www.jped.com.br
ORIGINAL
ARTICLE
Respiratory
allergy
to
moth:
the
importance
of
sensitization
to
Bombyx
mori
in
children
with
asthma
and
rhinitis
夽
,
夽夽
Laura
M.L.
Araujo
a,∗,
Nelson
A.
Rosário
Filho
b,
Carlos
A.
Riedi
caChildandAdolescentHealth,HospitaldeClínicas,UniversidadeFederaldoParaná(UFPR),Curitiba,PR,Brazil
bPediatrics,HeadoftheAllergyandPediatricImmunologyService,HospitaldeClínicas,UniversidadeFederaldoParaná(UFPR),
Curitiba,PR,Brazil
cPediatrics,HospitaldeClínicas,UniversidadeFederaldoParaná(UFPR),Curitiba,PR,Brazil
Received14June2013;accepted2August2013 Availableonline20December2013
KEYWORDS
Sensitization; Moth; Asthma; Rhinitis
Abstract
Objective: thisstudyaimedtoprepareasilkwormmoth(Bombyxmori)antigenicextractand toperformskinpricktestswiththisextractinpatientswithallergicrespiratorydiseases;to evaluateserumspecificimmunoglobulinE(IgE)toBombyxmoriusingImmunoCAP®systemand toreportthefrequencyofpositivitybetweenthetwomethodsandwithclinicaldata.
Methods: thiswas across-sectional study with 99 children andadolescents diagnosedwith asthmaand/orallergicrhinitis,whohadskinreactivitytoatleastoneofthesixaeroallergens tested.Clinicaldatawereevaluated:skinpricktestswithBombyxmoriin-houseextract,and totalandspecificIgEanalysisusingImmunoCAP®wereperformed.
Results: thefrequencyofBombyxmorispecificIgEwasfoundtobe52.5%and60%usingthe skinpricktestandImmunoCAP®,respectively.Anassociationbetweenapositiveskintestfor Bombyxmoriandthepresenceofallergicrhinitis,atopicdermatitis,andurticariawasobserved, butthesamewasnottrueforasthmaorallergicconjunctivitis.Therewasnorelationwiththe severityofasthmaorrhinitissymptoms.
Conclusions: ahighfrequencyofsensitizationtoBombyxmoriwasobservedinaselected pop-ulationofpatientswithrespiratoryallergic diseasesinthecityofCuritiba,stateofParaná, Brazil.Theextractpreparedfromthewingsofthismothspeciesiseffectiveindemonstrating thissensitivity.
©2013SociedadeBrasileiradePediatria.PublishedbyElsevierEditoraLtda.Allrightsreserved.
夽 Pleasecitethisarticleas:AraujoLM,RosárioFilhoNA,RiediCA.Respiratoryallergytomoth:theimportanceofsensitizationtoBombyx
moriinchildrenwithasthmaandrhinitis.JPediatr(RioJ).2014;90:176---81.
夽夽StudyconductedatAllergyandPediatricImmunologyService,HospitaldeClínicas,UniversidadeFederaldoParaná.
∗Correspondingauthor.
E-mail:[email protected](L.M.L.Araujo).
0021-7557/$–seefrontmatter©2013SociedadeBrasileiradePediatria.PublishedbyElsevierEditoraLtda.Allrightsreserved.
PALAVRAS-CHAVE
Sensibilizac¸ão; Mariposa; Asma; Rinite
Alergiarespiratóriaàmariposa:importânciadasensibilizac¸ãoàBombyxmoriem crianc¸ascomasmaerinite
Resumo
Objetivo: Prepararextratoantigênicodamariposadobicho-da-seda(Bombyxmori)erealizar testescutâneoscomesseextratoempacientescomdoenc¸asrespiratóriasalérgicas,avaliarIgE séricaespecíficaparaBombyxmoriusandoosistemaImmunoCAP®erelatarafrequênciade positividadeentreosdoismétodosecomdadosclínicos.
Métodos: Estudotransversalcom99 crianc¸aseadolescentescomdiagnóstico deasmae/ou rinite alérgica, que apresentaram reac¸ão cutânea apelo menos um dos seis aeroalérgenos testados. Osdadosclínicosforamavaliados;testescutâneoscomextratodeBombyxmorie análisedeIgEtotaleespecíficaporImmunoCAP®foramrealizados.
Resultados: AfrequênciadeIgEespecíficaparaBombyxmorifoide52,5%e60%, respectiva-mente,pelotestecutâneoeImmunoCAP®.Foiobservadaumaassociac¸ãoentreotestecutâneo positivoparaBombyxmorieapresenc¸aderinitealérgica,dermatiteatópicaeurticária,mas omesmonãoocorreuparaaasmaouconjuntivitealérgica.Nãohouverelac¸ãocomagravidade dossintomasdeasmaourinite.
Conclusões: Altafrequênciadesensibilizac¸ãoàBombyxmorifoiencontradaemumapopulac¸ão selecionadadepacientescomdoenc¸asalérgicasrespiratóriasnacidadedeCuritiba,estadodo Paraná,Brasil.Oextratopreparadoapartirdasasasdessaespéciedemariposaéeficazem demonstraressasensibilidade.
©2013SociedadeBrasileiradePediatria.PublicadoporElsevierEditoraLtda.Todososdireitos reservados.
Introduction
Sensitization toinhalant allergens is a risk factor for the
developmentofallergicdiseasessuchasasthmaand
rhini-tis.Knowledgeaboutsensitizingallergensandtheirdegree
of exposure in differentenvironments is essentialfor the
diagnosis and treatment of allergic respiratory diseases.
DermatophagoidespteronyssinusandBlomiatropicalismites
arethemainsensitizersforpatientsdiagnosedwithasthma
andallergicrhinitis.1,2
The participationofinsectsinallergicrespiratory reac-tionshasbeendiscussedfordecades.3Themostextensively
studied insect has been the cockroach, whose domestic
infestationis acauseofasthma andisconsidered tobea publichealthissue.4Therehavebeendescriptionsofasthma andrhinitistriggeredbyspeciesoffliesandmosquitoessuch asthemayflyandthecaddisfly.5,6Astudyconductedwith asthmaticpatientsinthecityofSãoPaulo,Brazil,evidenced positiveskinpricktest(SPT)withmosquitoextractin32.5% ofcases,andpositiveSPTwithmothextractin65%ofcases.7
There have been several reports of individuals who,
during the process of silk production, developed
respira-tory allergic diseases. While caringfor silkworm cocoons,
workers are exposed directly to their inhalant antigens,
presentfromtheselectiontothehatchingofcocoons,when
thereis contactwithdustfromthe moths’ wings.8 These
allergens can trigger asthma,9 rhinitis, and conjunctivitis symptoms.10
The silkworm moth has cross-reactivity with other
species of moths and butterflies; it has been shown that
patientswithrespiratoryallergicdiseasescandevelop symp-toms from environmental exposure totheir allergens.11,12 Concentrationsofmothantigensverifiedby
radioimmunoas-say in samples of dust in the external environment (not
home)fora periodof threeyearswerehigh andat levels
comparabletothoseofpollen.
Skin tests with moth extract in allergic patients had
45% reactivity in this population.13 In 1997, allergy skin tests in atopic children in the city of Curitiba, state of Paraná,Brazil,detected38.4%ofpositivitytomothextract
(1:20Heteroceraweight/volume),thesecondmostfrequent
aftertheDermatophagoidespteronyssinusmite(97.5%).It
wassuggestedthat the highrate of sensitizationtomoth
requiredabetterevaluationofitsclinicalrelevance.14 Theaimofthisstudywastodeterminethesensitivityto
BombyxmoribySPTusingsilkwormmothwingantigensand
specificserumimmunoglobulinE(IgE)inchildrendiagnosed withasthmaand/orallergicrhinitis.
Methods
Thiswasacross-sectionalstudywithnon-probabilistic
samp-lingof99childrenandadolescentsofbothgenderswitha
diagnosisof asthma and/orallergicrhinitis treatedat the
outpatientallergyclinic ofAllergyandPediatric
Immunol-ogyService, HospitaldeClínicas, UniversidadeFederaldo
Paraná, with positive SPT for at least one of the
fol-lowing antigens: Dermatophagoides pteronyssinus, Blomia
tropicalis, Blattella germanica, Lolium multiflorum, dog
epithelium,orcatepithelium.
Thediagnosisandclassificationofrespiratoryallergic
dis-eases(asthmaandrhinitis)followedtherecommendations
of the Global Initiative for Asthma (GINA)15 and Allergic
RhinitisanditsImpactonAsthma(ARIA),16respectively.
TheallergenextractofBombyxmoriwaspreparedfrom
thewings of this speciesusingthe following method:the
materialfromtheinsectwasmaceratedwithapestleina
ether.Forantigenextraction,the approximateamount of 2gof insect particles was placed in 20mL of cold sterile
buffer solution(PBS), mixedfor six minutes, andallowed
tostand for 48hours at 8◦C. The mixture wasthen
cen-trifugedfor15minutesat12,000rpm,andthesupernatant wasfilteredusingsterilepolyethersulfonefilters(Millipore
Express®PLUSMembrane Filters,USA)containingporesof
0.2micrometersindiameters.
After bacteriological sterility tests, the filtrate was
diluted in 50% glycerin, 1:20 (weight/volume). This final
concentration was chosen to allow for comparison with
theresultsof asimilarstudy conductedin1997, inwhich allergyskintestswereperformedwithallergenextract stan-dardizedat1:20fromHeterocerasp.moth(Hollister-Stier
Laboratories®,USA)inchildrendiagnosedwithasthmaand
allergicrhinitis.14 This concentrationof 1:20 is commonly
usedby manufacturersofglycerinated allergenic extracts
forSPT. The allergen extractwasstoredin bottleswitha dropperandkeptrefrigeratedat4◦C.17
AllsubjectsunderwenttheSPTwiththeextractprepared fromBombyxmoriaccordingtothefollowingtechnique:the volarsurfaceoftherightforearmwascleanedwith70%ethyl alcohol;then,adropoftheextractwasappliedontheskin andthelocationmarkedwithskinmarkerpen.Thepositive controlwasperformedwithhistamineataconcentrationof
10mg/mL(IPI/ASAC,Brazil)andthenegativecontrolwith
50%saline/glycerol(IPI/ASAC,Brazil)witha3-cmdistance
betweenthem.Thepuncturetechniquewasusedwitha
27-mmsterileneedleappliedsuperficiallyontheskinsurface atanangleof20◦.After15minutes,thereadingofthe
reac-tionwasperformedwiththeaidofamillimeter-scaleruler.
Thetestwasconsideredpositivewhenthemeanofthetwo
perpendiculardiametersofthepapulewas≥ 3mm.18
SerumsampleswereseparatedintoaliquotsinEppendorf
tubes at -80◦C until measurement of total IgE and
spe-cificIgEtoallergensfromBombyxmori,Dermatophagoides
pteronyssinus,Dermatophagoidesfarinae,Blomiatropicalis,
Blattella germanica, dog epithelium, and cat epithelium
throughtheImmunoCAP®method.Eachallergenwas
cova-lentlycoupledtoasolidphaseandreactedwithspecificIgE antibodiescontainedintheserumsampleofpatients.
In the case of Bombyx mori, the antigen coupled to
the solid phase is derived fromthe wings of this kind of
moth, andthe allergen components are notavailable for
thisinsect.Then,nonspecificantibodies wereremovedby
washingandanti-IgEantibodieswereadded,boundtothe
-galactosidase enzyme. After incubation, the antibodies
(anti-IgE-enzyme) that were not bound were removed by
a new washing process. The enzyme substrate contained
in the development solution was added to the reaction
medium.Thereactionwasthendiscontinuedbyaddingthe
stopsolution,andthefluorescencewasmeasured,whichis proportionaltotheamountofspecificIgEinthesample.The concentrationoftotalIgEwasexpressedinkU/Landforthe specificIgE,kUA/L.19 Values >0.7kUA/L wereconsidered positive.
TheresearchprojectwasapprovedbytheEthics
Commit-teeonHumanResearchoftheHC-UFPR;parentsorguardians signedtheinformedconsentandparticipantsolderthan12
yearsoldsignedthetermofagreement.
Forstatisticalanalysis,thesoftwareprogramStatistica (Statsoft®,USA)wasused.Measuresofcentraltendencyand
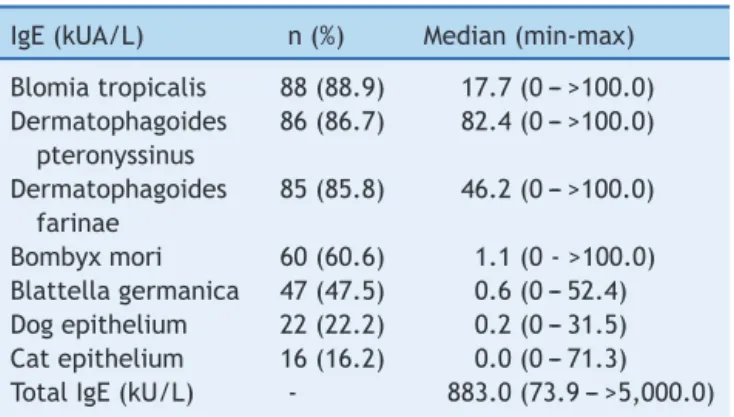
Table 1 Frequency ofpositivity tospecific IgEs through Immunocap®(>0.7kUA/L)andserumlevelsofspecificand totalIgEs.
IgE(kUA/L) n(%) Median(min-max)
Blomiatropicalis 88(88.9) 17.7(0--->100.0) Dermatophagoides
pteronyssinus
86(86.7) 82.4(0--->100.0)
Dermatophagoides farinae
85(85.8) 46.2(0--->100.0)
Bombyxmori 60(60.6) 1.1(0->100.0) Blattellagermanica 47(47.5) 0.6(0--- 52.4) Dogepithelium 22(22.2) 0.2(0--- 31.5) Catepithelium 16(16.2) 0.0(0---71.3) TotalIgE(kU/L) - 883.0(73.9--->5,000.0)
dispersionwereexpressedasmedian,minimumand
maxi-mum values.The nonparametric Mann-Whitney’s test was
usedtoestimatethedifferenceofvariableswithasymmetric
distribution.Theestimateddifferencebetweencategorical
variableswasperformedbyFisher’sexacttestandthe
chi-squared test.The univariatelogisticregressionmodelwas
usedtoestimatetheprobabilityofSPTpositivityaccording
tothelevelsofspecificIgEtoBombyxmori.Aminimumlevel
ofsignificanceof5%andaminimumtestpowerof85%were
consideredforalltests.
Results
Themeanageofpatientswas10.4+2.2yearswitharange
of6-15years(95%CI:9.9-10.8)and61(61.6%)weremales.
Eighty-sevenpatientshadasthma(87.9%)and95had(95.9%)
allergicrhinitis;12(12.1%)hadonlyrhinitis,andfour(4.0%)
hadonlyasthma.
Among patients with asthma, 31 (35.6%) were
classi-fiedashavingtheintermittentorpersistentmildform,46
(52.9%)hadpersistentmoderate,andten(11.5%)had
per-sistentsevereasthma.Amongpatientswithallergicrhinitis,
24(25.3%)hadthemildform(intermittentorpersistentmild
rhinitis)and71(74.7%)hadpersistentmoderate/severe.
Eyesymptoms(eyepruritus,tearing,conjunctival
hyper-emia, or congestion) were reportedby 66.7% of patients.
Amongother allergicdiseases,atopicdermatitiswas
diag-nosedinnine(9.1%)patientsandurticariainsix(6.1%).
Apositive reactionat SPT tothe Bombyxmori extract
was observed in 52 patients (52.5%), the second in
fre-quencyafterdustmites;Dermatophagoidespteronyssinus,
82 (82.8%), and Blomia tropicalis, 69 (69.7%). The other
observed reactions were to cockroach (Blattella
german-ica),dogepithelium,ryegrass(Loliummultiflorum)pollen,
andcatepitheliumwith17.2%,16.2%,15.1%,and12.1%of
positiveSPTs,respectively.
Table 1 shows the frequency of positive ImmunoCAP® testsusingacutoffof0.7kUA/L;thelevelsofspecificIgE
andtotalIgEwereexpressedasmedians(minimumand
max-imumvalues).
Univariatelogisticregressionanalysisshowedagood pos-itiveassociation betweenSPTand levelsof specificIgEto
Table2 AssociationbetweenSPTpositivityforBombyxmoriandotherskintests(n=99).
Allergens SPTBOMBYX+
n=52(%)
SPTBOMBYX ---n=47(%)
p
Dermatophagoidespteronyssinus 43(82.7) 39(83.0) 1.00
Blomiatropicalis 36(69.2) 33(70.2) 1.00
Blattellagermanica 08(15.4) 09(19.1) 0.79
Loliummultiflorum 09(17.3) 06(12.8) 0.58
Dogepithelium 11(21.1) 05(10.6) 0.18
Catepithelium 08(15.4) 04(8.5) 0.36
Fisher’sexacttest.
Table3 SerumlevelsoftotalandspecificIgEaccordingtoSPTpositivityforBombyxmori(n=99).
IgE(kUA/L) SPT+
(n=52)
SPT ---(n=47)
p
Dermatophagoidespteronyssinus 88.8(0.0---146.0) 62.6(0.0---144.0) 0.75 Dermatophagoidesfarinae 50.3(0.0---127.0) 36.2(0.0---126.0) 0.77
Blomiatropicalis 24.2(0.0---113.0) 11.9(0.0---111.0) 0.07
Blattellagermanica 1.0(0.0---52.4) 0.3(0.0---37.3) 0.002
Dogepithelium 0.2(0.0---6.3) 0.2(0.0---31.5) 0.13
Catepithelium 0.0(0.0---71.3) 0.0(0.0---32.5) 0.14
Bombyxmori 3.1(0.0---100.0) 0.4(0.0---83.0) <0.001
TotalIgE(kU/L) 1107.0(73.9---5,000.0) 772.0(93.0---5,000.0) 0.13
Mann-Whitney’stest
Table4 FrequencyofpositivityforspecificIgEs(>0.7kUA/L)accordingtoSPTpositivityforBombyxmori(n=99).
Allergens SPT+
n=52(%)
SPT ---n=47(%)
p
Dermatophagoidespteronyssinus 45(52.3) 41(47.7) 1.00
Dermatophagoidesfarinae 45(52.9) 40(47.1) 1.00
Blomiatropicalis 48(54.5) 40(45.4) 0.34
Blattellagermanica 30(63.8) 17(36.2) 0.04
Dogepithelium 16(72.7) 06(27.3) 0.05
Catepithelium 11(68.7) 05(31.2) 0.18
Bombyxmori 39(65.0) 21(35.0) 0.003
Fisher’sexacttest.
TherewasanassociationbetweenSPTpositivityfor
Bom-byx mori and the presence of allergic rhinitis (p=0.04),
atopicdermatitis (p=0.03), and urticaria(p=0.02).
How-ever,therewasnoassociationwiththepresenceofasthma
(p=0.36)andeyesymptoms(p=0.14).Likewise,therewas
nodifferenceintheseverityofasthma(p=0.73)orallergic
rhinitissymptoms(p=0.37).
RegardingthefrequencyofpositivityforBombyxmoriin
relationtoother SPTs,therewasnosignificantassociation
(Table2).
TheanalysisofserumlevelsoftotalandspecificIgE
anti-bodies in relation to the SPT for Bombyx mori tended to
behigherinpatientssensitizedtothemoth.However,only
the serum levels of specific IgE to cockroach and to the
mothweresignificantlyelevatedinthesepatientswhowere positiveattheSPTforBombyxmori(Table3).
There wasasignificantly higherfrequencyof positivity ofspecificIgEstodogepithelium,Blattellagermanica,and Bombyxmori(Table4).
Discussion
Theprevalenceofrespiratoryallergicdiseaseshasincreased
worldwide in the last decades.20,21 In Brazil, the same
trendofincreaseinasthmaandrhinitissymptomshasbeen
observed.22 Knowledge about sensitizing aeroallergens is
essentialfordiagnosis,treatment,andprevention.2
Dust mites are the main allergenic sources in patients
with asthma and allergic rhinitis.23,24 The importance of insectinhalantantigenshasbeendiscussedfordecades.3,7In
thisstudy,themothwasthesecond mostfrequent
aeroal-lergenregardingpositivityverifiedbySPTandspecificIgE serumlevels,suggestingthatthisinsectshouldbe consid-eredasensitizingagentforpatientswithasthmaandallergic rhinitis.
Thestudyparticipantswerenotsilkindustryworkers,and
therefore had nooccupational exposure to Bombyx mori.
Kinoand Oshima evaluatedasthmatic patients at random
radioallergosorbenttest(RAST)intheseraofoveronethird ofcases.Theauthorsconcludedthattheinsectsareeasily attractedtotheartificiallightofhouseholdsandmaycause sensitizationandrespiratoryallergysymptoms.[11]
A group of asthmatics with nohistory of occupational
exposure(n=50)showedafrequencyofpositivityattheSPT forBombyxmoriof68%,12higherthanthatobservedinthe presentpatients(52.5%);bothstudiesusedantigenicextract preparedfromthewingsofmoths.Thisdifferencemayhave
occurredbecausethe populationwasstudied inJapan, in
smallernumbers,consistedofadults,andhaddifferentlife
habitsandexposuretodifferentclimateandenvironment.
ThisstudyshowedanassociationbetweenSPTpositivity
forBombyxmori andthecorrespondingspecificserumIgE
byImmunoCAP®,whichdemonstratestheeffectivenessof
theextractmadetotestsensitizationtomoth.Moreover,in patientswithpositiveSPT, levelsofspecificIgEtoBombyx moriwerehigher.
WhenevaluatingthefrequencyofpositivitytospecificIgE formothaccordingtotheseverityofallergicrhinitis,itwas
observedthatpatientswithmoreseverediseasehadmore
positiveserumspecificIgEsformothbytheRASTmethod,25 differentlyfromthatfoundforthepopulationofthepresent study,inwhichtherewasmorepositivityattheSPTfor
Bom-byxmori in patients withrhinitis; however, therewas no
correlationwithsymptomseverity.Thisdifferencemaybe
explainedbythedifferentmethodsusedtodetectspecific
IgEandbythe highernumberof participantsin thestudy
conductedinJapan.
Althoughtheassessedpopulationofpatientswithatopic
dermatitis and urticaria was small, more dermatological
allergicdiseaseswerediagnosedinthosereactivetomoth.
This couldbe exploredin the future, asthere have been
fewreportsofallergicandirritantreactionsintheskinafter contactwithmoths.26,27
Itisknownthatthereiscross-reactivitybetweeninsect allergens.ItwasdemonstratedbyRAST-inhibitionassaythat thereis cross-reactivity between similar species, such as butterflyand moth,12 but alsobetween different species, suchasmothandmosquito.28
The molecularallergytechniquesallowed forthe
iden-tification of the major allergen of Bombyx mori larvae
(Bom m 1), which is constituted by arginine kinase
pro-tein and displays cross-reactivity with arginine kinase
fromthe cockroach.29 In the study,it was observed that
patients reactive to Bombyx mori at the SPT showed
a higher frequency of positivity and higher levels of
serum specific IgEs for Blattella germanica, which could
beexplainedby thecross-reactivity between them.
How-ever,thesamedidnotoccurwhencomparingthepositive
skin reactions between moth and cockroach, but the
immunochemical analysis of these allergens wasnot
per-formed.
Moreover,inhibition testswithextracts frommothand
mitesshoweddifferencesbetweentheirantibodies,
demon-stratingthatthereisnocross-reactivitybetweenthem.12In
thestudy,therewasnoassociationbetweenthefrequency
of skin reactivity at the SPT to mite and moth antigens
(Dermatophagoides pteronyssinus and Blomia tropicalis).
Similarly, there was no association between positivity at
the SPT for Bombyx mori and the presence of specific
serum IgE to mites (Dermatophagoides pteronyssinus,
Dermatophagoidesfarinae,andBlomiatropicalis),probably
becausethereisnocross-reactivitybetweenthem.
Onestudyfrom Chinaidentifiedanotherallergen
com-ponentofBombyxmori(Bomm7),alsoobtainedfromthe
larvaeofthisinsect,butconsistingoftropomyosinprotein.30 It isconsidered a pan-allergen,abletoshow broad cross-reactivitywithcomponentsofotherinsectssuchasDerp10 (fromtheDermatophagoidespteronyssinusmite)andBlag7 (fromtheBlattellagermanicacockroach).31,32Therefore,to verifytrue allergicsensitizationor cross-reactivity,future
studies should be performed, based on molecular allergy
diagnosis, butusingallergenic componentsof theBombyx
mori moth and notof itslarvae as acause of respiratory
allergicsymptoms.33,34
Thisstudywasthefirstonsensitizationtothesilkworm
moth performed in Brazil, and showedthe importance of
Bombyxmoriasasensitizingallergeninchildrenand adoles-cents diagnosedwithallergic respiratorydiseases(asthma and/or rhinitis).Ahighfrequencyof sensitizationto
Bom-byx mori wasfound in patients evaluatedby SPTwithan
extract prepared from the wings of moths; these results
wereconfirmedbyImmunoCAP®,awell-establishedmethod
fordetectionofspecificserumIgE.
Theidentificationofthisaeroallergen(moth),together withtheother groupsthat composetheprofile ofallergic sensitizationinthispopulation,shouldmaketheirtreatment moreefficient,asitwillallow foradjustmentsin environ-mentalcontrolprocedures,aswellasprovidenewspecific
immunotherapyoptionsinthefuture.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
References
1.CustovicA,SimpsonA.Environmentalallergenexposure, sen-sitisationandasthma:fromwholepopulationtoindividualsat risk.Thorax.2004;59:825---7.
2.DutraBM,Rosário NA,Zavadniak AF.Alérgenos inaláveisem Curitiba:umarevisãodesuarelevânciaclínica.RevBrasAlerg Imunopatol.2001;24:189---95.
3.PerlmanF.Insectsasinhalantallergens.JAllergyClinImmunol. 1958;29:302---28.
4.Arruda KL, Vailes LD, Ferriani VP, Santos AB, Pommés A, ChapmanMD.Cockroachallergensandasthma.JAllergyClin Immunol.2001;107:419---28.
5.FeinbergAR,FeinbergSM,Benaim-PintoC.Asthmaandrhinitis frominsectallergens.JAllergy.1956;27:437---44.
6.KinoT, ChiharaJ, Fukuda K, Sasaki Y, Shogaki Y, Oshima S. Allergy to insects in Japan III High frequency of IgE anti-body responses to insects (moth, butterfly, caddis fly, and chironomid)inpatientswithbronchialasthma immunochem-ical quantitation of the insect related airborne particles smaller than 10m in diameter. J Allergy Clin Immunol. 1987;79:857---66.
7.MendesE,LacazCS.Alergiaainsetos.In:Alergianasregiões tropicais.SãoPaulo:EditoraUniversidadedeSãoPaulo;1965. p.89.
9.InasawaM,HorikoshiK,TomiokaS.Astudyofbronchialasthma relatedtosilkwormculturing.JapanJAllergol.1973;22:142.
10.KobayashiS.Occupationalasthmaduetoinhalationof pharma-cologicaldustsandotherchemicalagentswithsomereference tootheroccupationalasthmasinJapan.Allergology. Amster-dan:ExcerptaMedica;1974.p.53.
11.KinoT,OshimaS.AllergytoinsectsinJapanI.Thereaginic sen-sitivitytomothandbutterflyinpatientswithbronchialasthma. JAllergyClinImmunol.1978;61:10---6.
12.KinoT,OshimaS.AllergytoinsectsinJapanII.Thereaginic sen-sitivitytosilkworminpatientswithbronchialasthma.JAllergy ClinImmunol.1979;64:131---8.
13.WynnSR,SwansonMC,ReedCE,PennyND,ShowersWB,Smith JM.Immunochemicalquantitation,sizedistribution,and cross-reactivityoflepdoptera(moth)aeroallergensinsoutheastern Minnesota.JAllergyClinImmunol.1988;82:47---54.
14.RosárioNA,VilelaMM.Quantitativeskinpricktestsandserum IgE antibodies in atopic asthmatics. J Invest Allergol Clin Immunol.1997;7:40---5.
15.Global Iniciative for Asthma --- GINA. Bethesda: Global
Ini-ciative for Asthma. Global Strategy for Asthma Treatment
and Prevention, 2006. [cited 2012 Feb 10]. Available from:
http://www.ginasthma.org/pdf/GINAReport2010.pdf
16.BousquetJ,KhaltaevN,CruzAA,DenburgJ,FokkensWJ,Togias A,etal.AllergicRhinitisanditsImpactonAsthma(ARIA)2008. Allergy.2008;63:8---160.
17.LierlMB,RiordanMM,FischerTJ.Prevalenceofinsect allergen-specificIgEinallergicasthmaticchildreninCincinnati.OhioAnn Allergy.1994;72:45---50.
18.NelsonHS.InvivotestingforimmunoglobulinE-mediated sensi-tivity.In:LeungDYM,SampsonHA,GehaR,SzeflerSJ,editors. Pediatricallergy-principlesandpractice.2nded.Oxford: Else-vier;2011.p.250-8.
19.Método de avaliac¸ão de IgE específica e total ---
Immuno-CAP PHADIA®. [cited 2012 Feb 12]. Available from:
http://www.phadia.com
20.Worldwide variation in prevalence of symptoms of asthma, allergic rhinoconjunctivitis and athopic eczema: ISAAC. The InternationalStudyofAsthmaandAllergiesinChildhood(ISAAC) SteeringCommitteeLancet.1998;351:1225---32.
21.LaiCK,BeasleyR,CranJ,FoliaskiS,ShahJ,WeilandS. Inter-national Study of Asthma and Allergies in Childhood Phase Three Study Group Global variation in the prevalence and severityofasthmasymptoms:phasethreeoftheInternational StudyofAsthma and Allergiesin Childhood(ISAAC). Thorax. 2009;64:476---83.
22.Solé D,WandalsenGF,Camelo-Nunes IC,Naspitz CK. ISAAC---Brazilian Group Prevalence of symptoms of asthma, rhinitis and atopiceczemaamongBrazilianchildrenandadolescents identified bytheInternational StudyofAsthma andAllergies in Childhood (ISAAC)- Phase 3. J Pediatr (Rio J). 2006;82: 341---6.
23.NaspitzCK,SoléD,JacobCA,SarinhoE,SoaresFJ,DantasV, et al. Sensibilizac¸ãoa alérgenos inalantesealimentares em crianc¸asbrasileirasatópicas,peladeterminac¸ãoinvitrodeIgE totaleespecífica-ProjetoAlergia(PROAL).JPediatr(RioJ). 2004;80:203---10.
24.Fernandez-CaldasE,LockeyRF.Blomiatropicalis:amitewhose timehascome.Allergy.2004;59:1161---4.
25.SuzukiM,ItohH,SugiyamaK, TakagiI,NishimuraJ,Kato K, etal.CausativeallergensofallergicrhinitisinJapanwith spe-cial reference to silkworm moth allergen. Allergy. 1995;50: 23---7.
26.DinehartSM,ArcherME,WolfJE,McgravanMH,ReitzC,Smith EB.Caripitoitch:dermatitisfromcontactwithHylesiamoths. JAmericanAcademyDermatol.1985;13:743---7.
27.OoiPL,GohKT,LeeHS,GohCL.Tussockosis:anoutbreakof dermatitiscausedbytussockmothsinSingapore.Contact Der-matitis.1991;24:197---200.
28.KomaseY,SakataM,AzumaT,TanakaA,NakagawaT.IgE anti-bodiesagainstmidgeand mothfoundinJapanese asthmatic subjects and comparison ofallergenicitybetween these two insects.Allergy.1997;52:75---81.
29.LiuZ,XiaL,Wu Y,XiaQ,XenJ,RouxKH.Identification and characterizationofanargininkinaseasamajorallergenfrom silkworm(Bombyxmori)larvae.IntArchAllergyClinImmunol. 2008;50:8---14.
30.ZhongBX.Proteindatabankforseveraltissuesderivedfromfive instarofsilkworm.YiChuanXueBao.2001;28:217---24.
31.Aalberse RC. Allergens from mites: implications of cross-reactivity between invertebrate antigens. Allergy. 1998;53:47---8.
32.JeongKY,LeeJ,LeeIY,ReeHI,HongCS,YongTS.Allerginicityof recombinantBlag7.Germancockroachtropomyosin.Allergy. 2003;58:1059---63.
33.MariA.Whendoesaproteinbecomeanallergen?Searchingfor adynamicdefinitionbasedonmostadvancedtechnologytools. ClinExpAllergy.2008;38:1089---94.