Arbuzov Institute of Organic and Physical Chemistry Academy of Sciences of the Republic of Tatarstan. As we celebrate the 150th anniversary of the theory of chemical structure of organic compounds, we honor A.M.
CONGRESS SCHEDULE
BUTLEROV CONGRESS SCIENTIFIC PROGRAMME
Minkin (Russia)
- Yu. Antipin (Russia)
- S. Miller (USA)
- F. Vasilevskiy (Russia)
- Olivucci (Italy)
- A. Katsyuba (Russia)
- V. Kazakov (Russia)
- I. Madzhidov (Russia)
- V. Vasilyev (Russia)
- Yu. Balakina (Russia)
- K. Kadirov (Russia)
- F. Khlebnikov (Russia)
- W. Bruce (UK)
- C. Larock (USA)
- Thomas (UK)
- P. Ananikov (Russia)
- F. Tietze (Germany)
- Bonifazi (Belgium)
- A. Balova (Russia)
- A. Kuznetsov (Russia)
- A. Kuropatov (Russia)
- R. Kurbangalieva (Russia)
- K. Saikia (India)
- V. Roeschenthaler (Germany)
- D. Shutalev (Russia)
- R.D. Correia (Brazil)
- G. Fedenok (Russia)
- F. Mironov (Russia)
- Hissler (France)
- Mikolajczyk (Poland)
- G. Kulinkovich (Estonia)
- Hey-Hawkins (Germany)
- P. Kukhar (Ukraine)
- Allen (USA)
- Suzenet (France)
- L. Fedushkin (Russia)
- M. Prokhorov (Russia)
- V. Zaitsev (Russia)
- A. Miluykov (Russia)
- Kawashima (Japan)
- L. Odinets (Russia)
- V. Parfenova (Russia)
- Saloutin (Russia)
- M. Muzafarov
- N. Gavrilov (Russia)
- Masson (France)
- J. Knöelker (Germany)
- O. Bachurin (Russia)
- Romeo (Italy)
- R. Metz (Germany)
- R. Milaeva (Russia)
- V. Kudryavtsev (Russia)
- Z. Voloshin (Russia)
- Yu. Fedorov (Russia)
- Galkina (Russia)
- Semenov (Russia)
- Inokuchi (Japan)
- B. Nawrozkij (Russia)
- Gorshkova (Russia)
- S. Fedorov (Russia)
- Lammertsma (Netherlands)
- E. Nikolaev (Russia)
- A. Baltina (Russia)
- G. Gabibov (Russia)
- König (Germany)
- S. Antipin (Russia)
- P. Gromov (Russia)
- P. Fedin (Russia)
- A. Fedorova (Russia)
- A. Evtugyn (Russia)
- B. Melnikova (Russia)
- A. Karpichev (Ukraine)
- Gorbatchuk (Russia)
- A. Kostin (Russia)
- L. Selektor (Russia)
- N. Ushakov (Russia)
- V. Yakimansky (Russia)
- G. Mirochnik (Russia)
- N. Chvalun (Russia)
- N. Kataeva (Russia)
- A. Mikhalitsyna (Russia)
- Z. Vatsadze (Russia)
- A. Livshits (Russia)
- S. Oshchepkov (Russia)
- Semionova (Russia)
- Makosza (Poland)
- Gevorgyan (USA)
- V. Gulevskaya (Russia)
- D. Shteingarts (Russia)
- I. Belen’kii (Russia)
- Galkin (Russia)
- V. Pestov (Russia)
- A. Utepova (Russia)
- S. Lyakhovnenko (Russia)
- V. Aksenov (Russia)
- B. Sheremetev (Russia)
- M. Starosotnikov (Russia)
- U.W. Maes (Belgium)
- J. Collins (USA)
- A. Dyakonov (Russia)
- D. Gridnev (Japan)
- S. Novikov (Russia)
- V. Rostovskii (Russia)
- V. Ivanova (Russia)
- N. Kornev (Russia)
- Yu. Sukhorukov (Russia)
- D.Kiselev (Russia)
- R. Akhmetova (Russia)
- Trifonov (Russia)
- Korshin (Israel)
- G. Zlotin (Russia)
- P. Krasnov (Russia)
Cyclization of functionalized (buta-1,3-diynyl)arenes in the synthesis of endiyne systems fused to heterocycles. Design and synthesis of membrane-anchored bacterial inhibitors as potential drugs for the treatment of Alzheimer's disease.
Abstracts
NMR, EPR, MRI
AFM, SPM
Mollet E. Leemans
- L. Eremenko
The report examines examples of the use of various organic ligand blocks in the chemical assembly of polynuclear metal architectures. Arbuzov Institute of Organic and Physical Chemistry, Kazan Scientific Center of the Russian Academy of Sciences, Department of Physical Chemistry, Kazan, Russia.
A. Suturina 1
Studying the electron density distribution in organoselenium compounds and its complexes using modern quantum-chemical approaches (Quantum Theory of Atoms in Molecules, Interactive Quantum Atom) revealed the special features of the selenium atom as an electron donor. It was shown that the changes in the properties of the selenium lone pair depend significantly on the type of electron acceptor.
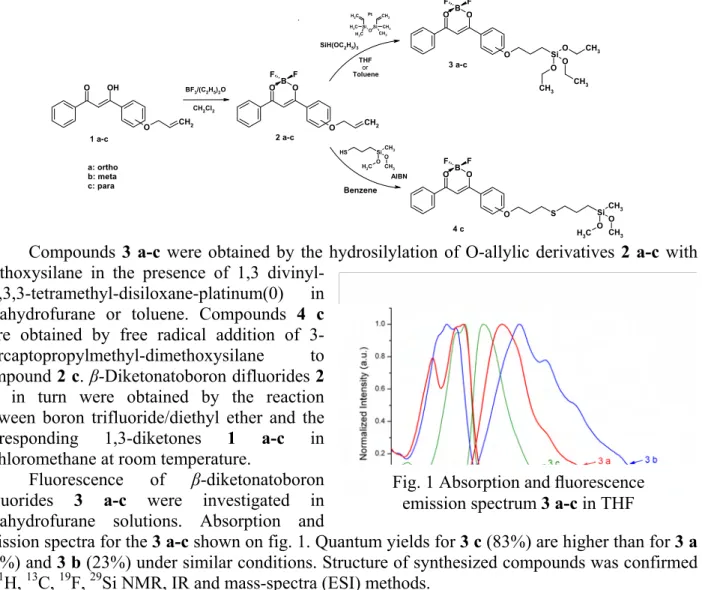
Yu. Balakina
1 - The Institute of Organic and Physical Chemistry, Kazan Scientific Center of the Russian Academy of Sciences, Kazan, Russia.
K. Kadirov 1
S. Konev
Detailed interpretation of the experimental Fourier-transform IR spectra of 1–4 was performed on the basis of the calculation of their vibrational spectra. DEHYDRODIMERIZATION OF PROPYNOL Institute of Chemical Kinetics and Combustion, Siberian Branch of the Russian Academy of Sciences, Novosibirsk, Russia.
G. Fedenok
- Sushev A.N. Kornev
The strong band 1597 cm-1 shows clear changes of the optical density depending on the substituents in the aromatic ring. In the 13C NMR spectra of synthesized ketoximes, the signal of the CH3 group is expressed in the 18-19 ppm.
SUBSTITUTED TETRAHYDROINDOLE DERIVATIVES
Through these works, we investigate the possibility of using products of chlorination of ethylbenzene with subsequent chloromethylation of received monochloroethylbenzenes in the synthesis of derivatives of styrene. It has been determined that under optimal conditions for chlorination, the maintenance of monochloroethylbenzenes in chlorination products is 55.8%.
Azizov N.S. Nabiyev
This program was used for the construction of the initial structure of solvent molecules (acetone) around ((CH3)3P and (CH3)3PCS2) molecules. Theoretical Organic Chemistry: Poster Presentation P-12 USING THE THEORETICAL INFORMATION TOPOLOGICAL INDEXES FOR CALCULATION OF.
Khabibullin
TERTIARY PHOSPHINES WITH UNSATURATED CARBOXYLIC ACIDS AND THEIR DERIVATIVES Kazan Federal University, Department of Chemistry, Kazan, Russia A.V. 1 - Kazan Federal University, Department of Chemistry, Kazan, Russia 2 - Kazan Federal University, Department of Physics, Kazan, Russia A.V.
M. Aminova 2
Theoretical Organic Chemistry: Poster Presentation P-14 PROBING THE REACTION MECHANISM OF TERTIARY PHOSPHINES WITH THEIR PANATE. OF THE CHEMICAL STRUCTURE OF SUBSTANCE 1 - Kazan State University of Energy Engineering, School of Materials Science and Technology, Kazan, Russia.
S. Sirotkin 1
- G. Uryadov 1
This approach describes reactions in the gas phase where the energy of molecules is determined by translational motion. In the liquid phase, the reagent molecules are in a "cage" of solvent molecules.
B. Krivolapov
Theoretical Organic Chemistry: Poster Presentation P-17 EXPERIMENTAL AND THEORETICAL INVESTIGATION OF NON-COVALENT INTERACTIONS AND CRYSTAL STRUCTURES OF SOME COMPOUNDS. Chiral” Organic Chemistry: Oral report O-101 INTRAMOLECULATE HOMOLYTIC C-ALKENYLATION VERSUS REDUCTIVE HETEROCYCLIZATION Bridged IN.
Korshin L. Konstantinovski
Bilokin
The phenomenon of chirality is considered one of the central topics in modern organic chemistry. Without questioning the importance and multifaceted character of the manifestations of chiral discrimination in living systems, we want to draw the attention of organic practitioners to some of the less well-known, but no less interesting aspects of the phenomena of chiral recognition.
E. Siyutkin
We have shown that efficient CR of heterocyclic amines can be achieved while using 2-arylpropionyl and N-protected amino acids derivatives as solvents. To explain the peculiarities of CR process in the course of acylation of racemic amines with chiral acyl chlorides, we used quantum chemical calculations (DFT-D/B3LYP/TSVP, COSMO solvation model) and NMR spectroscopy.
Karasik 1 S. Balueva 1
A. Rubtsova
New S-germyl and S-plumbyl derivatives of aryldithiophosphonic acids were prepared based on reactions of ammonium salts of the corresponding acids with triorganylchlorogermanes, chloroplumbane and diphenyl dichloroplumbane. S-germyl and S-plumbyl derivatives of aryldithiophosphonic acids with chiral centers have notable antimicrobial activities.
PROTECTED (S)-AMINO ACYL CHLORIDES
Chiral” Organic Chemistry: Presentation of the poster P-254 INVERSION OF STEREOSELECTIVITY IN THE ACELYATION OF HETEROCYCLIC AMINES Z. Tetracoordinated organophosphorus thioacids, their ammonium salts and esters containing chiral centers are of interest from the point of view of fundamental organic chemistry and the development of simple methods of obtaining new products by biological activity.
V. Orlov
IN ASYMMETRICAL CATALYTIC PROCESSES 1 - Ryazan Branch of Moscow State Open University, Department of Physics and Chemistry, Ryazan, Russia. 2 - Ryazan State University, Department of Chemistry, Ryazan, Russia 3 - Leibniz-Institut fur Katalyse an der Universitat Rostock e.V., Rostock, Germany.
Shiryaev 1 K.N. Gavrilov 2
Chiral” Organic Chemistry: Poster Presentation P-261 BROMINE-LITHIUM EXCHANGE UNDER IN SITU CONDITIONS: AN EFFICIENT ONE-POT. SYNTHESIS OF CHIRAL LIGANDS FOR TRANSITION METAL CATALYSIS Institute of Organic Chemistry, University of Cologne, Cologne, Germany.
B. Kasenova 2
OHAUS- Japan (scales), SHIMADZU - Japan (the widest range of analytical equipment), SARTORIUS - Germany (laboratory equipment), IMPLEN - Germany (spectrophotometers of a new generation with patented technology "drop sampling"), SHINKO - Japan (weighing equipment) , METTLER TOLEDO – Switzerland (scales, pH meters, analytical instrumentation for thermal analysis, titrators, electrochemical equipment, laboratory), DAIHAN – Korea (laboratory equipment), ULAB – Germany (advanced laboratory equipment), J.T. Baker – Netherlands-USA (high purity chemicals, pharmacopoeia standards and basic principles of polyethylene glycol figs), RHEOTEST – Germany (viscometers and rheometers rotamers), HAIER – Germany (biochemical equipment), TRILOGICA – Germany (viscometers, rheometers, moisture analyzers, dilatomers , bursting machines,) , HUBER – Germany (the widest range of thermostats), LEKI – China (scales and spectrophotometers), SNOL-THERM – Russia (muffle, tube, tube furnaces, furnaces), CEMICO – Russia (electrochemical devices), ESCO – Singapore (chemical) and bioreactors), INFRASPAK-ANALYT – Russia (electrochemical devices), UNICO – (spectrophotometers), LUMEX».
Green Lee
Advanced Methods of Organic Synthesis: Plenary Lecture PL-9 THE NATURE OF THE CATALYST IN THE TRANSITION-METAL-CATALYZED CARBON-HETEROATOM AND. Advanced Methods of Organic Synthesis: Oral Report O-15 CYCLISATION OF FUNCTIONALIZED (BUTA-1,3- DIYNYL)ARENES IN ENDIINE SYNTHESIS.
A. Berdnikov 1
Advanced Methods of Organic Synthesis: Oral Report O-20 A NOVEL REORDERING OF QUINOXALIN-2(1H)-ONE FOR THE SYNTHESIS. Prins cyclization has long been used for the synthesis of oxygen heterocyclic compounds, especially substituted tetrahydropyrans.
D. Shutalev Fesenko
Advanced Methods of Organic Synthesis: Oral Report O-23 RECENT ADVANCE IN THE SYNTHESIS OF TETRAHYDRO- AND DIHYDRO-1H-1,3-DIAZEPINE-2-. Advanced Methods of Organic Synthesis: Oral Report O-24 REGIO- AND STEREOSELECTIVE HEK ARYLYATION OF CINNAMATES AND ALLYLAMINE.
Prediger 1 J.G. Taylor 2
Institute of Chemical Kinetics and Combustion, Siberian Branch of the Russian Academy of Sciences, Novosibirsk, Russia. It was established that, depending on reaction conditions and substrate structure, stabilization of the intermediate takes place either with the preservation of the heterocycle size and the formation of indazole derivatives or with cycle expansion and the formation of cinnoline derivatives.
S. Vshivkova
1 - Institute of Technical Chemistry, UB RAS, Perm, Russia 2 - Perm State University, Chemical Department, Perm, Russia 3 - Institute of Natural Sciences of Perm University, Perm, Russia G.G. Previously, we synthesized a series of 4,6-di-substituted pyrimidines with thiophene and pyrrole groups attached to the pyrimidine core via α-C atom of heterocycle [3].
Aniskov
- Barabanov 1 A.V. Polukhin 1
- S. Baranovskyy
So, the construction of new antioxidants based on calix[4]resorcinols seems to be very promising. St. Petersburg State Institute of Technology (Technical University), Department of Organic Chemistry, St. Petersburg, Russia.
S. Bogachenkov
- A. Litvinov
Advanced Methods of Organic Synthesis: Poster Presentation P-26 NEW DIFFERENTLY SUBSTITUTED IZATIN DERIVATIVES: SYNTHESIS AND REACTIONS Z. Advanced Methods of Organic Synthesis: Poster Presentation P-28 EXPERIMENTAL AND THEORETICAL INVESTIGATION OF BASE-CATALYZED.
A. Cheshkov
HYDROXYDECAHYDROQUINAZOLINE-2-THIONE Moscow State Academy of Fine Chemical Technology, Institute of Organic Chemistry, Moscow, Russia. Advanced Methods of Organic Synthesis: Poster Presentation P-31 N-ACYLBENZOTRIAZOLE-MEDIATED MICROWAVE-ASSISTED SYNTHESIS OF PROTECTED AND NOVEL UNPROTECTED FERROCENOYLAMIDOAMINO ACIDS 1 - Anadolu University, Department of Chemistry, Eskisehir, Turkey.
S. Kulikov
According to X-ray analysis, the minor product is a fourteen-membered bis-dithioacetal with transposition of the substituents. The stereochemical study of the oxidation of seven-membered cyclic dithioacetals to monosulfoxides and disulfoxides was also carried out.
Hayvali
Advanced Methods of Organic Synthesis: Poster Presentation P-37 SYNTHESIS OF 2, 3-SECOTRITERPENE MONO-AND DICONJUGATES WITH AMINOLOCHOLS 1 - Institute of Technical Chemistry, Biologically Active Compounds, Perm, Russia. Advanced Methods of Organic Synthesis: Poster Presentation P-39 THE THREE-COMPONENT REACTION OF ISONITRILES WITH THIOPENOLS AND GEM-.
A. Mironov
Kalinin N.V. Nastapova
- Yanilkin I.A. Litvinov
M. Rybakov 1
A. Knyazeva
This implies that by careful choice of reaction temperature it is possible to selectively react one or both aziridine rings with C60, leading to C60 dyads and triads, respectively. Enikolopov Institute for Synthetic Polymer Materials of RAS, Laboratory for the Synthesis of Organoelement Polymers, Moscow, Russia.
M. Surin 2
- Kurdyukova 1 N.A. Derevyanko 1
The fluorene acidity and the methylene group activity in the cyanine condensation increase with such a substitution. Advanced Methods of Organic Synthesis: Poster Presentation P-49 NITRONORBORNE DERIVATIVES IN THE REACTION OF 1,3-DIPOLAR CYCLOADDITION.
Kuzhaeva 1
A variety of thioethers of the 2(5H)-furanone series 2-10 obtained from mucochloric acid 1 were selectively oxidized to their corresponding sulfones 11-19 or sulfoxides 20-25 in good yields. The reaction of 3,4-dithio-substituted derivatives 26 and 27 with m-chloroperbenzoic acid provides a method to selectively functionalize the sulfur atom bound to carbon atom C3 of the lactone ring, leading to new monosulfoxides 28 and 29 .
A. Romanova
The structure of the synthesized compounds was confirmed by IR-, 1H and 13C NMR-spectroscopy, and the purity and composition by TLC and elemental analysis. The structure of the obtained compound (IV,V) was confirmed by IR-, NMR- and X-ray structural analysis.
I. Guseynov 2
The resonance signals of the protons of the vinyl group in stereoisomers have been shown to differ. It is found that the protons of HA, HB and HC in trans isomers are shifted to a weaker field compared to the protons of cis isomers.
V. Dubrovskiy
- M. Timoshenko
Investigating the chemical properties of the pyrimidinones 2, we revealed the unprecedented reaction with hexamethylenetetramine (HMTA). Bäcklund (MIRB) addition reaction with the formation of an olefinic compound and upon interaction with the “soft” bases – nucleophiles – sodium enolates prepared from dimethyl malonate, malononitrile and methyl acetoacetate, give products of the Michael-induced cyclization reaction – tetrahydrothiophene-S, S-dioxide derivatives.
W 2 NaH, THF
- Meshkovaya
- K. Latypov
- Shamilov I.D. Nizamov
- S. Pankova
- Plemenkov 1 Ya.V. Veremeychik 1
- Plemenkov 1 O.A. Lodochnikova 2
- Kuznetsov
- Fershtat V.V. Kuznetsov
- Prishchenko M.V. Livantsov
- Salamci 1 K. Ecer 1
- I. Tokareva
Advanced Methods of Organic Synthesis: Poster Presentation P-76 SITE, STRUCTURE AND CHEMICAL PROPERTIES OF N-SUBSTITUTED 2(3)-IMINO-2,3-. Advanced Methods of Organic Synthesis: Poster Presentation P-86 NEW THREE-COMPONENT REACTION OF ISOCYANIDES WITH THIOCARBAMATES AND.

OF FLUORINATED BENZAZINES
- V. Votintseva
- Galkina 1 O.N. Kataeva 1
- K. Voronina 2
- A. Starikova 1
Arbusov Institute of Organic and Physical Chemistry Kazan Scientific Center of the Russian Academy of Sciences, Department of Organoelemental Chemistry, Kazan, Russia. Postovsky Institute of Organic Synthesis (Ural Division of the Russian Academy of Sciences), Yekaterinburg, Russia.