w w w . r b h h . o r g
Revista
Brasileira
de
Hematologia
e
Hemoterapia
Brazilian
Journal
of
Hematology
and
Hemotherapy
Original
article
Human
leukocyte
antigen
allele
linkage
disequilibrium
and
haplotype
structure
in
volunteer
bone
marrow
donors
of
Paraná
State
Paulo
Rincoski
Costantino
∗,
Suelen
Camargo
Zeck,
Waldir
Antonio
da
Silva,
Maria
da
Grac¸a
Bicalho
UniversidadeFederaldoParaná,Curitiba,PR,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received9November2016 Accepted17January2017 Availableonline21February2017
Keywords:
Geneticpolymorphism Linkagedisequilibrium Transplantation Histocompatibility HLAantigens
a
b
s
t
r
a
c
t
Background:Bonemarrowtransplantationhasbeenusedinthetreatmentofvarious dis-eases,especiallyhematologicdiseases.Thesuccessofthistreatment,amongotherfactors, requireshumanleukocyteantigens(HLA)compatibilitybetweenpatientanddonor. Know-ingthehumanleukocyteantigensallelegroupandhaplotypefrequenciesaswellasthe linkage disequilibrium betweenalleles ofdifferent humanleukocyte antigens locican shortenthesearchtimeforacompatiblebonemarrowdonor.
Objective:Toassembleandanalyzedataonhumanleukocyteantigensfrequencies avail-ableintheLaboratoryofImmunogeneticsandHistocompatibility(LIGH)databaseofthe UniversidadeFederaldoParanáaddinganestimationoftheHardy–Weinbergequilibrium andlinkagedisequilibrium.
Methods:Thesamplewascomposedofsevenpopulationsgroupedbyself-declaredancestry or inferredfromthesurnameasfollows:LaboratoryofImmunogeneticsand Histocom-patibilitydatabase(allgroups),descendantsofItalians,Poles,andAsians,Afro-Brazilians, Mulattos(mixedancestry)andAmerindians.Humanleukocyteantigensgenotypingwas carriedoutusingthepolymerasechainreaction-sequencespecificprimers(PCR-SSP)and -sequencespecificoligonucleotide(PCR-SSO)technologies.
Results:Therewerehighfrequencies oftheHLA-A*02,HLA-B*35andHLA-DRB1*13allelic groups inall groups.Thesame wasobservedforthe HLA-A*01-B*08-DRB1*03haplotype exceptforAsiandescendants.Itwasobservedthatthehumanleukocyteantigens Labo-ratoryofImmunogeneticsandHistocompatibilitydatabaseandtheAsiangrouparenotin Hardy–Weinbergequilibrium.TheItalian,Polish,Asian,MulattoandAmerindian descen-dantsshowedhaplotypesincompletelinkagedisequilibrium.Ourresultswerecompared withdataonthehumanleukocyteantigensintheParanápopulationavailablefromthe BrazilianVoluntaryBoneMarrowDonorRegistry(REDOME)anddatapublishedonthe pop-ulationofCuritibaandthenorthernregionofParaná.
∗ Correspondingauthorat:LaboratóriodeImunogenéticaeHistocompatibilidadedaUFPR(LIGH-UFPR)RuaFranciscoH.dosSantoss/n– JardimdasAméricas,CentroPolitécnico,SetordeCiênciasBiológicas,DepartamentodeGenética,Sala31,81530-990Curitiba,PR,Brazil.
E-mailaddress:[email protected](P.R.Costantino). http://dx.doi.org/10.1016/j.bjhh.2017.01.006
Conclusions: HaplotypesfrequentintheAsiangroupwerenotthemostfrequentlyobserved intheLaboratoryofImmunogeneticsandHistocompatibilitydatabaseandtheNational BoneMarrowDonorRegistryforthestateofParaná.Linkagedisequilibriuminformation mayproveusefulinthesearchforbonemarrowdonorsforpatientsawaitingasuitable donor.
©2017Associac¸ ˜aoBrasileiradeHematologia,HemoterapiaeTerapiaCelular.Published byElsevierEditoraLtda.ThisisanopenaccessarticleundertheCCBY-NC-NDlicense (http://creativecommons.org/licenses/by-nc-nd/4.0/).
Introduction
Bonemarrowtransplantationisatreatmentforvarious blood-relateddiseases,suchasleukemias,lymphomas,andsevere anemia.Amongotherfactors,compatibilityofhuman leuko-cyteantigens(HLA)betweenpatientanddonorismandatory forasuccessfulbonemarrow transplantation.Thislessens the chance of graft rejection and the occurrence of graft versus host disease (GVHD). Although immunosuppressive drugsreducethe possibilityofrejection,HLA compatibility isthefactorthatprovidesthebestprognosisfora success-fulprocedure.1 CurrentlyHLA-A,-Band-DRB1are matched
asoneofthefirstphasecriteriainthesearchofcompatible donorsforrecipientsonthewaitinglist.
HLA diversity reflects extraordinary polymorphism and polyallelismatmultipleclassIandclassIIlocithathave co-dominantexpressions.Recombinationeventsbetweenalleles inthemajorhistocompatibilitycomplex(MHC)regionsoccur witha frequencyof2–4%.1 For this reason,HLAgenes are
inheritedinatightclusterofcloselylinkedgenesor haplo-types.Theycodeforthemostimmunogenicproteinsknown todateandknowledgeabouttheHLAprofileofpatientand donorisanimportantissueinthecontextofcellandtissue transplantation.1
AccordingtoVogel&Motulsky,thegenesintheHLA sys-tempresentstrikingdeviationsfromequilibrium.2Lewontin
&Kojimacreatedthetermlinkagedisequilibrium(LD)that, similartootherterms,doesnotdescribethetruemeaning, thatis,thenon-randomassociationofallelesattwoormore loci.ThewordsLDcanalsorefertogeneslocatedondifferent chromosomesthatsegregateindependentlyinapopulation witharecentpastofpanmicticbreedingorgeneslocatedat adjacentgeneticloci.3LDischaracterizedwhenthefrequency
ofassociationbetweenallelesishigherorlowerthan what wouldbeexpectedconsideringtheindividualallele frequen-cies.ThepatternofLDvariesmarkedlybetweenpopulations anddependsontheevolutionaryhistory.Themore genera-tions ofpanmixia,the closerthe population willbetothe equilibrium.
TheBrazilianpopulation,whoserootsarebasedonan eth-nicmatrixofEuropean,AfricanandAmerindianorigins,has beenpluralizedandincreasedwiththediversityandgenetic compositionofvariousmigratoryflowsofpopulationswith differentbackgrounds.EachBrazilianregionhasitsown eth-nicparticularity.InParanáState,locatedinsouthernBrazil, thereisaprevalenceofEuropeans,mainlyGermans,Italians andPoles.Currently,theHLAdiversityofthe Paraná popu-lation isrelated tothe ancient populations thatarrived in
thestateattheendofthe19thcentury.Accordingtoethnic self-classificationinthe2010census,70.06%ofthepopulation statedtheywereWhite,25.35%Mulattos,3.15%Black,1.19% Asiansand0.25%wereIndigenous.4
For patients on the waiting list for whom there is no HLA-identical sibling donor, a search for an unrelated hematopoieticstemcell(HSC)donorcanbeinitiatedusingthe BrazilianVoluntaryBoneMarrowDonorRegister(REDOME). Currently,morethan4,000,000potentialHSCdonorsare reg-istered on REDOME nationwide with 3858 patients on the BrazilianBone Marrow RecipientRegister(REREME)waiting for a compatible HLA match. Despite the increase in the number of HLA-typed volunteer donors, a significant per-centageofpatientswillstillnotbetransplantedinBrazilor internationally.5
Thesearchcanbeperformedfirstinthegroupthatshares the same ancestry where, theoretically, there is a greater chancetofindaHLA-compatibledonor;haplotypedifferences are foundbetweendifferentethnicgroups.Haplotypesthat arerareinsomepopulationsarecommoninothers.
The primary goal of the current study was to bring togetherpreviousstudiesonHLAfrequencies6–10andtoadd
estimations of the Hardy–Weinberg equilibrium and LD to understand the allele and haplotype structuresbetter ina representative sampleofbonemarrowvolunteerdonorsin ParanáState.
Methods
A total of 121,305 volunteer bone marrow donors in the LaboratoryofImmunogeneticsandHistocompatibility(LIGH) database ofthe Universidade Federaldo Paranáwere used inthisstudy.InBrazil,theREDOMEguidelinesrecommend that transplants should have high-resolution matches for
HLA-A, -B, -C, -DRB1 and -DQB1 (10/10 matches) between donorandrecipientbyDNA-basedmethods.Inthepresent studythelow/mediumresolutiontypingforHLA-A,-Band -DRB1requirementsforregisteringintheREDOMEwereused. Informedwrittenconsentwasobtainedfromallparticipants.5
Self-classificationandclassificationofancestryby surnames
evaluation that considers skin and hair pigmentation, eye melanizationandfacialfeaturessuchasnoseandlipshape. Theterm‘Brown’isanattempttosynthetizeavarietyof classi-ficationsthatemergesfromtheextensivemiscegenationthat existsintheBrazilianpopulation.11
Theancestry orracialbackgroundwasobtainedby self-assessmentinthe Brazilian censusofthe IBGE. Aiming to add efficiency to sampling strategies, surnames were also included,mainlyforItaliansandPolesasproxiesforethnic origin.Inthecontextofpopulationgenetics,certainsurnames are unique to a particular ethnic group and may contain informationrelatedtofamilygroupsorancestryoftheir bear-ers.Severalresearchersusedsurnamesastoolstoevaluate average consanguinity, to assess population isolation and structure,andtoestimatetheintensityanddirectionalityof migrations.12
This study sample was grouped according to self-classificationor inferredancestry accordingto surname as follows:LIGHdatabase(allgroups:n=121,305),Italian(n=212), Polish (n=277), Asian (n=1681),Afro-Brazilian descendants (n=3822),Mulatto(Brazilianmixedancestry; n=14,553)and Amerindians (n=704). Ancestry inferred by surnames was especiallyhelpfulfortheclassificationofItalianandPolish people.
Thealleleand haplotypefrequenciesofall groupswere comparedwithHLAfrequenciesforParanáStateavailablein theREDOMEdatabase.5
Bloodwas collected in 4-mLtubes containing ethylene-diaminetetraaceticacid(EDTA)anticoagulantinbloodbanks of the Centro de Hematologia e Hemoterapia do Paraná (Hemepar) or in the blood bank of Erasto Gaertner Hospi-tal(Curitiba,PR).Thecollectedmaterialwastransportedto the LIGH where DNA extraction was performed by one of thefollowingtechniques:salting-out,EZ-DNAkit(Biometrix),
iPREP® Purelink® gDNA Blood Kit (Life Technologies) or Biopur kit (Biometrix). HLA-A, HLA-B and HLA-DRB1 typing was performedbythepolymerase chainreaction-sequence specificprimers(PCR-SSP)methodusingtheMicro-SSP-ABDR Low/MediumResolutionkit(OneLambda)orthepolymerase chain reaction-sequence specific oligonucleotide (PCR-SSO) methodusingLabtypeSSOkits(OneLambda)forlow/medium resolutionaccordingtothemanufacturer’sinstructions. Sam-ples were read on a Luminex Labscan 100 apparatus(One Lambda).TheanalysiswasperformedusingtheHLAVisual (OneLambda)orHLAFusionsoftware(OneLambda).
Statisticalanalysis
The Arlequin software version 3.1113 was used to
cal-culate the allelic groups and haplotypes frequencies, the Hardy–WeinbergequilibriumandLD.
Results
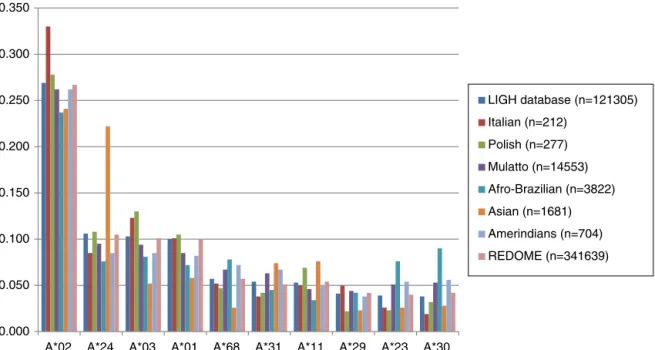
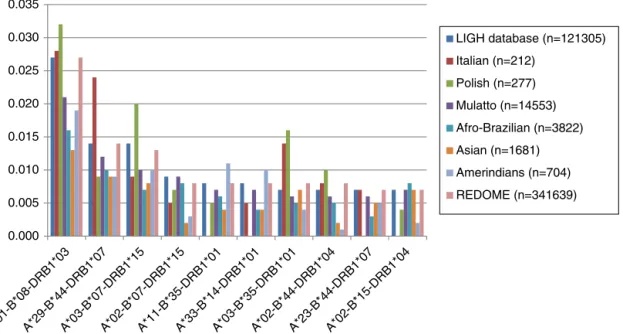
Thetenmostcommonallelicgroupsandhaplotypesinthe LIGHdatabaseareshowninFigures1–4.
The three HLA loci studied were in Hardy–Weinberg equilibrium for all groups except for the LIGH database:
HLA-A(p-value=0.000),HLA-B(p-value=0.000)andHLA-DRB1
(p-value=0.000);andAsian:HLA-A(p-value=0.000),HLA-B(p -value=0.000)andHLA-DRB1(p-value=0.001);Hardy–Weinberg equilibriumwassetforp-values>0.05(Table1).
AsfortheLIGHdatabase,thestrongestLDwasobserved for HLA-B*08-DRB1*03 (D′=0.540: r2=0.170: p-value=0.000). SeveralhaplotypesintheItaliangroupwereincompleteLD, e.g.HLA-A*69-B*39(D′=1.000:r2=0.075:p-value=0.000)aswell asforPolishdescendants:HLA-A*80-B*50(D′=1.000:r2=0.082:
0.000 0.050 0.100 0.150 0.200 0.250 0.300 0.350
A*30 A*23 A*29 A*11 A*31 A*68 A*01 A*03 A*24 A*02
LIGH database (n=121305)
Italian (n=212)
Polish (n=277)
Mulatto (n=14553)
Afro-Brazilian (n=3822)
Asian (n=1681)
Amerindians (n=704)
REDOME (n=341639)
0.000 0.020 0.040 0.060 0.080 0.100 0.120 0.140 0.160
B*39 B*14 B*40 B*18 B*08 B*07 B*51 B*15 B*44 B*35
LIGH database (n=121305)
Italian (n=212)
Polish (n=277)
Mulatto (n=14553)
Afro-Brazilian (n=3822)
Asian (n=1681)
Amerindians (n=704)
REDOME (n=341639)
Figure2–HLA-BtenmostfrequentallelicgroupsofLaboratoryofImmunogeneticsandHistocompatibilitydatabase comparedwithethnicgroupsaswellasBrazilianVoluntaryBoneMarrowDonorRegister(REDOME)dataforthestateof Paraná.
0.000
DRB1*13 DRB1*07 DRB1*04 DRB1*11 DRB1*01DRB1*15 DRB1*03 DRB1*08DRB1*14 DRB1*16 0.020
0.040 0.060 0.080 0.100 0.120 0.140 0.160 0.180 0.200
LIGH database (n=121305)
Italian (n=212)
Polish (n=277)
Mulatto (n=14553)
Afro-Brazilian (n=3822)
Asian (n=1681)
Amerindians (n=704)
REDOME (n=341639)
Figure3–HLA-DRB1tenmostfrequentallelicgroupsofLaboratoryofImmunogeneticsandHistocompatibilitydatabase comparedwithethnicgroupsaswellasBrazilianVoluntaryBoneMarrowDonorRegister(REDOME)dataforthestateof Paraná.
Table1–Hardy–Weinbergequilibrium(p-value>0.05)perlocusineachoftheethnicgroupsanalyzed.
HLA- LIGHdatabase (n=121,305)
Italian (n=212)
Polish (n=277)
Mulatto (n=14,553)
Afro-Brazilian (n=3822)
Asian (n=1681)
Amerindians (n=704)
A 0.000 0.240 0.204 0.143 0.658 0.000 0.320
B 0.000 0.837 0.169 0.134 0.846 0.000 0.405
DRB1 0.000 0.364 0.693 0.075 0.167 0.001 0.384
0.000
A*01-B*08-DRB1*03A*29-B*44-DRB1*07A*03-B*07-DRB1*15A*02-B*07-DRB1*15A*11-B*35-DRB1*01A*33-B*14-DRB1*01A*03-B*35-DRB1*01A*02-B*44-DRB1*04A*23-B*44-DRB1*07A*02-B*15-DRB1*04 0.005
0.010 0.015 0.020 0.025 0.030 0.035
LIGH database (n=121305)
Italian (n=212)
Polish (n=277)
Mulatto (n=14553)
Afro-Brazilian (n=3822)
Asian (n=1681)
Amerindians (n=704)
REDOME (n=341639)
Figure4–HLA-A-B-DRB1tenmostfrequenthaplotypesofLaboratoryofImmunogeneticsandHistocompatibilitydatabase comparedwithethnicgroupsandwiththeavailableBrazilianVoluntaryBoneMarrowDonorRegister(REDOME)dataforthe stateofParaná.
p-value=0.000).ConcerningtoMulattogroup,thestrongestLD valueswereobservedforhaplotypesHLA-A*01-B*59(D′=1.000:
r2=0.000: p-value=0.001) and HLA-B*59-DRB1*03 (D′=1.000:
r2=0.000: p-value=0.000)that were incomplete LD.As for Afro-Brazilians, the haplotype HLA-B*82-DRB1*11(D′=0.870:
r2=0.010: p-value=0.000) showed the strongest LD. Asian descendantspresenteddifferenthaplotypesincompleteLD, includingHLA-A*69-B*35(D′=1.000:r2=0.003:p-value=0.003). TheAmerindiansgroupalsoshowedhaplotypesincomplete LDsuchasHLA-A*80-B*15(D′=1.000:r2=0.006:p-value=0.005); LDwassetforp-values>0.05(Table2).
Discussion
HLAgenescodeforcellsurfaceproteinsthatactas biochem-icalsignatures.TheseHLAmarkersareclassifiedassixtypes labeledHLA-A,-B,-C, -DR,-DQ, and-DP. Atotal ofatleast ten genescode forthe HLAsystem givingeach person an immunesignaturedisplayingpeptides(foreignorself)forT lymphocytes.Therearemorethanhundredvariantsforeach ofthesegenesresultinginbillionsofcombinationsor indi-vidualHLAtypes that pose achallenge for cell and tissue transplantation.14
However, HLA diversity is a key factor of the adaptive immuneresponsedirectedtowardapathogenandan effec-tiveimmuneresponse,amongotherfactors,dependsonthe HLA alleles a person carries. HLA alleles differ mainly at exons 2 and 3 for class I genes, and exon 2 for class II genes. These exonscode for the peptide-bindingregion of HLA molecules. It has been assumed that polymorphisms of HLA alleles are functional as different HLA molecules bind differentsets ofrelated pathogen-derived peptides to presentthem toT-cellreceptors.Ithasalsobeen observed that HLA alleles that have a higher binding affinity to a
particular pathogenic peptide are better at clearing infec-tions.AccordingtoSanchez-Mazas, thedistributionofHLA allelesindifferentpopulationsmaybeaconsequenceofthis functionalpolymorphism.15AstoHLAalleles,heterozygous
individualshaveawiderpeptide-bindingregionrepertoireand hencetheymaymountabetterimmuneresponsetoa spe-cificpeptideepitopeofapathogencomparedtohomozygous individuals.
From thebiologicalpointofview,toknowwhich alleles are present inapopulationand their frequenciescould be informativeabouttheevolutionofHLApolymorphismsand thepopulation’sgeneticsusceptibilitytodisease.Forclinical transplantationpurposes,however,thisknowledgeprovides a morerealistic overviewofa populationconcerning their mostfrequentHLAallelesandhaplotypeswiththis informa-tion beingvery usefulinestimatingthe chancesoffinding a compatible donorfor a patient on the waiting list for a hematopoietic stem cell transplantation (HSCT). It is well known that a significant degree ofHLA matches between donorandrecipientimprovesoveralltransplantsurvival,rates ofengraftmentandreducestheincidenceofacuteandchronic GVHD.
Thethreemostcommonallelicgroups,HLA-A(-A*02,-A*24 and-A*03)andHLA-B(-B*35,-B*44and-B*15),foundintheLIGH databasecorroboratetheoverallfrequencyofthedata avail-ableintheREDOMEforthestateofParaná.5Thesefindingsare
inagreementwithapreviousstudyforthepopulationofthe northernregionofParanáState16aswellaswithaprevious
surveyconductedinthepopulationofCuritiba.1
rev
bras
hema
tol
hemoter.
2
0
1
7;
3
9(3)
:229–236
Table2–DescriptionofthestrongestlinkagedisequilibriumvaluesforthehaplotypesHLA-A/-B,HLA-A/-DRB1andHLA-B/-DRB1ineachoftheanalyzedgroups.
HLA- A*24-B*59 A*29-DRB1*07 B*08-DRB1*03
LIGHdatabase D′=0.441 D′=0.220 D′=0.540
(n=121,305) r2=0.001 r2=0.010 r2=0.170 p=0.000 p=0.000 p=0.000
HLA- A*69-B*39 A*34-B*08 A*02-B*45 A*33-B*58 A*03-B*47 A*02-B*56 A*69-DRB1*01 A*34-DRB1*04 B*45-DRB1*11 B*47-DRB1*15 B*48-DRB1*09
Italian D′=1.000 D′=1.000 D′=1.000 D′=1.000 D′=1.000 D′=1.000 D′=1.000 D′=1.000 D′=1.000 D′=1.000 D′=1.000
(n=212) r2=0.075 r2=0.027 r2=0.010 r2=0.282 r2=0.017 r2=0.010 r2=0.023 r2=0.023 r2=0.020 r2=0.029 r2=0.665 p=0.000 p=0.001 p=0.044 p=0.000 p=0.007 p=0.044 p=0.002 p=0.002 p=0.003 p=0.001 p=0.000
HLA- A*80-B*50 A*36-B*53 A*25-B*56 A*31-B*47 A*36-DRB1*11 A*80-DRB1*14 B*47-DRB1*13 B*81-DRB1*12 B*56-DRB1*12
Polish D′=1.000 D′=1.000 D′=1.000 D′=1.000 D′=1.000 D′=1.000 D′=1.000 D′=1.000 D′=1.000
(n=277) r2=0.082 r2=0.199 r2=0.034 r2=0.042 r2=0.010 r2=0.040 r2=0.010 r2=0.160 r2=0.080 p=0.000 p=0.000 p=0.000 p=0.000 p=0.010 p=0.000 p=0.010 p=0.000 p=0.000
HLA- A*01-B*59 A*43-DRB1*07 B*59-DRB1*03
Mulatto D′=1.000 D′=0.430 D′=1.000
(n=14,553) r2=0.000 r2=0.000 r2=0.000 p=0.001 p=0.020 p=0.000
HLA- A*69-B*55 A*69-DRB1*11 B*82-DRB1*11
Afro-Brazilian D′=0.817 D′=0.590 D′=0.870
(n=3822) r2=0.116 r2=0.000 r2=0.010 p=0.000 p=0.000 p=0.000
HLA- A*69-B*35 A*11-B*67 A*68-B*78 A*69-DRB1*04 B*78-DRB1*13 B*73-DRB1*04
Asian D′=1.000 D′=1.000 D′=1.000 D′=1.000 D′=1.000 D′=1.000
(n=1681) r2=0.003 r2=0.036 r2=0.011 r2=0.001 r2=0.002 r2=0.006 p=0.003 p=0.000 p=0.000 p=0.029 p=0.005 p=0.000
HLA- A*80-B*15 A*02-B*56 A*02-B*47 A*03-B*54 A*74-B*78 A*80-DRB1*16 B*54-DRB1*15 B*78-DRB1*07
Amerindians D′=1.000 D′=1.000 D′=1.000 D′=1.000 D′=1.000 D′=1.000 D′=1.000 D′=1.000
(n=704) r2=0.006 r2=0.004 r2=0.004 r2=0.008 r2=0.028 r2=0.020 r2=0.010 r2=0.010 p=0.005 p=0.018 p=0.018 p=0.001 p=0.000 p=0.000 p=0.000 p=0.010
HLA-A*11andHLA-B*40werethethirdmostcommonallelic groupsintheAsiangroup,thusdisagreeingwiththefindings ofBardietal.16whoreportedHLA-B*40asthemostprevalent
inthisgroup.
ClassIIHLA-DRB1*13,-DRB1*07and-DRB1*04werethemost frequent allelic groups in the LIGH database, thus agree-ing,inpart, withdataavailableinREDOMEforthestateof Paraná5 that reported HLA-DRB1*11 as the most common.
TheywerealsothemostcommonallelicgroupsofMulattos andAmerindians.HoweverinAsianstheHLA-DRB1*15allelic groupwasthesecondmostcommon,inagreementwitha pre-viousstudycarriedoutinthepopulationofnorthernParaná state.16
The three most frequent haplotypes ( HLA-A*01-B*08-DRB1*03, HLA-A*29-B*44-DRB1*07, HLA-A*03-B*07-DRB1*15) foundintheLIGHdatabase areinlinewiththethreemost common haplotypes found in REDOME for the state of Paraná.5 Bicalho et al.1 and Ruiz et al.17 reported similar
results.Thesehaplotypeswerealsothemostfrequentamong Mulattos.ThemostcommonhaplotypeinAsians( HLA-A*24-B*52-DRB1*15–datanotshown)wasalsothemostfrequent inthestudybyBardietal.forthisethnicgroup.16
Table1showsthatallgroupsareinHardy–Weinberg equi-libriumfortheHLA-A,-Band-DRB1lociexceptfortheLIGH databaseandtheAsiangroupwherethenumberofobserved heterozygotes is smaller than expected. This fact possibly occursbecausetheLIGHdatabaseiscomposedofdonorsfrom differentpartsofParanáStateorlivingincitieswithhigh con-centrationsofpeoplesharingthesameancestralbackground (Asian) that determine regional differences inthe popula-tion.Additionally,theconditionofstrictlyrandommarriages, whichisfundamentaltotheaccuracyoftheHardy–Weinberg equilibrium,maynotoccur.
TheobservedLDbetween HLAalleles providesvaluable informationaboutthestructureofthepopulation.Itmaycast somelightonhowevolutionaryforces,suchasnatural selec-tion,geneconversion,mutations,affectHLAgenefrequencies. Further,it canpavethe wayforabettercomprehensionof allelicinteractionsthatgeneratethealleliccombinations,or haplotypes,indifferentpopulationsbecauseoftheirabilities tobindtopeptidesofpathogensintheenvironment.Hence, certainhaplotypecombinationsaremorelikelytobefound togetherthanothers.Theseassociationsofallelescanarise throughtheirco-evolutionaryinteractionwithpathogens.In clinicalsettings,individualconservedhaplotypeswithin eth-nicgroupscouldbeakeyfactortoincreasetheoddsoffinding acompatibleHLAdonor.
ThehaplotypesinLDHLA-A*34-B*08(D′=1.000)and
HLA-A*03-B*47(D′=1.000)fortheItaliangroupandHLA-A*36-B*53 (D′=1.000) and HLA-A*36-DRB1*11 (D′=1.000) for the Polish group are in agreement withthe resultsobtained by San-tosforCaucasians. Moreover,the HLA-A*11-B*67(D′=1.000) haplotypeinthe Asian group isinline withthe resultsof Santos.18
Complete LD (D′=1.000)was widelyfound between the analyzedethnicgroups, indicatingthe existenceoflinkage betweenoneallelicgroupandanother thereby characteriz-ingaparticularhaplotype.Whatisstrikingisthecorrelation (r2)betweenallelicgroupswithcomplete LDwhichis0.000 orveryclosetothis.Thisfactindicatesthattheseparticular
allelic groups are present at completely different frequen-cies,forexample,theMulattogroupshowedHLA-A*01-B*59
(D′=1.000andr2=0.000)withtherespectiveHLA-A*01(0.085) andHLA-B*59(0.000034)frequencies.So,amongMulattosin thepopulationofParanástate,HLA-B*59isalmostalways asso-ciatedwithHLA-A*01.Nevertheless,thecontraryisnottruefor
HLA-A*01.Thistypeofanalysisisofutmostimportanceinthe searchforbonemarrowvolunteerdonors,especiallyforthe HLAlaboratoryteamwhenfacinggenotypingambiguities.
LD,thenon-randomassociationofallelesatdifferentloci, canprovidevaluableinformationonthestructureof haplo-types.Thisisoftenthebasisforevaluatingtheassociationof geneticvariationswithhumantraitsinunrelatedsubjects.19
TheknowledgeofHLAgenesandhaplotypesinthepopulation structureisrelevantnotonlyforgeneticstudiesbutin dis-easesusceptibilityandorgantransplantation.Inregardstoa mixedpopulationlikeBrazilians,thesizeandgeneticdiversity ofREDOMEshouldaccomplishtherequirementthatthe num-berofdonorsregisteredinthenationaldatabasecanensure thateachpatientonthewaitinglistfindsacompatibledonor asearlyaspossible.HLAfrequenciescanbeusedtoestimate theprobabilityoffindingidenticalunrelatedvolunteerbone marrowdonors.AsallelesatcloselylinkedHLAlocitendto beassociatedatthepopulationlevel(e.g.inLD)itiscommon sensethatpatientswithHLAhaplotypesinstrongLDhavea higherprobabilityoffindingHLA-identicalunrelateddonors thanothers.20
Conclusions
AllstudiedgroupswereinaccordancewiththeHLAgenetic profile ofthe Paranápopulation data foundin REDOMEin respecttotheallelicgroupsandhaplotypefrequenciesexcept fortheAsiangroup.Regionalgeneticdifferencesmay influ-encetheHardy-Weinbergequilibrium21asseenintheAsian
groupandtheLIGHdatabase.TheanalysisofLDofHLA hap-lotypesisoftheutmostimportancetofindcompatiblebone marrowvolunteerdonorsandshouldbecarefullyconducted considering the group ancestry and regional peculiarities. Theseresultscontributetothestudyofthepopulation genet-icsoftheParanáState.TheinformationonLDbetweenHLA allelicgroupsmayshowitsvalueandpracticalusein trans-plantationgeneticswhenthepurposeistofindcompatible bonemarrowdonors.
Conflict
of
interest
Theauthorsdeclarenoconflictsofinterest.
Acknowledgements
r
e
f
e
r
e
n
c
e
s
1. BicalhoMG,RuizTM,CostaSM,ZacariasFR.HaplótiposHLA maisfrequentesemdoadoresvoluntáriosdemedulaósseade Curitiba,Paraná.RevBrasHematolHemoter.2002;24(4):306–9. 2. VogelF,MotulskiAG.Humangenetics:problemsand
approaches.3thedBerlin:SpringerScience&Business Media;1997.p.851.
3. LewontinRC,KojimaK.Theevolutionarydynamicsof complexpolymorphisms.Evolution.1960;14:458–72. 4. Etnias–EstadodoParaná.Availablefrom:http://www.
cidadao.pr.gov.br/modules/conteudo/conteudo.php?conteudo =77.[cited13February2015].inpress.
5. RedeBrasildeImunogenética.Availablefrom:
http://imunogenetica.org/resultados/perfil-genomico-do-redome-rereme/[cited12December2014].inpress. 6. CostantinoPR,BicalhoMG.Diversidadesolidáriaem
ítalo-brasileirosdosudoestedoParaná:descric¸ãodas frequênciasHLAgrupoalélicasehaplotípicas.In:XIII CongressodaSociedadeBrasileiradeTransplantedeMedula Óssea,2009,Curitiba–PR.RevBrasHematolHemoter.2009.p. 31.Anais.p.29.
7. ZeckSC,ZacariasFR,BicalhoMG.Frequênciaalélicae haplotípicadoslocosHLA-A,-Be-DRB1empopulac¸ão brasileiracomdescendênciapolonesa.In:XIIICongressoda SociedadeBrasileiradeTransplantedeMedulaÓssea,2009, Curitiba–PR.RevBrasHematolHemoter.[online].2009[cited 2015-02-05].Availablefrom:http://www.sbtmo.org.br/ userfiles/anais/arquivo20140514170041.pdf
8. CostantinoPR,SilvaWA,BicalhoMG.Frequênciasdosgrupos alélicosehaplótiposdoslocosHLA-A,-Be-DRB1emuma amostramiscigenadadapopulac¸ãoparanaense.In:XVIII CongressodaSociedadeBrasileiradeTransplantedeMedula Óssea,2014,BeloHorizonte-MG.SBTMO2014XVIII CongressodaSociedadeBrasileiradeTransplantedeMedula Óssea.2014.Anais.p.68.
9. CostantinoPR,SilvaWA,BicalhoMG.Descric¸ãodas
frequênciasgrupoalélicasehaplotípicasdoslocosHLA-A,-B e-DRB1empopulac¸ãoafrodescendente.In:XIIICongresso BrasileirodeTransplantes.2013[cited2015-02-05],Available from:http://www.sistemaparaevento.com.br/evento/ abto2013/trabalhosaprovados/naintegra/577 10.ZeckSC,CostantinoPR,BicalhoMG.Descric¸ãodas
frequênciasgrupoalélicasehaplotípicasdoslociHLA-A,-Be -DRB1empopulac¸ãoparanaensecomancestralidade
oriental.In:XVICongressodaSociedadeBrasileirade TransplantedeMedulaÓssea,2012,RibeirãoPreto-SP.Rev BrasHematolHemoter.v.34.Anais.p.19-19.2012. 11.PenaSD,DiPietroG,Fuchshuber-MoraesM,GenroJP,Hutz
MH,KehdyFD,etal.Thegenomicancestryofindividuals fromdifferentgeographicalregionsofBrazilismoreuniform thanexpected.PLoSOne.2011;6(2):e17063.
12.DarluP,BloothooftG,BoattiniA,BrouwerL,BrowuerM, BrunetG,etal.Thefamilynameassocio-culturalfeatureand geneticmetaphor:fromconceptstomethod.HumBiol. 2012;84(2):169–214.
13.ExcoffierL,LavalG,SchneiderS.Arlequinver.3.0:an integratedsoftwarepackageforpopulationgeneticsdata analysis.EvolBioinformOnline.2005;1:47–50.
14.BodmerW,McKieR.TheBookofMan[Chapter7];1997,pp 109–124.
15.Sanchez-MazasA,Fernandez-Vi ˜naM,MiddletonD, HollenbachJA,BuhlerS,DiD,etal.Immunogeneticsasa toolinanthropologicalstudies.Immunology.
2011;133(2):143–64.
16.BardiMS,JarduliLR,JorgeAJ,CamargoRB,CarneiroFP, GelinskiJR,etal.HLA-A,BandDRB1alleleandhaplotype frequenciesinvolunteerbonemarrowdonorsfromthenorth ofParanastate.RevBrasHematolHemoter.2012;34(1):25–30. 17.RuizTM,DaCostaSM,RibasF,LuzPR,LimaSS,BicalhoMG.
Humanleukocyteantigenallelicgroupsandhaplotypesina Braziliansampleofvolunteerdonorsforbonemarrow transplantinCuritiba,Parana,Brazil.TransplProc. 2005;37(5):2293–6.
18.SantosPS.Análisededesequilíbriodeligac¸ãoede frequênciashaplotípicasHLA-A,HLA-BeHLA-DRB1na amostradedoadoresvoluntáriosdemedulaósseadoLIGH [monografia].Curitiba:UniversidadeFederaldoParaná;2005. p.87.
19.SchaidDJ.Linkagedisequilibriumtestingwhenlinkagephase isunknown.Genetics.2004;166(1):505–12.
20.VojvodicS,PopovicS,Macukanovic-GolubovicL.Implications ofHLAlinkagedisequilibriumphenomenoninfinding unrelatedvolunteerbonemarrowdonors.ResearchGate. 2007;3–4:178–82.