.
WaveIII,in relationtowaveI
Coefficient Standarderror pvalue WaveV,inrelation towaveIII
Coefficient Standarderror p-Value
Constant 4.004 0.017 0.000 Constant 5.770 0.016 0.000
WaveIRE 0.583 0.071 0.000 WaveIIIRE 0.810 0.066 0.000
MaleRE 0.087 0.021 0.000 MaleRE 0.073 0.021 0.001
Constant 4.026 0.017 0.000 Constant 5.782 0.015 0.000
WaveILE 0.609 0.080 0.000 WaveIIILE 0.850 0.059 0.000
MaleLE 0.069 0.022 0.002 MaleLE 0.084 0.019 0.000
RE,rightear;LE,leftear.
blood leadlevelsinthestudied childrenprobablyreflects
theimpactofthemeasurestakenbythemunicipal
govern-mentandthecompanyresponsibleforthecontaminationof
theareaaroundthefactory.Eventhoughthestudywasstill
inprogress,theparticipantsweretreatedduetolead
poi-soning,streets andpublicspacesnear thebattery factory
werepaved,andtheaffectedpopulationreceived
informa-tionto avoid theconsumption of localproducts from the
communitygarden,aswellasotherinformationrelatedto
generalhealth.
Althoughstudiesconductedwithworkersexposedtolead
showconsistent resultsthatindicateauditoryeffects, the
data on the effects of environmental lead exposure on
theauditory systemof childrenarecontradictory.13,25---28,31
Schwartz and Otto10 suggested that evoked potentials
can provide an early indication of lead contamination
whencomparedtobehavioralprocedures,but subsequent
studies,27,28,31 including the present, study have failed to
confirmthatassertion.
The present study showed noassociation between low
levels of lead in blood and the absolute latencies and
interpeakintervalvaluesobtainedintheBAEPassessmentof
childrencontaminatedbylead.Theregressionmodelusedin
thestudy(whichincludedthevariablesage,estimatedblood
leadlevels,thetimebetweenaudiologicalassessmentand
collectionofblood,andgenderofparticipants)showedthat
theonly significantassociation wasobservedbetween the
absolutelatenciesofwavesIIIandVandgender(Table2).
Themalechildrendemonstratedsignificantlyhigher
laten-ciesandinterpeakintervals comparedtofemales, butthe
changeswerenotrelatedtolevelsofleadinblood.
TheassociationbetweentheabsolutelatenciesofBAEP
and gender was expected, as the literature reports that
BAEPsareaffectedby genderandage, duetoanatomical
andfunctionaldifferencesbetweengenders.Thisfindingis
importantconsideringthatalthoughsomestudiesevaluated
acontrolgroupmatchedtothetestgroupinrelationto
gen-derandage,thestatisticalmodelfrequentlyusedtoshow
thepresenceofadose---effectassociationdidnot
necessar-ily considerthe influenceof theseindependent variables.
Inone suchstudy,for instance, whichreportedan
associ-ationbetweenauditoryfunctionandtheblood leadlevel,
adecrease in thecorrelation wasdemonstratedafter the
inclusionofageandgenderinthestatisticalanalysis.18
Thus, unlike the results described by other
investigators,13,24---26 the present study found no
associ-ationbetweenBAEPandthecumulativebloodleadlevel,in
agreementwiththeresultsobtainedbyCounteretal.27,28,31
Leadlevelsin thepresentstudy werelowerthanthosein
theaforementionedstudies.Thethresholdandlead
poison-inglevelnecessary toinduce aneuropathology areyet to
beestablished.InascientificopinionoftheNordicExpert
Group on the association between occupational exposure
tochemicals and theincidenceof hearingloss, thegroup
definedtheno-observed-adverse-effectlevel(NOAEL) and
thelowest-observed-adverse-effectlevel(LOAEL)forlead,
inanimalsandhumans,specificallyregardingimpactonthe
auditorysystem.23ANOAELof35---40
g/dLandaLOAELof
55g/dLinbloodwereidentifiedinmonkeys.
In adult humans, the effects on the central auditory
system have been associated with current exposures and
weightedmeanlife-timeofleadconcentrationsinbloodof
approximately28---57g/dL,dataobtained mainlyin
stud-iesrelatedtooccupationalexposures.Inthepresentstudy,
alterationsinBAEPwereobservedinchildrenlivingin
lead-contaminated areas, with an estimated mean blood lead
levelsof12.2g/dL.
TheseresultssuggestthattheBAEPwasnotsensitiveto
assess the auditory system in children with low levels of
leadinblood.Neurotoxicityappearstobethepredominant
effectintheauditorysystemintheeventofleadexposure
(insteadof ototoxicity). Consideringthis perspective, the
recordingofotherevokedpotentialsorbehavioralteststo
investigatechangesincentralauditoryprocessingappearto
bemoresensitiveproceduresfortheearlyidentificationof
lead-induceddisorders,asdescribedintheliterature.
The association between lead exposure and the
cen-tralauditoryprocessingskills wasinvestigated1 andhigher
lead levels in blood in the prenatal, neonatal, and
post-natalperiodswereassociatedwithpoorercentralauditory
processingskills, asdemonstrated by a filtered word test
(screeningtestforauditoryprocessingdisorders[SCAN]).
However,thedichoticdigitstestandtheauditoryfusion
test---revised (AFTR)were usedtoevaluateasub-sample
of20childrenfromthepresentstudygroup,32asa
feasibil-itystudy.Thechildrencontaminatedbyleadshowedlower
performanceinrelationtoclinicalnormativedata;however,
therewasnocorrelationbetweenbloodleadlevelsand
audi-toryprocessingskills.ThelonglatencyauditoryevokedN2
potentialand cognitive P3were alsorecorded in another
sub-sampleof73children.33
The N2 potential latency increased with the
concen-tration of lead in blood (p=0.030), but no significant
inbloodandlatency(p=0.821)ortheamplitudeofP3
poten-tial(p=0.411).ConsideringthatN2potentialisendogenous
and is highly related to attention,34 this finding suggests
thatleadpoisoningmayberelatedtoattentiondeficit,as
previouslyreported.2Whiledifferentprotocolsof
audiolog-icalassessment wereperformed in individuals exposed to
solvents,35 furtherstudiesareneededtoidentifytheideal
procedurestoassesstheauditoryeffectsoflead
contami-nation.
Evidenceofeffectsofleadoncognition,including
intel-ligence quotient (IQ), led the Center for Disease Control
andPreventiontoestablishthelevelofleadinbloodabove
5g/dLasthepopulation-basedreferenceforchildren,
aim-ingatprimarypreventiveintervention.29
Disclaimer
Mention of any company or product does not constitute
endorsementbytheCentersforDiseaseControland
Preven-tion(CDC),National Institute for OccupationalSafetyand
Health(NIOSH).Thefindingsandconclusionsinthisreport
arethoseofthe authorsanddonot necessarilyrepresent
theviewsoftheNationalInstituteforOccupationalSafety
andHealth.
Conclusion
No association was observed between the wave
compo-nentsof theBAEP andthe estimated blood leadlevelsof
12.2g/dL(±5.7g/dL)inchildrenexposedtolead.BAEP
doesnotseemtobethemostsensitivemethodtoevaluate
childrenwithlowbloodleadlevels.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
References
1.DietrichKN,SuccopPA,BergerOG,KeithRW.Leadexposureand thecentralauditoryprocessingabilitiesandcognitive develop-mentofurbanchildren:theCincinnatileadstudycohortatage 5years.NeurotoxicolTeratol.1992;14:51---6.
2.BellingerD.Verylowleadexposuresandchildren’s neurodevel-opment.CurrOpinPediatr.2008;20:172---7.
3.Lanphear BP, Dietrich K, AuingerP,CoxC.Cognitivedeficits associatedwithbloodleadconcentrations<10microg/dLinUS childrenandadolescents.PublicHealthRep.2000;115:521---9.
4.Lanphear BP, Hornung R, Khoury J, Yolton K, Baghurst P, BellingerDC,etal.Low-levelenvironmentalleadexposureand children’sintellectualfunction:aninternationalpooled analy-sis.EnvironHealthPerspect.2005;113:894---9.
5.Canfield RL, Henderson CR, Cory-Slechta DA. Intellectual impairmentinchildrenwithbloodleadconcentrationsbelow 10microgramperdeciliter.NEnglJMed.2003;348:1517---26.
6.FinkelsteinY,MarkowitzME,RosenJF.Low-levellead-induced neurotoxicityinchildren:anupdateoncentralnervoussystem effects.BrainResRev.1998;27:168---76.
7.Department of Health and Human Services. 2020 Topics & Objectives. Environmental Health. Available from: http:// healthypeople.gov/2020/topicsobjectives2020/overview.aspx? topicId=12[updated10.04.13,accessed11.09.13].
8.Brasil Ministério do Trabalho. Artigo 24/GM, 29 de dezem-bro,1994.ProgramadeControleMédicodeSaúdeOcupacional [OccupationalHealth SurveillanceProgram].Diário Oficialda União1994;PoderExecutivo,Brasília,DF.
9.CDC, Centers for Disease Control and Prevention [Internet]. AdvisoryCommittee on Childhood LeadPoisoning Prevention Controland Prevention.Low levelleadexposure harms chil-dren:a renewedcallforprimary prevention;2012.Available from:http://www.cdc.gov/nceh/lead/acclpp/finaldocument 010412.pdf[accessed11.09.13].
10.SchwartzJ,OttoD.Bloodlead, hearingthresholds,and neu-robehavioraldevelopmentinchildrenandyouth.ArchEnviron Health.1987;42:153---60.
11.SeppalainenAM,HernbergS.Afollow-upstudyofnerve conduc-tion velocities in leadexposed workers. Neurobehav Toxicol Teratol.1982;4:721---3.
12.SingerR,ValciukasJA,LilisR.Leadexposureandnerve conduc-tionvelocity:thedifferentialtimecourseofsensoryandmotor nerveeffects.Neurotoxicology.1983;4:193---202.
13.HoldesteinY,PrattH,GoldsherM,RosenG, ShenhavR,Linn S,etal.Auditorybrainstemevokedpotentialsinasymptomatic lead-exposedsubjects.JLaryngolOtol.1986;100:1031---6.
14.LilleF,HazemannP,GarnierR,DallyS.Effectsofleadand mer-curyintoxicationsonevokedpotentials.JToxicolClinToxicol. 1988;26:103---16.
15.ArakiS,MurataK,YokoyamaK,UchidaE.Auditoryevent-related potential(P300)inrelationtoperipheralnerveconductionin workersexposedtolead,zinc,andcopper:effectsofleadon cognitivefunctionandcentralnervoussystem.AmJIndMed. 1992;21:539---47.
16.Discalzi GL, Capellaro F, Bottalo L, Fabbro D, Mocellini A. Auditorybrainstemevokedpotentials(BAEPs)inlead-exposure workers.Neurotoxicology.1992;13:207---9.
17.Kovala T, Matikainen E, Mannelin T, Erkkilä J, Riihimäki V, HanninenH,etal.Effectsoflowlevelexposuretoleadon neu-rophysiologicalfunctionsamonglead batteryworkers. Occup EnvironMed.1997;54:487---93.
18.ForstLS,Freels S,PerskyV.Occupational leadexposureand hearingloss.JOccupEnvironMed.1997;39:658---60.
19.FarahatTM,Abdel-RasoulGM,El-AssyAR,KandilSH,KabilMK. Hearingthresholdsofworkersinaprintingfacility.EnvironRes. 1997;73:189---92.
20.Araki S, Sato H, Yokoyama K, Murata K. Subclinical neuro-physiologicaleffectsoflead:areviewonperipheral,central andautonomicnervoussystemeffectsinleadworkers.AmJ IndMed.2000;37:193---204.
21.WuTN,ShenCY,LaiJS,GooCF,KoKN,ChiHY,etal.Effects oflead andnoise exposureson hearingability.Arch Environ Health.2000;55:109---14.
22.HwangYH, Chiang HY, Yen-Jean MC, Wang JD. The associa-tion between low levels of lead in blood and occupational noise-inducedhearinglossinsteelworkers.SciTotalEnviron. 2009;408:43---9.
23.JohnsonAC,MorataTC. Occupational exposureto chemicals andhearingimpairment.ArbeteOchHalsa.2010;44:177.
24.Otto D, Robinson G, Baumann S, Schroeder S, Mushak P, KleinbaumD,etal.5-Yearfollow-upstudyofchildrenwith low-to-moderateleadabsorption:electrophysiologicalevaluation. EnvironRes.1985;38:168---86.
25.Osman K, Pawlas K, Schutz A, Gazdzik M, Sokal JA, Vahter M.LeadexposureandhearingeffectsinchildreninKatowice, Poland.EnvironRes.1999;80:1---8.
26.ZouC,ZhaoZ,Tang L, ChenZ,DuL. Theeffectofleadon brainstemauditoryevokedpotentialsinchildren.ChinMedJ (Engl).2003;116:565---8.
28.Counter SA, Vahter M, Laurell G, Buchanan LH, Ortega F, Skerfving S.High lead exposure and auditorysensory-neural functioninAndeanchildren.EnvironHealthPerspect.1997;105: 522---6.
29.CDC,Centersfor DiseaseControl and Prevention.Preventing lead poisoning in young children; 1991. Available from:
http://www.cdc.gov/nceh/lead/Publications/books/plpyc/ contents.htm[accessed11.09.13].
30.ANSI.AmericanNationalStandardsInstitute.Maximum permis-sibleambientnoiselevelsforaudiometrictestrooms.NewYork, NY:AmericanNationalStandardInstitute,ANSI;1991.
31.CounterSA.Brainstemneuralconductionbiomarkersin lead-exposedchildrenofAndeanlead-glazeworkers.JOccupEnviron Med.2002;44:855---64.
32.MoraesTFD,Gonc¸alvesTS,SalvadorKK,LopesAC,Alvarenga KF,Feniman MR.Relationshipbetweenleadinthebloodand performance in the abilities from hearing process. Int Arch Otorhinolaryngol.2012;16:39---43.
33.AlvarengaKF,Bernardez-BragaGRA,ZuckiF,DuarteJL,Lopes AC,FenimanMR.Correlationanalysisofthelonglatency audi-toryevokedpotentialN2andcognitiveP3withtheleveloflead poisoninginchildren.IntArchOtorhinolaryngol.2013;17:41---6.
34.McPherson DL,BallachandaB.Middle and long latency audi-toryevokedpotentials.In:RoeserRJ,ValenteM,Hosford-Dunn H, editors. Audiology diagnosis New York: Thieme; 2000. p. 471---501.