J. Braz. Chem. Soc. vol.19 número6
Texto
Imagem
Documentos relacionados
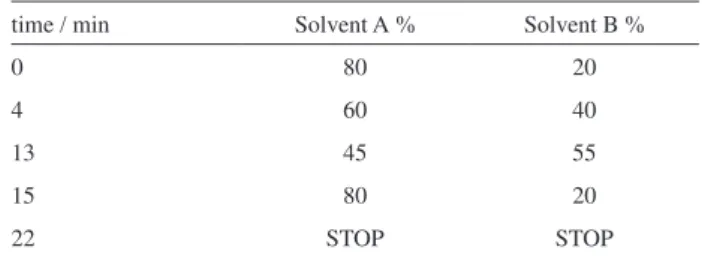
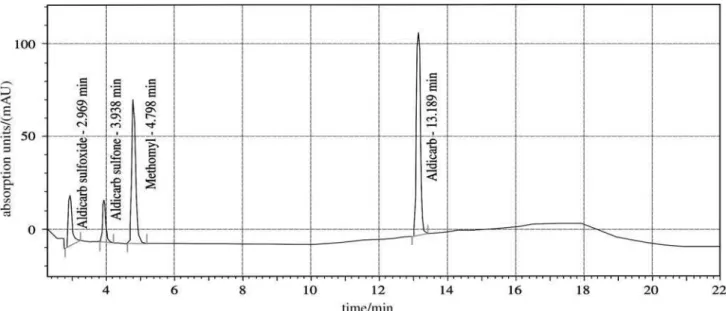
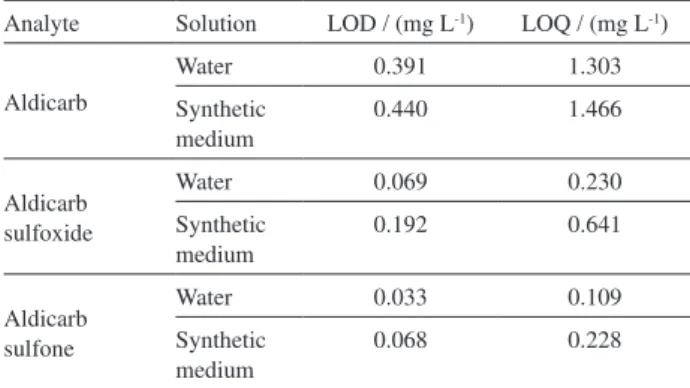
Thus, the aim of this study was to validate a simple and reliably HPLC-UV method using solid-liquid extraction followed by an isocratic chromatographic elution for
Development and validation of RP-HPLC method for simultaneous estimation of cefepime hydrochloride and amikacin sulphate in injection dosage form. J Pharm Sci
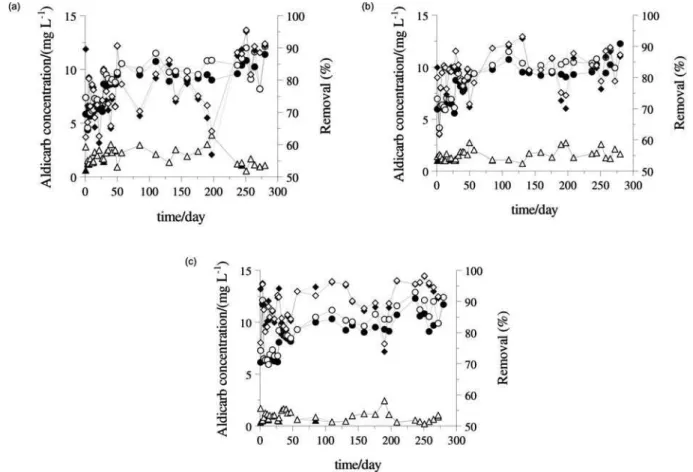
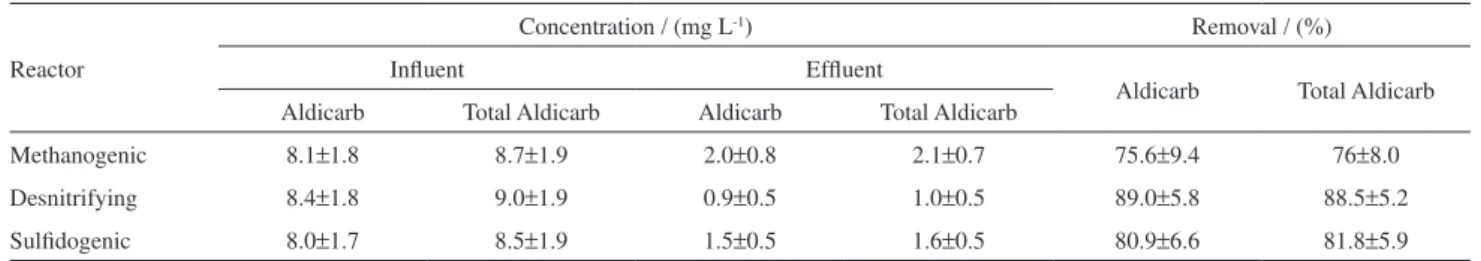
amazonensis infective juveniles and different root exudates (cucumber, tomato, garlic, bean, soybean and corn) and control treatments (water and aldicarb), in Petri
The present study describes the development and subsequent validation of simple and accurate stability indicating RP-HPLC method for the determination of sparloxacin and
The present work describes the development of a new high performance liquid chromatographic (HPLC) method for the determination of Ceixime trihydrate under diferent stress conditons
The investigation includes the determination of organic impurities, inorganic impurities and volatiles, validation of the HPLC-DAD method, stability studies under transport
Validation parameters of the developed HPLC method for simultaneous determination of sesquiterpene lactones in the dichloromethane fraction of the hydroalcoholic extract obtained
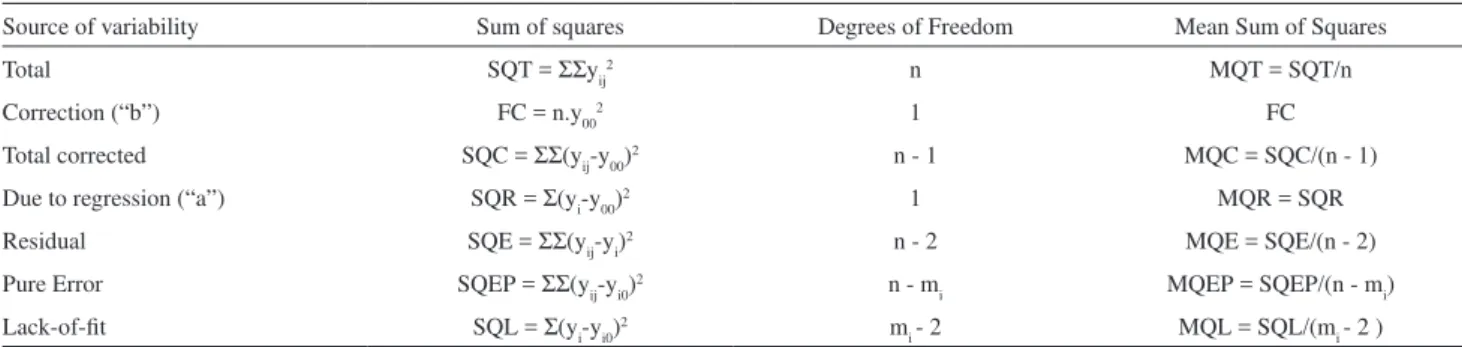
Taking advantage of the representation of a time series data by its trajectory matrix of Hankel constructed using Singular Spectrum Analysis, as well as of its decomposition