Universidade
de
lÚvora
Departamento
de
Biologia
Mestrado
de
Biologia
e
Ecologia
do
Litoral
Mariúo
Effects of
field
metal
contamination
on
spsrm
motility
and
RNA/IINA
ratio
in two
echinoderm
species
Efeito
da
acumulnção
de
metais
pesados na
motilidade
do
esperma
e
no
índice
ARN/ADN
de populações de duas
espécies
de
equinodermes
Ana Isabel
dos
Ramos
Catarino
Orientação:
Dr.
Fhilippe Dubois (Université Libre
de
Bruxelles,
Bélgica)
Dr.
Hemique Nogueira
Cúral
(Faculdade
de
Ciências
daUniversidade
deLisboq
Portugal)
Ests dissertação
ruãoinclui
as
críticas
e sugestõesfeitas
pelo
júri
Universidade
de
Évora
Departamento
de
Biologia
Mestrado
de
Biologia
e
Ecologia
do
Litoral
Mariúo
Effects of
Íield
metal
contamination
on
sperm
motility
and
RNA/IINA
ratio in
two
echinoderm
species
Efeito
da
acumulação
de metais pesados na
motilidade
do
esperma
eno
Índice
ARN/ADN
de populações de duas
espécies
de
equinodermes
Ana Isabel
dos
Ramos
Catarino
,)tí4,1
A
Orientação:
Dr.
Philippe
Dubois (Université
Libre
deBruxelles,
Bélgica)
Dr.
Henrique Nogueira Cabral (Faculdade
de
Ciências
daUniversidade
deLisboa, Portugal)
Esta dissertação não inclwi ss
críticas
e sugestõesfeitas pelo
iúri
Este projecto teve o apoio da Fundação para a Ciência e Tecnologlq sendo a autora bolseira de mesffado: SFRII/B]p,{12138712005.
Parte
deste projecto
foi
apresentado
na
tY
Inturnational
Echinoderm
Confererrce,New
Hampshire,USA: Effects
of
Íield
metal
contamination on
sperm
motility and RNA:DNA ratio in
Ágqias
rubens (apresentação oral por Ana Catarino).Contents
AcrcvowtmGdENTs
2 4 AgSTRACT 5 Rnsunno INIRODUCTION 6 Rgrgnm.ICEs 9Effects
of field
metal contaminationon
speÍmmotility
andRNA/DNA
ratio
in
two
echinoderm
species..
... 13Fnqnr
coNsrDERATroNs
...41RprrxsNcEs...
-.-43Acrolowr,uncnanNTs
I
amthankful
to Dr.
Philippe
Duboisfor
acceptingthe
supervisionof
this
project for
the companionship,
for all
the suppoú,for
answeringall my
questions,for
believingin
my work,
for all
the patience.I
am most grateful for everything.I
am thankfulto Dr.
HenriqueCúral
for
also acceptingúe
supervisionof this projec!
for all
the support andall
the concernaboú
tJrework, for
replyingto all my
doubts and forall
the advice. Thank you very much.I
am thankfirl
to
Dr.
Michel
Jangoux,for
welcoming
me
in
the
Ivlarine Biology
LúoraÍory
of the FreeUniversity
of Brussels(ULB,
Belgium).I
am thankful to Cristina Nunes as she was thelink
that connected me to Brussels.I
am also thankful to all the people from the Marine Lab(ULB), for
welcoming me,for
all
your help
andyour
support.I
am most gratefulto
Thierry,
Viviane
andEdith,
to Catherine and Julie, toPhilipp
Pernetfor all
the precious helpwith
the work, to David,Pol
and Guillemettefor
answeringall
my
questions and alsofor work
support,to
Julie Simonfor all
the
companionship andfinally
to
Claire,
for
friendship, companionship and all the help.Merci
à tous!I
am very thankful
to
Dr.
Usha Punjabi and
Dr.
Kris
Peetersfrom the
Antwerp
University Hospital (UZA\ Belgium),
for
their
availabilrty
on helping me with
the CASA analysis.I
am alsoftankful to Dr.
Evelyne Bertrandfrom the
St-PierreHospital,
Brussels,for
helping me
with
the sperm quality analysis.I
am thankfulto
SergeAron
andMyriam
Heueftz for letting me use their lab facilities.I
am thankful to Benjamin Ciotti, Elaine Caldarone and Vanessa Fonseca for all the helpon
the RNAIDNA
ratios
determination andvery
useful
tips
about the nucleic
acids exfraction method.To all the anonymous, that somehow
I
have not mentioned, my appreciation.Words are
not
enoughto
thank
everybodythat
has helped mewith
this
project, who supported me and who believed in me. Thank you ever so much!Now
in Portuguese - Agora em português:Gostaria de agradeser ao gang de Bnrxelas, especialmente ao núcleo
duro!
Estou-vos eternamente g,iatapela companhi4amizade, apoio, alegria.... Foram indispensáveis! Até breve...Gostaria de agradecer a todos os meus amigos, espalhados pelos sete cantos do mundo,
que tanto
me
apoiaram,que tiveram
disponibilidade
e
paciência para
ouvir!
Um
agradecimento especial à Beta, à Joaninha e, cliaro está, à JuAna!
Muito
obrigada pela vossa amizade incondicional.Gostaria de agradecer à
miúa
tiaAdília
e família" por me terem recebido e pelo apoio.E
finalmente!
A
vocês,
"malta-lá-de-casa":Mãe
e
Pai, André
e
Cataina!
Foram excepcionais.Eu
sei
que possocontar
sempre com vocês,haja
o
que houver!Muito
obrigada, por tudo.
Arsrru.cr
Effes
of
field
metal contamination on sperm
motility
andRNA/DNA ratio in
two
echinoderm species
This
study aimedto
assess the effectsof field
metal contaminationon
spennmoülity
and
RNAIDNA
ratio
in two
populationsof
echinoderms; Asterias rubens and Echinus acutus. Thesespcies
occur naturally along
a
contaminationgradient
of
cadmium, copper,lmd
andzinc,
in
a Norwegianfiord (Sorfior$.
Spermmotility
was quantifiedwith
thehelp
of
a
computer assisted sperm analysis system(CASA).
TheRNA/DNA
ratios were assessed based on
a
l-dye, (Ethidium bromidell-enzyme
(RNase)96-well
microplate
fluorometric
assay.Although both
species appearedto
readily
accumulate metals, neither spermmotility
parameters inA.
rubens norRNAIDNA
in
the bodywall
of
both
spcies
were affected. TheRNAIDNA
ratiosin
A.
rubenspyloric
caeca weresignificantly higher
in
the
most
contaminatedstation.
Even thought
this fiord
is considered ahighly
contaminated place, theseppulations
seemto
tolerate these metal contamination levels.Rnsunno
Efeito da
acumulação
de
metais
pesadosna motilidade do
esperma
e no
índice
ARN/ÀDN
de populações de duas espécies de equinodermesEste estudo teve como objectivo
verificar
o efeito da acumulação de metais pesados namotilidade do
espermae na
razÃoARN/ADN
em
indivíduosde
duas populações de equinodermes: Asterias rubens e Echinus acutus. Estas espécies ocoffem natrnalmenteao longo de
um
gradientede
contaminação de cádmio, cobre, chumboe zinco,
numfiorde
norueguês (Sorfiord).A
motilidade do espermafoi
quantificada com oauxílio
deum
sistemainfomático
especializado(CASA).
A
raz-áoRNA/DNA
foi
determinada através de ensaiosutilizando
brometo de etídio como corante fluorescente. Embora se tenhaverificado
acumulaçãode
metais pesadosnos
tecidosdos indivíduos,
não
seregistaram diferenças significativas na motilidade do esperma e na razáa
ARN/ADN
do tegumentoem função
do
grau de
contaminaçãodo
ambiente. Apenasos
valores daraz,ão
ARN/ADN
do
cecopilórico
deA.
rubensforam
significativamente maiores na estaçãomais
contaminada.Estas
populaçõesparecem então
tolerar
os
níveis
de contaminantes existentes no Sor§ord.INTnouucrroN
Echinoderrrs are considered good environmental indicators, because they exhibit a
high
sensitivityto
environmental changes, havea
sedentaryway
of
life
and integratelocal
pllution
(Guillou
et
al.,
2000; Coteuret
al.,
2003a).In
particular,
it
has been shownthat they
accumulate metals asa function
of
the
contaminationlevel
of
the environment (e.g. Warnauet
aI.,
1998;Guillou
et
a1.,2000; Coternet al.,
2003b and references therein). Some are also consideredto
be key-species,playing a
structuringrole in
benthic ecosystems (Menge etal.,
L994). Therefore,ffiy
effectof
a contaminant on populations of these species may affect the whole community. In the last decades the useof
echinoderm,like
sea urchins andstarfis[
has been sftongly recommendedby
ecotoxicologists
(Dinnel
etal.,
1987,1988; 1,989; Coteur et al., 2003a) and international environmental agencies (e.g. USEPA'
1994_ Environment Canada,1997).The Ssrfiord,
locatedin
SouthwestNorway, holds at its
head threelarge
metalsmelters.
For
more
than
80
yeaÍs
their
waste
was
dischargedinto this
fiord,
contaminaüng severelyits
sedirnents and making the Sorfiord oneof
the most polluted fiords in the world. The dischargesonly
started to be reducedfrom
1986 on andin
L992 remedial actions also took place: the metal containing sludge started to be deposited intomountain
cavesand
sedimentsin
shallow water,
closeto
oneof
the
smelters, were cappedby
animprmeúle
membranein
orderto
limit
metal release (see Coteur et al., 2003b;Gillan
etal.,
2A05; Ruus et a1., 2006 and references therein).Although
remedial actions have been takenin
recent years and dischargesinto the
fiord
were
exÍremelyreducd
the Sorfiord
still
remainsone
of
the most
contarninated areasin
Europe (OSPAR Commission,2000).
A
steepmetal
contamination gradient occursfrom
the head towards the opening into the Flardangerfiord (Coteur et al. 2003b).In this
studytwo
echinoderm species, Asterias rubens and Echinus acutus,that
occtlr
all
along the Sorfiord, were chosen to assess heavy metal contamination effectsin
two
well
known biomarkers: speÍmmotility
andRNAIDNA
ratio.Experimental gamete exposure
to
metalsis
known
to
affect
echinodeÍms speÍm quality, theúility
of
sperm to successfullyfertilize
an egg, and embryogenesis (Warnauet
&1.,1996;
His
et
al.,
1999;
Larrain
et
&1., 1999).
Given that
sperm
motility
(progressive
movement)
is a
requirement
for
fertilization
and
is
correlated
with
fertilization
susoess,it
is
generally usedto
assess speÍmquality
(Kime
et
a1.,2001; Rurangwaet al.,
2004).
Spermmotility
can me
measuredeither
by an
observeror
automaticatly.
The
first
methodological
approachusually has
a
higher
degreeof
subjectivity associatedmainly
dueto
the choiceof
the observers,their
experience andtheir
interpretationof
movement. Computer assisted sperm analysis systems,known
asCASÀ
provide much more
accurate andreliable
results (Rurangwaet
ú.,2004). A
CASA
system refers to the physical equipment used tovísualae
anddigitize
static and dynamic sperm images, aswell
asto
the methods usedto
process and analyse them.A
simple
sprm
movement
video
analysis provides
standardizeresults
with
highreproducibility
(WHO,
1999;Kime
et
al.,
2A0|'
Rurangwaet
al.,
2004).The CASA
system has been extensively used
in
fish(Kime
et al., 200 I ; Rurangwaet
al. , 2004), butalso in ochinoderm sperm analysis
(Au et
a1.,2000:Au
et al., 2001;Lu
andWq
2005).In
laboratorial studiesit
has been demonstrated,with
the helpof
theCASA,
that echinoid sperrnmotility
decreasedwith
metal exposure, eitherfrom
spennitself (Au
et al., 2000)or
adults chronic contamination(Au et
a1.,2001). These results suggest that speÍmmotility
studies may be a good approachin
evaluating echinoderm reproductive impairment byfield
metal contamination.The
RNA/DNA ratio
has been
usedas
a
biochemical
growth rate
indicator,providing growth
rates
and
metabolic
status estimatesof
a
wide
range
of
marine organisms,for
instancelarval
Íishes(Buckley
et
al.,
L999), crustaceans (Rosa andNunes, 2003), molluscs
(Melzner
et
al.,
2005), corals
(Meesterset
al., 20A»
and echinoderms (Watts and Lawrence, 1990; Frantzis etal.,
1992; Liyana-Pathiranaet
al.,2002).
This
index has been provento
be sensitiveto
environmental changes (Meesters et al., ?002; Rosa and Nrures, 2003) and positively correlatedwith
protein synthesis and growth (Barron andAdelman,
1984; Wang andStickle,
1988; RosaandNunes,2003).
Higher growth
rates are
expected
to
eúance
total
ribonucleic
acid
(RI'{A)
concenfiations, hence the raüo of
RNA
to deoxyribonucleic acid(DNA).
TheRNAs
are necessaryfor
the
biosynthesísof
proteins and
their
number and
activity
varies
in
Íesponse
to
the requisitefor
protein
synthesis. TheDNA
levelsin
cells
remain rather constant and, thus, are used to normalize the measuredRNA
concentrations. Hence, theRNAENA
ratio is an index ofcell's
metabolic rate (Buckley et a1., 1999).Since
the
1970s thisratio
has been broadly used, although different methods have beendwelopd.
Thefirst
methodto
be used was the UV-based nucleicacid
analysis. However, now-a-daysmainly
spectrofluorometric nucleic acid analysis is used, which isconsidered a more sensitive and rapid method and as precise as the former one (Buckley
et
al.,
1999" Caldaroneet
al., 2001). The most commonly spectrofluorometric methodused employs
the
fluorophore
ethidium bromide
(EB:
3,8-diamino-6-phenyl-5-ethylphenanthridinium bromide),uúich
bindsto
nucleic acids,g1ing
a measureof
thetotal
sample amount of them. The enryme RNase is then added, in order to digestRNA,
being the remaining fluorescence attributed
to
DNA
and thusproüding
also a way to back calculate theRNA
amount @uckley et al,1999; Caldarone et al., 2001).Growth impainnent due
to
contaminant exposure has been evidenced by a declineof
this ratio
in
fish,
crustaceans and molluscs (Barron andAdelman,
1984; Wang andStickle,
1988;Miliou
etal.,
1998;Wo
et
al.,
1999;Adham,2002).
These datz strongly supportthe use of theRNAIDNA
ratioto
assess the effects of contamination on growth rate in echinodeÍms.In the recent years, an increasing emphasis has been dedicated to the evaluation
of
contamination effects in marine organisms. Several European andworldwide
legislative documents have introduced monitoring and environmental assessment studies as a wayof
diagnosis and remedial measures development. The present study aimedto
evaluatethe
effectsof
heavymetal
contaminationin
two
speciesof
echinodermsin
a heavily contaminatedford
in Norway,
and thusto
contributeto
a better understandingof
thislSSUe.
Rrc,rnnmgcn§
Adham,
K.G.
2AA2. Sublethal effects of aquaticpollution in
Lake Maryüt on theAfrican
sharptooth
catfish,
Clarias gariepinus
@trchell,
1822).
Journal
of
Applied
Ichthyolog
L8,87-94.
Au, D.W.T.,
ChiLnE,M.W.L.,
Wu,
R.S.S. 2000.Effects
of
cadmium and phenol onmotility
and
ultrastructureof
seaurchin
and
mussel spermatozoa.Archives
of
Env ironment
al
C ont aminat ion and Tox i col ogt
38, 45 5 463 .Au, D.W.T.,
Lee,C.Y.,
Chan,K.L.,
Wu, R.S.S.2001. Reproductiveimpairmentof
seaurchins upon chronic
exposureto
cadmium.PaÍt
I:
Effects
on
gamete qualtty. EnvironmentalPoll*ion
lll,
1-9Buckley,
L.,
Caldarone,E.,
Ong,
T.-L.
1999.RNA:DNA
ratio
and other
nucleic acid-based indicatorsfor
growth
andcondition
of
marine fishes.Hydrobiologia
40I,
265-277.Barroq
trrrÍ.G., Adelman,I.
1984. Nucleic acid, protein content, and growttr of larvalfish
sublethally
exposedto
various toxicants. Canadian
Journal
of
Fisheries and
Aquatic Sciences 41, 141-150.
Caldarone,
E.M.,
Wag[er,M.,
Onge-Burns, J.S., Buckley, L.J. 2A0L Protocol and gurdefor
estimating nucleic acidsin
larval fish
using a fluorescence microplats reader.u.s.
deparhnentof
commerce,Northeast Fisheries
ScienceCenter
Reference DoçttmenÍ, 0 1 - I I ; 22pp.Coteur,
G.,
Gosselin,P.,
Wantier,
P.,
Chambost-Iúanciet,Y.,
Danis,
8.,
Penret, P.,r#amar1
M.,
Dúois,
P.
2003a. EchinodeÍmsas bioindicators,
bioassays, and impact assessment toolsof
sediment-associated metals and PCBs inúe
North
Sea. Archtxes of Erwironmental Contamination andToxicologt
45,190-242.Coteur,
G", Perne! P., Gillan,
D.,
Joly, G.,
Maage,
4.,
Dubois,
P.
2003b. Field
contamination
of
the
starfi sh Asterias ruberzsby
metals.Part
1: short- andlong-term
accumulation
along
a
pollution
gradient.Erwironmental Toxicolog,t and
Chemistry 22, 2136-2144.
Dinnel, P.4., Link, J.M.,
Stober,Q,
1987.Improved
methodologyfor
a
sea urchin spermcell
bioassayfor
marine waters. Archives of Environmental Contamination andToxicologt
16, 23-32.Dinnel,
P.4.,
Pagano,G.G.,
Oshida,P.S.
1988.A
seaurchin test
systemfor
marine environmentalmonitoring.
In:
Burke,R.,
Mladenov, P., Lambert, P., Parsley, R. (Eds.), EchinodermBiology
-
Proceedingsof
the
6th
International Echinoderm Conference.A. A.
Balkema, Rotterdam, pp.6lL-619.
Dinnel,
P.4,, Link,
J.M,
Stober,
Q.J.,
Letounreau,
M.W.,
Roberts,
W.E.
1989. Comparativesensitivity
of
seaurchin
sperm bioassayto
metals
and pesticides. Archives of Environmental Contamination andToxicologt
18,748-755.Environment Canada. 1997. Méthode d'essai biologique: essai sur
la
fécondation chezles
échinides (oursins
verts
et
oursins plats).
Raport SPE
llRMlz7.
Ottawa, Environment Canada, 109pp.Frantzis,
A,,
Grémare,A.,
VétiorUG.
1992.Growth
rates andRNA: DNA
ratios
in
Paracentrotus
lividus
(Echinodennata: Echinoidea)fed on
benthic macrophytes.Joarnal
of Experimental MarineBiologt
andEcolog
156,125-138.Gillan, D.C.,
Danis,8.,
Pernet, Ph.,Joly,
G., Dubois Ph. 2005. Structureof
sediment-associatedmicrobial
communities along a heavy-metal contamination gradientin
úe
marine
environment.
Applied
And
Environmental
Microbiolog,t 7l(2),
679Ã9A.
Guillou,
M.,
Quiniou,
F.,
Huart, El.,
Pagano,G.
2000.
Comparisonof
embryonic developmentand metal
contaminationin
several populationsof
the
sea urchinSphaereehinus
granularis
(Lamarck) exposed
to
anthropogenic
pollution. Archives of Environmental Contaminntion andToxicologt
39,337-344.His, E.,
Heyvan5,
I., Geffar{
O.,
Montaudouin,
X.d.
1999.A
comparison between oyster (Crassostrea gigas) and sea urchin {Paracentrotusltvidus)
larval bioassays fortoxicological
studies. Water Research 33,fiA6-nl8.
Kime, D.E., Look, K.J.W.V.,
McAllister,
B.G., Huyskens, G.,Rurangw4 E.,
Ollevier, F. 2001. Computer-assisted sperm analysis(CASA)
as atool for
monitoring speÍmquality
in fish.
Comparative Biochemistry andPltysiologt
Part C:
Toxicologt
&
Pharmacolog
1 30, 425-433 .Lanaiq
A-,
Riveros,
4.,
Silv4
J., Bay-Schmit[
E.
1999.Toxicity
of
metals
and pesticides using the spermcell
bioassaywith
the
sea urchinArbacia
spatuligera.Bulletin
of Environmental Contamination andToxicologt
62,749-757.Liyana-Pathirana,
C.,
Shúidi,
F.,Whittick, A.,
Hooper,R.
2002.Effect
of
season andartificial diet on arrino
acids and nucleic acids
in
gonadsof
green sea urchinSnongilocentrotus
droebachiensis. Comparative BiochemistryAnd
Plrysiologt
A-Molecular And Inte grat ive Physiologt
133, 3 89-398.Lu,
X.Y.,
Wn,
R.S.S. 2005.UV
induces reactive oxygen species, damages sperm, andimpairs fertilisation
in
the
sea urchinAnthocidaris
crassispina.Marine Biologt
148, 51-57.
Meesters,
E.H., Nieuwland,
G.,
Duineveld, G.C.A.,
Koh
4.,
Bak, R.P.M.
2002.RNA/DNA ratios
of
scleractinian corals
suggest acclimatisatiorladaptationin
relation
to light
gradients andturbidity
regimes.Marine Ecologt
Progress Series 227,?3.3-239.Melzner,
F.,
Forsythe,J.W., Lee, P.G., Wood,
J.8.,
Piatkowski,
fJ.,
Clemmeseq
C. 2005. Estimating recent growth in thecútlefish
Sepiafficinalis:
are nucleic acid-basedindicators
for
growth and
condition
the
methodof
choice?Journal
of
Experimental Marine
Biolog
andEcologt
311,37-5L.Menge,
B.À,
Berlow,8.L.,
Blanchette,C.4.,
Navarrete, S.4., Yamada" S.B. 1994. The keystone species concept-
variation
in
interaction sfiengthin
a
roc§
intertidal hâbitat. Ecological luíonographs 64,249486.
Milior4
H.,
Zaboukas,
N.,
Moraitou-Apostolopoulou,
M.
1998.
Biochemicalcompsition, growth
and survival
of
the
guppy,
Poecilia reticulata,
üring
chronic
sublethal
exposure
to
cadmium. Archtves
of
Erwironmental Contamination and Toxicolog,t 35, 58-63.OSPAR Commission. 2000.
Quality
Status Report 2000, Regiontr
-
GreaterNorth
Sea.OSPAR Commission, London. 136 +
xiii
pp.Ros4
R,, Nunes,M.L.
2A0É. Seasonal changesin
nucleic acids, amino acids and protein contentin
juvenile
Norway
lobster (Nephrops norttegicus).Marine
Biologt
143,565-572.
Rurangwa,
E., Kime,
D,E., Ollevier, F.,
Nash, J.P. 2004. The measurementof
spennmotility
and factors affecting
spermquality
in
culturedfish.
Aquaculture
234,1-28.
Ruus,
À,
Norman,W.G.,
Maage,4.,
Skei,J.2006.
PCB-containingpaint
and plaster causedexteme
PCB-concentrationsin
biotafrom
the Sorfiord (Western Norway) -A
case study. MarinePollution Bulletin
52, 100-103.US EPA.
1994. Seanrchin, Arbacia
punctúata,
fertilization test method
1008.0. In: Short-term methodsfor
estimating the chronictoxicity of
effluents and receivingwaters
to
marine
and
esfuarine organisms.
2"d edition.
US
Environmental Protection Agency, report EPA/60014-911103. Cincinnati, OH, 300-340 pp.Wang
S.Y., Stickle,W.B.
1988. Biochemical composition of the blue crab Callinectes sapidu"s exposedto
the
water-solublefraction
of
crude oil.
Marine Biologt
98,
23-30.
Warnau,
M.,
Iaccarino,M.,
Biase,4.D.,
Temara,4.,
Jangoux,M.,
Dubois, P., Pagano,G.
1996. Spermiotoxicrtyand embryotoxicrty
of
heavymetals
in
the
echinoid Paracentrotus lividus, Erwironmental Toxicologt and Chemistry 15, L931-1936.Wanraq [!{.,
Bíondo,
R.,
Temara,4.,
Bouquegleau,J.-M.,
Jangoux,M.,
Dubois,
P.1998-
Distribution of healy
metalsin
the echinoid Paracentrotuslividus from
theMediterranean Posidonia oceanica ecosystem:
seasonal
and
geographical variations.Journal
of Sea Research 39,267-280.Watts, S.A., Lawrence,
J.M.
1990.The effect
of
reproductive state, temperature, andsaliniry
on
DNA
and
RNA
levels and
actiüties
of
metabolic
enrymesof
thepyloric
caÊcain the
sea stiarLuidia
clathrata
(Say).Pltysiological
Zoologt
63, I 196-12 15.WHO.
1999.WHO
laboratory manualfor
the examination of human semen andsperm-cervical
mucus interaction.4e Edition.
WorldHealth
Organization, Cambridge,106 pp.
Wo, K.T., Lam,
P.K.S.,
Wu,
R.S.S.
L999.A
comparisonof
growth
biomarkersfor
assessing sublethal effects
of
cadmium on a marine gastropod, Nassarius festivus.llariÍre
P ollut ion Bull etin
39, 165 -17 3 .Effects of
Íield
metal conúamination on sperm
mofility
and
RNA/DNA
ratio
in two
echinoderm
species
Ana
I.
Catarinol'2'*; HenriqueN.
Cabral3;Kris
Peetersa; PhilippePernef;
Usha Purf abia; Philippe Dubois2(Papr
submitted to the scientific journalÁquatic
Toxicologt, Elsevier)'Universidade de Evora (Portugal)
2lúoratoire
de Biologie Marine, Université Libre de
Brurelles
@elglum)3Faculdade
de Ciências da Universidade de Lisboa (Portugal)
aM
Laboratory, Antwerp Universlty Hospital(Jz{,Belgium)
*Corresponding author: Ana
Isúel
Catarinoaddress:
Laboratoire de Biologie MarineUniversité
Libre
deBnxelles,
Campus du Solbosch,CP 160/15, avenue F.D. Roosevelt 50, 1050 Bruxelles,
Belgium
Abstract
This
study
aimedto
assessthe
effectsof
field
metal
contaminationon
speÍmmotility
andRNAIDNA
ratio in
echinodenns.In
orderto
do so, populations af Asteriasrubens
andEchinus
acuttts,that
occur naturally
along
a
contaminationgradient
of
cadmium, copper, lead and
zinc,
in
a Norwegtanfiord
(Sorfiord),
were studied. Spermmotility,
a
measureof
spermquality,
was quantifred using acompúer
assisted spenn analysis system(CASA), The
following
motion
parametersof individual
speÍm cells were assessed:curvilinear
velocity (VCL),
sfraightline
velocity
(VSL),
average pathvelocity (VAP).
The
RNA/DNA
ratios were
assessed basedon
a
l-dye, (Ethidiun
bromide,
EByl-enzyme
(RNase)96-well
microplate fluorometric assay.Although
both species appearedto
readily accumulate metals, neither spermmotility
parameters in ,4. rubensnor RNA/DNA ratio
in
the body
wall
of
both
specieswere
affected.
TheRNAIDNA
raüos
in
A.
rubens
pyloric
caeÇawas
significantly
higher
in
the
most contaminated station. However,this
washighly
dependenton a
few
possible outliers and when these values were removedfrom
analysis, significant differences disappeared. Even thought theSor{ord is
considered ahighly
contaminated place, populations of 24.rubens amd E'. acutus seem to be able
to
survive under these conditions and possiblyto
reproduce.
Keywordsz Asterias rubens; Echinus acutus;
Sorfiord;
metal contamination;
spermmotility;
RNAIDNA
ratio1.
Introduction
Numerous echinoderms play
a
structuringrole
in
benthic ecosystems where they act as key-species (Mengeet
al., 1994). For that reason, any effectof
a contaminant on populationsof
these speciesmay affect the whole
community.It
is well
known
that echinoderms readily accumulatehealy
metals and effects from molecularto individual
levels have been documented
both
in
laboratory andfield
conditions (Temaraet
al., 1997c; Warnau etaI.,
1998; den Besten etal.,
2001; Coteur et a1.,2003a,c; Danis et al.,20Aq. Exprimental
exposuresprovide
readily
interpretableresults and
üustworthy cause-effect relationships betweenmetal
contamination and impactson
echinoderms,namely
on
reproduction, steroid
metabolism,skeletal growth and immune function
(Voogt et
al.,
1987; den Besten et a1.,1989,l99l;
Temaraet
al., t997b;Au et
a1.,2001;Coteur
et al.,
2401,2005). Effects
of
metals
in
field
conüaminated populations are diverse.On the
one
hand
alkaline
phosphataseactívity
in
digestive
organsmay
besignificantly
reduced (Temara
et
a.1.,l997a,c),
the
immune system
can
be
either depressedor
stimulated (Coteur
eÍ
al.,
2003a,b)and
impaired
developmentof
theoffspring
have been relatedto
metal
contaminationof
the
progenitors(Guillou
et
al.,2000). On the other
han{
populationsof
starfish canmaintain
stable populationsin
heavily
contaminated environmentwith no
apparent effectson
eggs andfertiliration
rates (Temara et a1., 1998; Dubois et al., 2004).
In
the present study the effectsof
metalsin field
contaminated populationsof
the starfish Asterias rubens were assessed usingtwo
sensitive physiologlcal aspects that arecrucial
for
the maintenanceof
populations: spermmotility
andprotein
synthesis, thus growth. The latter was also investigated in the sympatric sea urchin Echinus actrtus.Sprm
motility
(progressive movement)is
a
requirementfor
fertilization
and is correlatedwith
fertilization
success.It
is
generally usedto
assess speÍmquality
(Au
et aL,ZAAl; Rurangwa et al., 2A0q. Furthermore,invitro
exposureof
gametes úo metals isknown
to
affect
spermquahty
(Au
et
al.,
200I)
and
embryogenesis (Warnauet
al.,1996)
of
echinoderms,a fact
that hasled
several enüronmental agenciesto
use these assaysas
standard procedures(e.g.
US EPA,
1994; Environment
Canada, 1997). However,the
effectsof field
metal contaminationon
echinoderm sperm qualrty have never been investigatedThe RNA/DNA ratio
hasbeen used as
a
biochemical
growth rate
indicator,proüding growth
rates
and
metabolic
status estimatesof
a
wide
range
of
marineorganisms,
for
instancefish
@arron andAdelman,
1984; Adham, 20A2; Fonseca et al.,2006),
cnrstaceans(Wang
and
Stickle, 1988), molluscs
(Melzrer
et
al.,
2005), cnidarians (Meesterset
a1.,2002) and echinoderms (Watts and Lawrence, 1990; Frantziset
al.,
1992; Liyana-Pathiranaet
al.,
2A0». The
use
of úis
ratio
is
basedon
the assumption that the amountof DNA' in
a given organism, isrelatively
constant under changing environmental conditions, sinceit
is mainly
a functionof
cell
number,whilst
total
RNA, which
is afimction
of ribosome number, is positively correlated urith protein synthesis, andthus,
$owth. Metal
exposuÍes resulted,in
numerous invertebrates andfish,
in
a decreaseof
RNA/DNA
raüo(Barron
and Adelman, 1984; Wang and Stickle, 1988; Adham,2A0».
The
preserúwork was
carried
out
in
the
Ssrfior{ a
southernbranch
of
theHardangerfior4
locatedin
WesternNorway.
Three smelters,built
at the
headof
theflord,
have dischargedtheir
wastesinto
it
for
more than 80 years, generating a hot spotof
metal contamination (see Temaraet
al.
1998 and references therein).
Inputs weresignificantly
reducedin
1986 andfollowed
by
remediation actions (see Coteuret
al.,2003c;
Gillan
et
al., 2005
an
referencesthere
in).
However, metals are
trappedin
sediments
of
the
fiord, limiting
netefflux to
the
open sea, butallowing
some internalrecycling.
The
release
of
these
sediment-assoçiatedmetals
is
significant and
is stimulatedby
theactivity
of
the benthos (see Temara et a1., 1998 and references there in). Populationsofr4.
rubens and E. acutus, occur naturally all along thefiord,
includingin
the most contiaminated sites.In
these sites,Á.
rubensis
effectively contaminatedby
metals (Coteur et al 2003c).
Metal
contaminationof
E'. acutusinthe
Sorfiord has never been assessed.2.
Materials
andMethods
2.1 Study
Area
andSampling
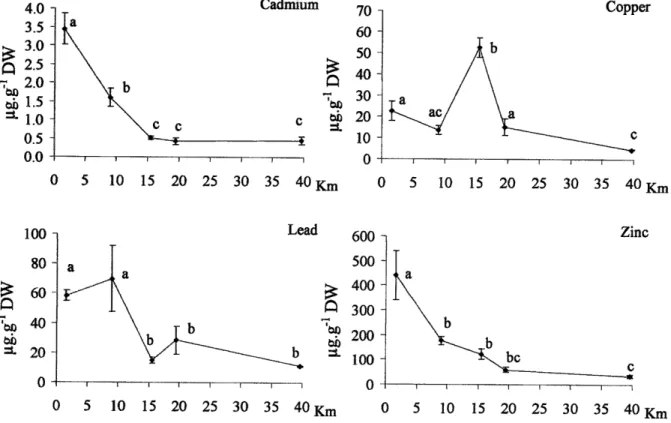
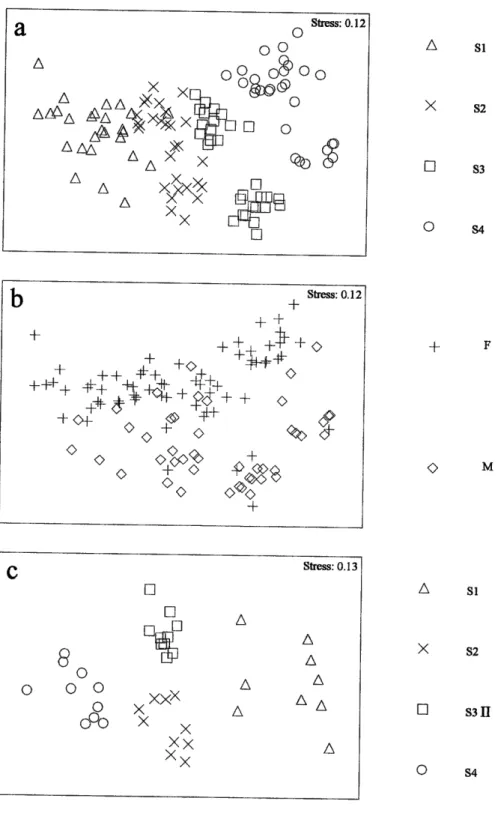
Sampling took place in the Sorfiord
(Fig.
1), which is 37 km long,l-2km
wide and390m maximum depth.
Individuals were
collected
during March
2006,
in
shallow water,by
SCUBA diving
in
5 different
stations at increasing distancesfrom
theford
head: S1, S2, 53 and 54 for Asterias rubens and SI,52,
S3tr
and54
for Echinus acutus(Fig. 1;
Túle
1).
The S4
station was
locatedjust
outside
the
Sorfiord,
in
the Hardangerfiord, towards the open sea.In
order to avoid exposure to the vertical gradientof
salinityan{
therefore,to
an osmotic shock, the animals were placedinto
sealedjars
filled with their
surroundingwater before
beingbrought
to
the
surface. These were aeratedwhile
transportedto the
laboratory locatedin Aga (FiS.
1). Samplesof
thetop
5cm layer
of
sediments were sollectedin
polyethylene containers,in
all
stations. These samples were stored at -20oC and then dried (60"C for 2 days) before metal analysis.For
sprm
motility
analysis, 2AA.
rubensindividuals
were collectedfrom
eachstation and brought
to
Belgium,
in
aeratedwater,
no later than
3
days after their
collection
They were then installedin
a closedcircuit
aquarium system andwithin two
weeks spsrm
motility
video recordings were carriedoú.
For
RNAIDNA
analysis around25
indivídualsof
A.
rubens andE.
acutus werecollected
per
station.
All
individuals were
processedwithin
around
5
hours
after collection. Prior to dissection, ann length of the starfish@),
from armtip
tomouú,
ffid
ambital diameter
of
sea urchins(D)
were measured;all
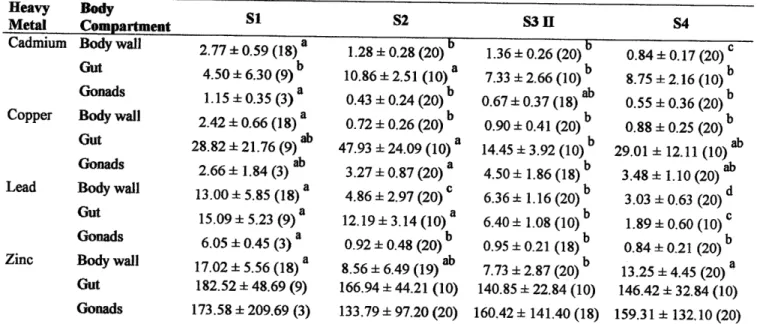
individuals were weighted (W). Samplesof
bodywall
andpyloric
caec4 fromÁ.
rubens, andof
bodywall
and gonads,from
E.
ncttÍtt§,
were
dissected
and
immediately
frozen
in
liquid
nitrogen
and subsequently stored at -80'Cuntil
RNAIDNA
determinations. The remaining bodywall,
pyloric
Gascaand
gonadsfrom
.4.
rubensand body
wall,
gut (after
removal
of
itscontent)
and
gonads,from
E
aeutuswere taken
from the
sameindividuals as
for
RNA/DNÀ
analysis, dried at 60"C for 2 days and storedfor
metal analysis.Males whose gonads were infected
by
ciliates(2
starfishin
S1for RNA/DNA),
hermaptrodite
individuals
(2A.
rubensin
Sl
for RNA/DNA
analysis) and individuals whose gender cannotbe
deterrnined(2
E.
acutusfrom
Sl)
werenot
firther
usedfor
both metal and
RNAIDNA
analysis.§afrord
10 km re §3u Trtanium I iron andsí
Odda ãnc smelterFig.
1.Location
of
the sampling sitesin
the
Sor$ord (SouthwesternNorway): 51,
52, S3, S3II,
S4.Table 1. Location and
conditions
of
the different
sampling
sites
in
the
Ssr$ord,Nonuay.
tn
stations S1, S2 and 54both
species were collected;on 53 only
A.
rubens was sampled whereas on S3II
it
was only E. acutus.Site Coordinates
Distance from the
hcad of the {iord
(km) Collection Depths (m) Water Temperature
cc)
Satinity (