ContentslistsavailableatSciVerseScienceDirect
Phytomedicine
jo u r n al h om e p a g e :w w w . e l s e v i e r . d e / p h y m e d
Cytotoxic
effect
of
leaf
essential
oil
of
Lippia
gracilis
Schauer
(Verbenaceae)
Rosana
P.C.
Ferraz
a, Diogo
S.
Bomfim
a, Nanashara
C.
Carvalho
b, Milena
B.P.
Soares
b,c,
Thanany
B.
da
Silva
d,
Wedna
J.
Machado
e,
Ana
Paula
N.
Prata
e,
Emmanoel
V.
Costa
d,
Valéria
Regina
S.
Moraes
d,
Paulo
Cesar
L.
Nogueira
d,
Daniel
P.
Bezerra
a,∗aDepartmentofPhysiology,FederalUniversityofSergipe,SãoCristóvão,Sergipe,Brazil bGonc¸aloMonizResearchCenter,Fundac¸ãoOswaldoCruz,Salvador,Bahia,Brazil cCenterofBiotechnologyandCellTherapy,HospitalSãoRafael,Salvador,Bahia,Brazil dDepartmentofChemistry,FederalUniversityofSergipe,SãoCristóvão,Sergipe,Brazil eDepartmentofBiology,FederalUniversityofSergipe,SãoCristóvão,Sergipe,Brazil
a
r
t
i
c
l
e
i
n
f
o
Keywords: Lippiagracilis Essentialoil Cytotoxicity Apoptosis Antitumor
a
b
s
t
r
a
c
t
Medicinalplantsareoneofthemostimportantsourcesofdrugsusedinthepharmaceutical indus-try.Amongtraditionalmedicinalplants,LippiagracilisSchauer(Verbenaceae)hadbeenusedforseveral medicinalpurposesinBraziliannortheastern.Inthisstudy,leafessentialoil(EO)ofL.graciliswas pre-paredusinghydrodistillation.FollowedbyGC–MSanalysis,itscompositionwascharacterizedbythe presenceofthymol(55.50%),asmajorconstituent.TheeffectsofEOoncellproliferationandapoptosis inductionwereinvestigatedinHepG2cells.Furthermore,micebearingSarcoma180tumorcellswere usedtoconfirmitsinvivoeffectiveness.EOanditsconstituents(thymol,p-cymene,␥-terpineneand myrcene)displayedcytotoxicitytodifferenttumorcelllines.EOtreatmentcausedG1arrestinHepG2 cellsaccompaniedbytheinductionofDNAfragmentationwithoutaffectingcellmembraneintegrity. Cellmorphologyconsistentwithapoptosisandaremarkableactivationofcaspase-3werealsoobserved, suggestinginductionofcaspase-dependentapoptoticcelldeath.Invivoantitumorstudyshowedtumor growthinhibitionratesof38.5–41.9%.Inconclusion,thetestedessentialoilofL.gracilisleaves,which hasthymolasitsmajorconstituent,possessessignificantinvitroandinvivoantitumoractivity.These datasuggestthatleafessentialoilofL.gracilisisapotentialmedicinalresource.
© 2013 Elsevier GmbH. All rights reserved.
Introduction
Naturalproductsareaninterestingsourceofdrugsusedinthe pharmaceuticalindustry.Amongthese,essentialoilsarecomplex mixturesofodoriferoussubstancesthatusuallypresentmultiple pharmacologyproperties.Eachoftheseconstituentscontributes tothebiologicaleffectsoftheseoils(Bakkalietal.2008).
Lippia gracilis Schauer (Verbenaceae), popularly known as “alecrim-da-chapada”and“candeia-de-queimar”,hadbeenused forseveralmedicinalpurposesinBraziliannortheastern.Among its folk medicinal uses, the treatment of cutaneous diseases, burns,wounds,ulcers,influenza,cough,sinusitis,bronchitis,nasal congestion,headache,jaundiceandparalysishavebeenreported (Pascualetal.2001;Albuquerqueetal.2007).Usually,itsleaves areusedtoprepareinfusionordecoctionand usedasatea,as well as a macerate in alcohol for topical application (Lorenzi and Matos2008).Somestudiesexaminingthepharmacological
∗Corresponding authorat: Department ofPhysiology,FederalUniversity of Sergipe,Av.MarechalRondon,JardimRosaElze,49100-000SãoCristóvão,Sergipe, Brazil.Tel.:+557921056644.
E-mailaddress:[email protected](D.P.Bezerra).
propertiesofL.gracilishave demonstratedthatitsleafessential oilpresentsantibacterial,molluscicidal,larvicidal,antinociceptive andanti-inflammatoryactions(Pessoaetal.2005;Silvaetal.2008; Mendes etal. 2010;Teles et al.2010).Theantinociceptive and anti-inflammatorypropertiesofmethanolicextractofleavesofL. gracilishavealsobeenreported(Guimarãesetal.2012).Recently, in ourcytotoxicdrug-screeningprogram, we demonstrated the cytotoxic activity of leaf essential oil of L. gracilis to several tumorcelllines(Ribeiroetal.2012).However,themechanisms underlyingtheseeffectswerenotexplored.
Inpresentwork,thechemicalcompositionofleafessentialoil (EO)ofL.graciliswascharacterizedbyCG–MS.Themechanisms involvedinEOcytotoxicactivitywereinvestigatedinHepG2cells. InvivoeffectsofEOinmicebearingSarcoma180tumorcellswere alsoevaluated.
Materialsandmethods
Cells
Cytotoxicity was determined in tumor cells using HepG2 (human hepatocellular carcinoma), K562 (human chronic
myelocytic leukemia) and B16-F10 (mouse melanoma), all donatedbyHospitalA.C.Camargo,SãoPaulo,SP,Brazil.Cellswere maintainedinRoswellParkMemorialInstitute-1640(RPMI-1640, Gibco-BRL,Gaithersburg,MD,USA)mediumsupplementedwith 10% fetal bovine serum (Cultilab, Campinas, SP, Brazil), 2mM l-glutamine(VetecQuímicaFina,DuquedeCaxias,RJ,Brazil)and 50g/mlgentamycin(Novafarma,Anápolis,GO,Brazil).Adherent cellswereharvestedbytreatmentwith0.25%trypsinEDTAsolution (Gibco-BRL,Gaithersburg,MD,USA).Allcelllineswereculturedin cellcultureflasksat37◦Cin5%CO
2 andsub-culturedevery3–4 daysto maintainexponential growth.Cytotoxicity experiments wereconductedwithcellsinexponentialgrowthphase.
Sarcoma180tumorcells,whichhadbeenmaintainedby pas-sagesintheperitonealcavityofSwissmice,wereobtainedfrom theLaboratoryofExperimentalOncologyattheFederalUniversity ofCeará.
Human lymphocytecells wereobtainedby primary culture. Heparinizedblood(fromhealthy,non-smokerdonorswhohadnot takenanydrugatleast15dayspriortosampling)wascollected andperipheralbloodmononuclearcells(PBMC)wereisolatedbya standardprotocolusingFicolldensitygradient(Ficoll-PaquePlus, GEHealthcareBio-SciencesAB,Sweden).PBMCwerewashedand resuspendedataconcentrationof0.3×106cells/mlinRPMI1640 mediumsupplementedwith20% fetalbovineserum,2mM glu-tamine,50g/mlgentamycinat37◦Cwith5%CO
2.ConcanavalinA (ConA,SigmaChemicalCo.StLouis,MO,USA)wasusedasa mito-gentotriggercelldivisioninT-lymphocytes.ConA(10g/ml)was addedatthebeginningofcultureandafter24h,cellsweretreated withthetestdrugs(BrownandLawce1997).
Forallexperiments,cellviabilitywasperformedbyTrypanblue assay.Over90%ofthecellswereviableatthebeginningofthe culture.
Animals
Atotalof36Swissmice(males,25–30g),obtainedfromthe cen-tralanimalhouseoftheFederalUniversityofSergipe,Brazil,were used.Animalswerehousedincageswithfreeaccesstofoodand water.Allanimalswerekeptundera12:12hlight-darkcycle(lights onat6:00a.m.).Animalsweretreatedaccordingtotheethical prin-ciplesforanimalexperimentationofSBCAL(Brazilianassociationof laboratoryanimalscience),Brazil.TheAnimalStudiesCommittee fromtheFederalUniversityofSergipeapprovedtheexperimental protocol(number60/2010).
Plantmaterial
L.gracilisleaveswerecollectedintheproximityofthe“Serra daGuia”[coordinates:09◦58′09′ ′S,37◦51′52′ ′W],Poc¸oRedondo, SergipeState,BrazilinNovember2006.Sampleswereprocessed andidentifiedaccordingtostandardprotocol(Morietal.1989), being deposited in the herbarium of the Federal University of Sergipe(ASE)underthe number18740. Thespeciewas identi-fiedbyDr.RaymondMervynHarley,RoyalBotanicGardens,Kew (England).
HydrodistillationandCG–MSanalysisoftheessentialoil
The essential oil from fresh leaves of L. gracilis (50g) was obtainedbyhydrodistillationfor2husingaClevenger-type appa-ratus(Amitel,SãoPaulo,Brazil).Theessentialoilwasdriedover anhydroussodiumsulphate,andthepercentagecontentwas calcu-latedonthebasisofthedryweightofplantmaterial.Theessential oilwasstored at−4◦C in asealed amberbottleuntil chemical analysis.Theextractionswereperformedintriplicate.
OH
Thymol p-Cymene -Terpinene Myrcene
Fig.1.Chemistrystructureofthymol,p-cymene,␥-terpineneandmyrcene.
Essential oil analysis of L. gracilis was performed on a Shimadzu QP5050A GC/MS system equipped with an AOC-20i auto-injector. A J&W Scientific DB-5MS (coated with 5%-phenyl-95%-dimethylpolysiloxane) fused capillary column (30m×0.25mm×0.25mfilmthickness)wasusedasthe sta-tionaryphase.Heliumwasthecarriergasat1.2ml/minflowrate. Column temperatures were programmed from 40◦C for 4min, raisedto220◦Cat4◦C/min,andthenheatedto240◦Cat20◦C/min. The injector and detector temperatures were 250 and 280◦C, respectively.Samples(0.5linCH2Cl2)wereinjectedwitha1:20 splitratio.MSweretakenat70eVwithascanintervalof0.5sand fragmentsfrom40–350Da.
Theretentionindiceswereobtainedbyco-injectingtheoil sam-plewithaC8–C18linearhydrocarbonmixture(vanDenDooland Kratz1963).ThevolatilecomponentswereanalyzedbyGC/MS,and identificationwasmadebycomparingretentionindicesandmass spectrawiththoseintheliterature(Adams2007),aswellasby com-puterizedmatchingoftheacquiredmassspectrawiththosestored intheNISTandWileymassspectrallibraryandotherpublished massspectra.Thepercentagecompositionofeachcomponentwas determinedbydividingtheareaofthecomponentbythetotalarea ofallcomponentsisolatedundertheseconditionswithoutresponse factorcorrection.
Purecompounds
Thymol(purity≥99.5%),p-cymene(purity 99%),␥-terpinene (purity≥97.0%)andmyrcene(purity≥90%)(Fig.1)wereobtained fromSigmaChemicalCo.StLouis,MO,USA.
Cellproliferationassay
20 (2013) 615–621 617
Analysisofmechanismsinvolvedinthecytotoxicactivity
Thefollowing experiments were performedtoelucidate the mechanismsinvolvedincytotoxicactionofEO.Forallexperiments, 2mlofasolutionofHepG2cells(0.7×105cells/ml)wereseeded in24-wellplatesandincubatedbyovernighttoallowthatthecells toadheretotheplatesurface.Then,thecellsweretreatedfor24h withEOatfinalconcentrationof2.5and5.0g/ml.Negative con-trolwastreatedwiththevehicle(0.1%DMSO)usedfordiluting thetesteddrug.Doxorubicin(1.0g/ml)wasusedasthepositive control.
Trypanbluedyeexclusiontest
CellproliferationwasassessedbytheTrypanbluedyeexclusion test.HepG2cellswereseededintheabsenceandpresenceofEO. After24hdrugexposure,cellproliferationwasassessed.Cellsthat excludedTrypanbluewerecountedusingaNeubauerchamber.
Cellcycledistribution
Cellswereharvestedinalysissolutioncontaining0.1%Triton X-100(SigmaChemicalCo.StLouis,MO,USA)and2g/ml pro-pidiumiodide(BioSource,USA).Cellfluorescencewasdetermined byflowcytometryinaFACSCaliburcytometer(BenctonDickinson, SanDiego,CA,USA)withCellQuestsoftware(BDBiosciences,San Jose,CA,EUA).Tenthousandeventswereevaluatedperexperiment andcellulardebriswasomittedfromtheanalysis.
Morphologicalanalysiswithhematoxylin–eosinstaining
Morphological changes were examined by light microscopy (OlympusBX41,Tokyo,Japan)usingImage-ProExpresssoftware (MediaCybernetics,Inc.SilverSpring,USA).Toevaluatealterations inmorphology,cellsfromcultureswereharvested,transferredto cytospinslides, fixedwithmethanol for 30s, and stained with hematoxylin–eosin.
Morphologicalanalysisusingfluorescencemicroscope
Morphological changes were examined using fluorescence microscope.Cellswerepelletedandresuspendedin25lsaline. Thereafter,1lofaqueoussolutionofacridineorange(AO,Sigma ChemicalCo.StLouis,MO,USA)andethidiumbromide(EB,Sigma ChemicalCo.StLouis,MO,USA)(AO/EB,100g/ml)wasaddedand thecellswereobservedunderafluorescencemicroscope(Olympus BX41,Tokyo,Japan).Threehundredcellswerecountedpersample andclassifiedasviable,apoptoticornecroticcells.
Cellmembraneintegrity
Thecellmembraneintegrity wasevaluatedbytheexclusion of propidiumiodide.Cell fluorescence wasdeterminedby flow cytometry,asdescribedabove.
Caspase-3activationassay
Caspase-3/CPP32 colorimetric assay kit (BioVision Incorpo-rated,CA,USA) wasused toinvestigatecaspase-3 activationin treatedcells basedonthecleavageof Asp-Glu-Val-Asp (DEVD)-pNA.Briefly,cellswerelysedbyincubationwithcelllysisbufferon icefor10minandthencentrifugedat10,000gfor1min.Toeach reactionmixture,50lcelllysate(100–200gtotalprotein)was added.Enzymereactionswerecarriedoutina96-wellflat-bottom microplate.
Invivoantitumorassay
The in vivo antitumor effect was evaluated using sarcoma 180ascitestumorcellsfollowingprotocolspreviouslydescribed (Bezerraetal.2008;Brittoetal.2012).Ten-dayoldsarcoma180 ascitestumorcells(2×106cellsper500l)wereimplanted sub-cutaneouslyintothelefthindgroinofmice.EOwasdissolvedin5% DMSOandgiventomiceintraperitoneallyonceadayfor7 consecu-tivedays.Negativecontrolwastreatedwiththevehicle(5%DMSO) usedfordilutingthetestedsubstance.5-Fluoruoracil(5-FU,Sigma ChemicalCo.StLouis,MO,USA)wasusedasthepositivecontrol. Atthebeginningoftheexperiment,themiceweredividedinto fourgroups,asfollows:Group1:animalstreatedbyi.p.injection ofvehicle5%DMSO(n=12);Group2:animalstreatedbyi.p. injec-tionof5-FU(25mg/kg/day)(n=8);Group3:animalstreatedby i.p.injectionofEO(40mg/kg/day)(n=8);Group4:animalstreated byi.p.injectionofEO(80mg/kg/day)(n=8).Thetreatmentswere startedonedayaftertumorinjection.Thedosagesweredetermined basedonpreviousarticles.Onday8,theanimalswereeuthanized, bycervicaldislocation,andthetumorswereexcisedandweighed. Thedrugeffectswereexpressedasthepercentinhibitionofcontrol. Bodyweightloss,organweightalterationandhematological analysesweredeterminedattheendofexperimentabove,as pre-viouslydescribed(Bezerraetal.2008;Brittoetal.2012).Peripheral bloodsampleswerecollectedfromtheretro-orbitalplexusunder lightetheranesthesiaandtheanimalswereeuthanizedbycervical dislocation.Aftersacrifice,liver,kidneyandspleenswereremoved andweighed.Inhematologicalanalysis,totalanddifferential leuko-cytecountsweredeterminedbystandardmanualproceduresusing lightmicroscopy.
Statisticalanalysis
Dataarepresentedasmean±SEM(orSD)orIC50valuesand their 95% confidence intervals (CI 95%) obtained by nonlinear regression. Differencesamong experimentalgroups were com-pared by one-way analysis of variance (ANOVA) followed by Newman–Keulstest(p<0.05).Allanalyseswerecarriedoutusing theGRAPHPADprogram(IntuitiveSoftwareforScience,SanDiego, CA,USA).
Resultsanddiscussion
Thepresentworkinvestigatedthephytochemicalandcytotoxic propertiesofleafessentialoilofL.gracilis.Itwaschemically char-acterizedbyCG–MSanalysis.TheeffectsofEOoncellproliferation andapoptosisinductionwereinvestigatedinHepG2cells. Further-more,micebearingSarcoma180tumorcellswereusedtoconfirm itsinvivoeffectiveness.
Table1
ChemicalconstituentsofleafessentialoilofL.gracilis.
RIa RIb Compounds %Peakareac
1 884 880 3-Methyl-3-buten-1-olacetate tr
2 924 924 ␣-Thujene 1.23±0.12
3 930 932 ␣-Pinene 0.40±0.00
4 973 974 -Pinene tr
5 988 988 Myrcene 4.03±0.29
6 1003 1002 ␣-Phellandrene 0.10±0.00
7 1005 1008 ␦-3-Carene 0.27±0.06
8 1014 1014 ␣-Terpinene 1.47±0.15
9 1022 1020 p-Cymene 10.80±1.35
10 1026 1024 Limonene 0.57±0.06
11 1035 1032 (Z)--Ocimene 0.20±0.00
12 1045 1044 (E)--Ocimene 0.20±0.00
13 1056 1054 ␥-Terpinene 5.53±0.57
14 1067 1065 cis-Sabinenehydrate tr
15 1082 1086 Terpinolene 0.10±0.00
16 1097 1095 Linalool 0.13±0.06
17 1169 1167 Umbellulone 0.30±0.18
18 1176 1174 Terpinen-4-ol 0.90±0.00
19 1226 1232 Thymolmethylether 10.53±1.40
20 1290 1289 Thymol 55.50±4.21
21 1294 1298 Carvacrol 0.20±0.00
22 1343 1349 Thymolacetate 3.30±0.44
23 1362 1369 Cyclosativene 0.20±0.00
24 1369 1374 ␣-Copaene 0.10±0.00
25 1377 1376 (E)-Methylcinnamate 0.30±0.10
26 1416 1417 (E)-Caryophyllene 1.43±0.06
27 1435 1439 Aromadendrene 0.20±0.00
28 1452 1452 ␣-Humulene 0.20±0.00
29 1469 1469d 2,6-Dimethoxyacetophenone 0.23±0.06
30 1487 1496 Viridiflorene 0.20±0.00
31 1491 1500 Bicyclogermacrene 0.17±0.06
32 1504 1505 -Bisabolene 0.27±0.06
33 1511 1511 ␦-Amorphene tr
34 1578 1582 Caryophylleneoxide 0.20±0.00
35 1581 1590 Globulol 0.10±0.01
TOTAL 99.36
tr,trace(meanvalue<0.10%).
aRI,retentionindicesonDB-5MScolumncalculatedaccordingtovanDenDool andKratz(1963).
b RI,retentionindicesaccordingtoAdams(2007). c Dataarepresentedasmean±SDofthreeanalyses. d RI,retentionindexaccordingtoTret’yakov(2008).
thelowestcontentofcarvacroland(E)-caryophyllenesuggeststhat thismaybeanotherchemotypethatitisanovelsourceofthymol. Threetumorcelllinesweretreatedwithincreasing concentra-tionsofEOanditsconstituents(thymol,p-cymene,␥-terpinene and myrcene)for 72h and analyzedby methyl-[3H]-thymidine incorporationassay.Table2showstheobtainedIC50 values.EO showedIC50valuesrangedfrom4.93to22.92g/ml forHepG2 andK562celllines,respectively.Amongitsconstituents,myrcene presentedtobethemostcytotoxiccompound,showingIC50values rangingfrom9.23to12.27g/mlforHepG2andB16-F10celllines, respectively.Thymol,p-cymeneand␥-terpineneshowed cytotox-icityonlyfor B16-F10,showingIC50 values of 18.23,20.06and 9.28g/ml,respectively. Doxorubicin,usedasthe positive con-trol,showedIC50valuesfrom0.03to2.92g/mlforB16-F10and K562celllines,respectively.Inaddition,thecytotoxicityofEOwas alsoevaluatedtonormal cells(PBMC).Theresults, presentedin Table2,showthatEOwasalsocytotoxictonormalcells.Noneof EOconstituentsshowedcytotoxicitytonormalcellsatthetested concentrations(IC50>25g/ml).
Accordingtoourcytotoxicdrug-screeningprogram,essential oilthat showsIC50 valuesbelow 30g/mland purecompound thatshowsIC50 valuesbelow1g/mlareconsideredpromising (Suffness and Pezzuto1990;Bezerra et al.2008).Therefore, EO is considered a potent cytotoxic agent. On the other hand, its constituentsthymol,p-cymene,␥-terpineneandmyrceneare
con-sideredweakcytotoxicagents.Thesecompoundswerepreviously Table
20 (2013) 615–621 619
Table3
EffectofleafessentialoilofL.gracilisoncellcycledistributionofhumanhepatocellularcarcinomaHepG2cellsafter24hincubation.
Drugs Concentration
(g/ml)
Cellcyclephases(%)
G1 S G2/M
Control – 62.49±2.12 10.76±1.57 17.63±1.86
Doxorubicin 1.0 39.31± 5.06* 9.64±1.73 54.07±7.90*
Essentialoil 2.5 72.08±1.10* 9.23±1.32 13.26±0.88
5.0 74.61±1.31* 8.87±0.85 12.48±0.78
Dataarepresentedasmeanvalues±S.E.M.fromtwoindependentexperimentsperformedinduplicate.Negativecontrolwastreatedwiththevehicle(0.1%DMSO)usedfor dilutingthetestedsubstance.Doxorubicinwasusedasthepositivecontrol.Tenthousandeventswereanalyzedineachexperiment.
*p<0.05comparedtocontrolbyANOVAfollowedbyStudent-Newman–Keulstest.
assessedagainsttumorcelllines.Among them,thymolshowed IC50 valueof∼60g/mltoHL-60cells and␥-terpineneshowed IC50valueof156.92g/mltoJurkatcells (Debetal.2011; Döll-Boscardinetal.2012).Probably,thepotentcytotoxicactivityof testedessentialoilmightbeattributedtomixtureofitsmainand minorconstituents.
SinceHepG2cellswereespeciallysensitivetoEOcytotoxicity, furtherstudieswereperformedwiththiscelllineusing concen-trationscorrespondingto2.5and5.0g/ml.Theseconcentrations werechosenbasedonitsIC50valueinthiscellline(4.93g/ml).
WhenanalyzedbyTrypanbluedyeexclusion,EOreduced prolif-erationofHepG2cellsinaconcentration-dependentmannerafter 24hincubation(p<0.05,Fig.2).
Cellcyclearrestisacommoncauseofcellgrowthinhibition.To determinewhetherEOcytotoxicityinductioninvolvesalterations incellcycleprogression,analysisofcellcycledistributionbyflow cytometrywereincludedinthisstudy.AllDNAsubdiploidinsize (sub-G1)wereconsideredasinternucleosomalDNAfragmentation. TheresultsoftheeffectofEOoncellcycledistributionshowed thattotalnumberofG1cellsincreased,indicatingcellcyclearrest duringthisphase(Table3).G1-phasecellcyclearrestcreatesan opportunityforcellstoeitherundergorepairorentertheapoptotic
pathwaytomaintaintissuehomeostasisandeliminatethemutated neoplasticandhyperproliferatingneoplasticcellsfromthesystem (Puccietal.2000).BesidestheincreasingofcellsinG1,itwasalso
Control Dox 2.5 5.0
0 5 10 15 20 25 30 35 40
* *
*
Essential oil (µg/ml)
Cell number (x10 4 cell/ml)
Fig.2. EffectofleafessentialoilofL.gracilisontheproliferationofhuman hepa-tocellularcarcinomaHepG2cellsmeasuredbyTrypanbluedyeexclusionmethod after24hincubation.Negativecontrolwastreatedwiththevehicle(0.1%DMSO) usedfordilutingthetestedsubstance.Doxorubicin(Dox,1.0g/ml)wasusedas thepositivecontrol.Dataarepresentedasmeanvalues±S.E.M.fromtwoorthree independentexperimentsperformedinduplicate.*p<0.05comparedtonegative controlbyANOVAfollowedbyStudent-Newman–Keulstest.
Control Dox 2.5 5.0 0
20 40 60 80 100
Essential oil
* *
*
*
(µg/ml) A
Cell numbers (%)
Control Dox 2.5 5.0
0 20 40 60 80 100
Essential oil B
(µg/ml))
Cell membrane integrity (%)
Control Dox 2.5 5.0
0 5 10 15 20
*
Essential oil (µg/ml)) C
*
*
Internucleosomal DNA fragmentation (%)
Control Dox 2.5 5.0
0.0 0.2 0.4 0.6
Essential oil
* *
*
(µg/ml)) D
Caspase 3 activation
(OD 405nm)
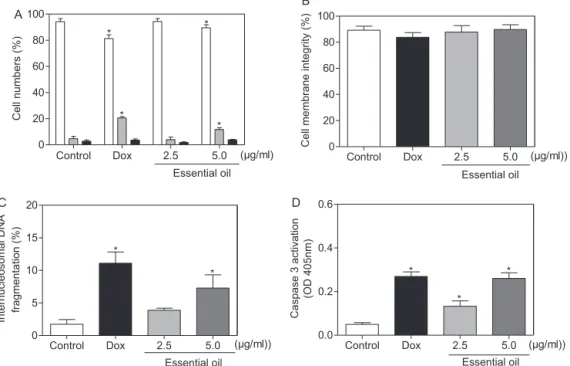
Fig.4. EffectofleafessentialoilofL.gracilisonviabilityofhumanhepatocellularcarcinomaHepG2cellsafter24hincubation.(A)Cellviabilitymeasuredbyfluorescence microscopeusingacridineorange/ethidiumbromide–viablecells(whitebar),apoptoticcell(graybar),necroticcell(blackbar).(B)Cellmembraneintegritymeasuredby flowcytometryusingpropidiumiodide.(C)InternucleosomalDNAfragmentationdeterminedbyflowcytometricusingpropidiumiodideandtritonX-100.(D)Caspase3 activationmeasuredbycolorimetricassay.Negativecontrolwastreatedwiththevehicle(0.1%DMSO)usedfordilutingthetestedsubstance.Doxorubicin(Dox,1.0g/ml) wasusedasthepositivecontrol.Dataarepresentedasmeanvalues±S.E.M.fromtwoorthreeindependentexperimentsperformedinduplicate.Forflowcytometryanalysis tenthousandeventswereanalyzedineachexperiment.*p<0.05comparedtonegativecontrolbyANOVAfollowedbyStudent-Newman–Keulstest.
observedanincreasingintheinternucleosomalDNAfragmentation (p<0.05,Fig.4C).
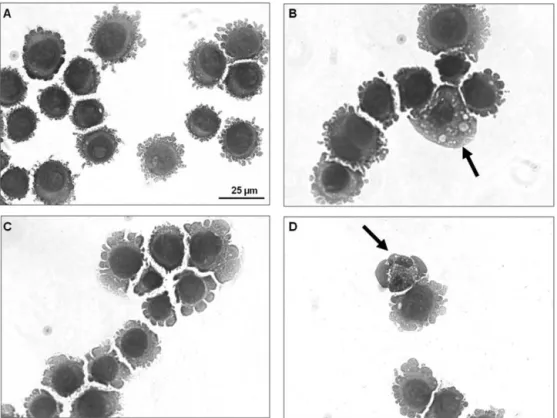
Morphological changes were investigated using hematoxylin–eosinstaining(Fig.3).Inpresenceof5.0g/mlofEO, cellspresentedmorphologyconsistentwithapoptosis,including cellvolumereduction,chromatincondensationandfragmentation of the nuclei condensation. Morphological changes were also investigatedusingAO/EB stainingand fluorescencemicroscopy, wherethepercentagesofviable,apoptoticandnecroticcellswere calculated.After24hofexposure, EO-treatedcellspresentedan increasednumber ofapoptoticcellsatconcentrationof5g/ml (p<0.05,Fig.4A).
EO did not disrupt membrane at any tested concentration (p>0.05, Fig.4B). In addition, as cited above, DNA fragmenta-tionincreasedin EO-treated cells (p<0.05, Fig.4C). These both modificationswerecompatiblewithapoptotic cells.Inaddition, a remarkable activation of caspase-3 was recorded in lysates fromHepG2cellstreatedwithEO(Fig.4D),suggesting caspase-dependentapoptoticcelldeath.
Apoptosis is a regulated cell death process that elimi-nates damaged or malfunctioning cells. It is characterized by phosphatidylserine exposure, loss of mitochondrial membrane potential, caspase activation, chromatin condensation, nuclear fragmentation, resulting in the phagocytosis of membrane-boundapoptotic bodies (Walsh and Edinger 2010).Herein, we demonstratedthatEOisabletoinducecelldeaththrough caspase-dependentapoptosispathwayinHepG2cells. Interestingly,Deb etal. (2011)reportedthat thymol, themain constituentof EO, is ableto induceHL-60 cell deathby apoptosispathway asso-ciatedwith the reactive oxygen species production, disruption ofmitochondrialmembranepotential,increaseinmitochondrial H2O2 production,adecreaseinBcl-2protein,anincreaseinBax protein levels and caspase-9, -8 and -3 activation. Moreover, thymol was also able to induce caspase-independent apopto-sis.
ToinvestigatewhetherOEhasinvivoantitumoractivity,mice were subcutaneously transplanted withsarcoma 180 cells and treatedbyintraperitonealrouteonceadayfor7consecutivedays withEO.TheeffectsofEOonmicetransplantedwithsarcoma180 tumorcellsarepresentedinFig.5.Tumorgrowthinhibitionrates were38.5–41.9%.Theinhibitionwassignificantatbothdosesin relationtothecontrolgroup(p<0.05).
Systemic toxicological parameters were also examined in EO-treatedmice.Forthese,bodyweightloss,organweight alter-ationandleukogramweredetermined.Nostatisticallysignificant changesinEO-treatedmicewereseeninanytoxicological param-etersanalyzed(p>0.05,datanotshown).Incontrast,5-FU,usedas thepositivecontrol,reducedthebodyweightsandspleenorgan weightsandinducedadecreaseintotalleukocytes(p<0.05,data notshown).
Control 5-FU 40 80
0.0 0.5 1.0 1.5 2.0 2.5
0 20 40 60 80 100
Tumor weight Inhibition
(mg/kg/day) Essential oil
* *
*
Tumor weight (g)
Inhibition (%)
20 (2013) 615–621 621
Inconclusion,thesedatapresentedthatthetestedleaf essen-tialoilofL.gracilisischemicallycharacterizedbythepresenceof thymol,asmajorconstituent,andpossessesinvitroandinvivo anti-canceractivities.Incell-basedassay,itwasabletoinduceG1arrest andcaspase-dependentapoptosisinHepG2cells.Inanimalmodel, itwasfoundtobeassociatedwithadecreaseintumorgrowth.
Conflictofinterest
Theauthorshavedeclaredthatthereisnoconflictofinterest.
Acknowledgements
ThisworkwasfinanciallysupportedbyCAPES(Coordenac¸ão de Aperfeic¸oamento dePessoal de NívelSuperior), CNPq (Con-selho Nacional de Desenvolvimento Cientifico e Tecnológico), FAPESB(Fundac¸ãodeAmparoàPesquisadoEstadodaBahia)and FAPITEC/SE(Fundac¸ãodeAmparoàPesquisaeàInovac¸ão Tecnológ-icadoEstadodeSergipe).TheauthorsthankElisalvaT.Guimarães andDanieleBrustolimforassistanceinflowcytometricdata acqui-sition.
References
Adams,R.P.,2007.IdentificationofEssentialOilComponentsbyGas Chromatogra-phy/MassSpectrometry,fourthed.AlluredPublishingCorporation,IL. Albuquerque,U.P.,Medeiros,P.M.,Almeida,A.L.S.,Monteiro,J.M.,LinsNeto,E.M.F.,
Melo,J.G.,Santos,J.P.,2007.Medicinalplantsofthecaatinga(semi-arid) vege-tationofNEBrazil:aquantitativeapproach.JournalofEthnopharmacology114, 325–354.
Bakkali,F.,Averbeck,S.,Averbeck,D.,Idaomar,M.,2008.Biologicaleffectsof essen-tialoils:areview.FoodandChemicalToxicology46,446–475.
Bezerra,D.P.,Pessoa,C.,Moraes,M.O.,Alencar,N.M.,Mesquita,R.O.,Lima,M.W., Alves,A.P.,Pessoa,O.D.,Chaves,J.H.,Silveira,E.R.,Costa-Lotufo,L.V.,2008.In vivogrowthinhibitionofsarcoma180bypiperlonguminine,analkaloidamide fromthePiperspecies.JournalofAppliedToxicology28,599–607.
Britto,A.C.S.,Oliveira,A.C.A.,Henriques,R.M.,Cardoso,G.M.B.,Bomfim,D.S., Car-valho,A.A.,Moraes,M.O.,Pessoa,C.,Pinheiro,M.L.B.,Costa,E.V.,Bezerra,D.P., 2012.Invitroandinvivoantitumoreffectsoftheessentialoilfromtheleavesof Guatteriafriesiana.PlantaMedica78,409–414.
Brown,M.G.,Lawce,H.J.,1997.Peripheralbloodcytogeneticmethods.In:Barch, M.J.,Knutsen,T.,Spurbeck,J.L.(Eds.),TheAGTCytogeneticsLaboratoryManual. Lippincott-RavenPublishers,Philadelphia,pp.77–171.
Deb,D.D.,Parimala,G.,Saravana,Devi,S.,Chakraborty,T.,2011.Effectofthymolon peripheralbloodmononuclearcellPBMCandacutepromyeloticcancercellline HL-60.Chemico-BiologicalInteractions193,97–106.
Döll-Boscardin,P.M.,Sartoratto,A.,Maia,B.H.,Paula,J.P.,Nakashima,T.,Farago, P.V., Kanunfre,C.C., 2012. In vitrocytotoxic potential ofessential oils of
Eucalyptusbenthamiianditsrelatedterpenesontumorcelllines.Evidence-Based ComplementaryandAlternativeMedicine,2012,342652.
Griffiths,M.,Sundaram,H.,2011.Drugdesignandtesting:profilingof antiprolif-erativeagentsforcancertherapyusingacell-basedmethyl-[3H]-thymidine incorporationassay.MethodsinMolecularBiology731,451–465.
Guimarães,A.G.,Gomes,S.V.,Moraes,V.R.,Nogueira,P.C.,Ferreira,A.G.,Blank,A.F., Santos,A.D.,Viana,M.D.,Silva,G.H.,Quintans,Júnior,L.J.,2012.Phytochemical characterizationandantinociceptiveeffectofLippiagracilisSchauer.Journalof NaturalMedicines66,428–434.
Lorenzi,H.,Matos,F.J.A.,2008.PlantasMedicinaisnoBrasil:NativaseExóticas, seconded.InstitutoPlantarum,NovaOdessa.
Mendes,S.S.,Bomfim,R.R.,Jesus,H.C.,Alves,P.B.,Blank,A.F.,Estevam,C.S.,Antoniolli, A.R.,Thomazzi,S.M.,2010.Evaluationoftheanalgesicandanti-inflammatory effectsoftheessentialoilofLippiagracilisleaves.JournalofEthnopharmacology 129,391–397.
Mori,S.A.,Silva,L.A.M.,Lisboa,G.,Coradin,L.,1989.Manualdemanejodoherbário fanerogâmico,seconded.CEPLAC,Ilhéus.
Neves,I.A.,deOliveira,J.C.S.,daCamara,C.A.G.,Schwartz,M.O.E.,2008. Chemi-calcompositionoftheleafoilsofLippiagracilisSchauerfromtwolocalitiesof Pernambuco.JournalofEssentialOilResearch20,157–160.
Pascual,M.E.,Slowing,K.,Carretero,E.,SánchezMata,D.,Villar,A.,2001.Lippia: traditionaluses,chemistryandpharmacology:areview.Journalof Ethnophar-macology76,201–214.
Pessoa,O.D.,deCarvalho,C.B.,Silvestre,J.O.,Lima,M.C.,Neto,R.M.,Matos,F.J., Lemos,T.L.,2005.AntibacterialactivityoftheessentialoilfromLippiaaff.gracilis. Fitoterapia76,712–714.
Pucci,B.,Kasten,M.,Giordano,A.,2000.Cellcycleandapoptosis.Neoplasia2, 291–299.
Ribeiro,S.S.,Jesus,A.M.,Anjos,C.S.,Silva,T.B.,Santos,A.D.C.,Jesus,J.R.,Andrade,M.S., Sampaio,T.S.,Gomes,W.F.,Alves,P.B.,Carvalho,A.A.,Pessoa,C.,Moraes,M.O., Pinheiro,M.L.B.,Prata,A.P.N.,Blank,A.F.,Silva-Mann,R.,Moraes,V.R.S.,Costa, E.V.,Nogueira,P.C.L.,Bezerra,D.P.,2012.Evaluationofthecytotoxicactivityof someBrazilianmedicinalplants.PlantaMedica78,1601–1606.
Silva,W.J.,Dória,G.A.,Maia,R.T.,Nunes,R.S.,Carvalho,G.A.,Blank,A.F.,Alves,P.B., Marc¸al,R.M.,Cavalcanti,S.C.,2008.EffectsofessentialoilsonAedesaegypti lar-vae:alternativestoenvironmentallysafeinsecticides.BioresourceTechnology 99,3251–3255.
Suffness, M.,Pezzuto,J.M., 1990. Assaysrelated tocancer drugdiscovery. In: Hostettmann,K.(Ed.),MethodsinPlantBiochemistry:AssaysforBioactivity. AcademicPress,London,pp.71–133.
Teles,T.V.,Bonfim,R.R.,Alves,P.B.,Blank,A.F.,Jesus,H.C.R.,QuintansJr.,L.J.,Serafini, M.R.,Bonjardim,L.R.,Araújo,A.A.S.,2010.Compositionandevaluationofthe lethalityofLippiagracilisessentialoiltoadultsofBiomphalariaglabrataand larvaeofArtemiasalina.AfricanJournalofBiotechnology9,8800–8804. Tret’yakov, K.V., 2008.Retention data.In:Linstrom, P.J., Mallard,W.G.(Eds.),
NIST Chemistry Webbok. National Institute of Standards and Technol-ogy, Gaithersburg, MD, NIST Standard Reference Database Number 69, http://webbook.nist.gov/chemistry/(accessedSeptember,2012).
vanDenDool,H.,Kratz,P.D.,1963.Ageneralizationoftheretentionindexsystem includinglineartemperatureprogrammedgas–liquidpartition chromatogra-phy.JournalofChromatography11,463–471.