SCIENTIFIC COMMUNICATION
Temporal distribution of five bat species (Chiroptera,
Phyllostomidae) from Panga Reserve, south-eastern Brazil
Wagner A. Pedro 1 Valdir A. Taddei 2
ABSTRACT. Temporal distribution of tive bat species from Panga Reserve, south·eastcrn Brazil (Chiroptera, Phyllostomidae). Dala aboul activity patterns, hourly and monlhly, 0 11 five phyllostomid bals, Glossopl/aga soricina (Palias, 1766),
Anoura caudirer (E. Geoffroy, 181 8), Ca rollia perspicillara (Linnaeus, 1758), Slltrn; -ra lilium (E. Geoffroy, 1810) and Platyrrhinus lineaws (E. Geoffroyi, 18 t O), studied
over a one year period aI lhe Panga Ecological Reserve, Uberlandia, Minas Gerais state, south-eastern Brazil, are reported al1d discussed.
KEY WORDS . Chiroptera, Phyllostomidae, lemporal activity, ecological niche, mi-gration
Some studies in ecology link differences in temporal activity patterns with niche shifts between species that are similar in ecology and morphometrics (ZARET &
RAND 1971; KUNZ 1973; PIANKA 1973 ; MARSHALL & MCWILLlAM 1982).Other studies point out relationships between temporal activity, houri y and monthly, with food availability in phyllostomid bats (SAZIMA & SAZIMA 1978; SAZIMA et aI. 1982 ; LEMKE 1984; MARINHO-FILHO & SAZIMA 1989; FISCHER 1992; FISCHER et aI. 1992). Herein, data about activity patterns, hourly and monthly, on five phyllostomid bats - Glossophaga soricina (Palias, 1766), Anoura caudifer (E. Geoffroy, 1818),
Carollia perspicillata (Linnaeus, 1758), Sturnira lilium (E. Geoffroy, 1810) and
Platyrrhinus lin.eatus (E. Geoffroy, 1810) - studied over a one year period at the Panga
Ecological Reserve, a 404 ha area South oI' Uberiândia, Minas Gerais State (ca. 19°09' _19° 11 ' S; 48"23' -48°24'W), ca. 800 m altitude, are reported and discussed. The regional c1imate is seasonal with a five-month dry-cool season (from May to Septem-ber) and a seven-month rainy hot season. The vegetation consists 01'50% celTado native scrub woodland interspersed with mesofitic forest, xeromorphic forest and grasslands. Bats were sampled month1y with mist nets opened at sunset and c10sed at sunrise. Nets were checked for bats every 15 minutes. The sunset and sunrise times corresponded to data obtained from the Observatório Nacional do Rio de Janeiro, Brazil ( 1990, 1991). A total of 46 ni ghts of collecting were made between June 1990
and May 199 1 (ca. 552 h of netting). More detailed information about the collecting methods and materiais are available in PEDRO & TADDEI (1997).
1) Laboratório de Chiroptera , Departamento de Apoio, Produção e Saúde Animal , Universidade Estadual Paulista. Caixa Postal 341 , 16050-680 Araçatuba , São Paulo, Brasil. 2) Universidade para o Desenvolvimento do Estado e da Região do Pantanal. 79003-010
Campo Grande, Mato Grosso do Sul, Brasil.
952 Pedro & Taddei
Slalistical analys is fo ll owed ZAR (1984), including Kolmogorov-Sm irnov test, to compare lhe di stributio n of hourly activity for di ffe rent species.
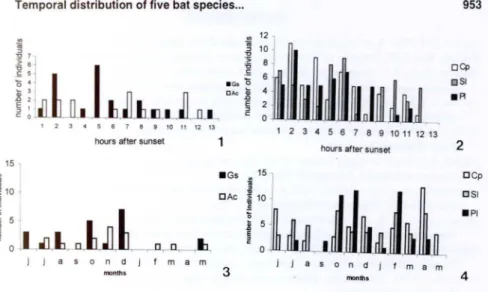
The frequency of capture for five Phyll ostomidae species during the ni ght are shown in figure I and 2. Except for the 3rd and 12th ho ur after sunset for G.
soricilla, ali five spec ies were caught throughout lhe ni ght with some di fferences of
intensity. In order of decreasing freque ncy,
C.
perspicillata was captured the mosl freq uent fo ll owed byS.
liliul17, P. lineatus, G. soricilla and with A. caudifer lhe least captured. Ali five species had their highest rate of capture、オイゥョ
セ@
lhe 1st a nd 2nd hours afler sunsel wi lh a second freque ncy peak during the 5th or 6 h hou r, and then a s li ght final increase in acti vity durin g lhe Iih
hour jusl before sunri se. In gene ral, ali five spec ies ex hibiled a noticeable reduclion in aClivily during the last lhird o f lhe evenin g from lhe 8th hour to sunri se. In comparison of the two neclari vorous species (Fig. I ), Glossophaga so ricina showed two aClivily pea ks durin8
lhe I SI hal f of the ni ght, and A. caudifer showed two acti vity pea ks during the 2n half o f th e ni ght. Glossophaga soricilla was lhe only species not lo exhibit a sli ght increase in capture during lhe II th hour after sun set (Fig. 1). The lhree frug ivorous spec ies (Fig.2), Ca rollia perspicillata, S. liliuln, and P. lineatus showed some similarity in hourl y
aClivity, but the prim ary difference was the absence of a second activ ity peak durin g lhe 6th hour afte r sun set for
C.
perspicillata.HEITH AUS et
ai.
( 1975) we re the first to suggest lhal temporal diffcrence in foraging mi g hl red uce competiti o n between so me frugivorous bats, but o nl y in lhe case of competiti o n by direcl interference durin g fo ragi ng because lh e fruils eate n in the beg inning of lhe night are not repl aced later in the ni g hl. T hu s, frugivorous bals feeding in the beginning of the ni g ht mi g hl have a compeli tive advanl age. A lth ough o ur data show sli ght diffe re nces in aCli vity pattern s for the frugivorous bats, lh ere were no obvi ous shifts in freque ncy peaks to SUppOrl lhe idea lh at there was direct competiti o n for the same food re o urces. The observed differences in hourly act ivi ly of frugivorousC.
perspicillata ,S.
liliurIJ and P.lil/ eatus may be re laled to specific aspects of th e bio logy of these spec ies, o r
reflect lhe differenccs of diel as discussed in PEDRO & TADDEI ( 1997): Carollia
perspicillaTa feeds mainly o n Piper, S. liliuln o n Solanum , a nd P. lin eat us o n
Cecrop ia . at lhe Panga Eco log ica l Reserve.
It was also suggested by HEITHAUS et
ai.
( 197 5), lhat te mpo ral di ffe re nces in foragin g may be important lo the neclarivorous bals lh at ex pl oit resources lh at are more readil y renewabl e. MARI NHO-FILHO & SAZIMA (1989) extended thi s ass umpli on lO the in sectivorous bats and a few studies support lhe hYPolhes is of te mporal segregati o n in insectivorous and nectarivorous bat spec ies (BROWN 1968; K UNZ 1973 ; M ARSH ALL & M CWILLlAM 1982). The resulls presented here, allh ough not slati sli call y s ignifi cant, show some differe nces in lhe ho url y activity pattern s in the nectarivo rous pair-wi se gro up of G. soricina-A . ca udifer. However, the re was no ev idence from o ur data lo suggest this because the re were no obvi ous diffe rences in acti vity paltern s between the two spec ies of neclari vorous bats.In general lhe nectarivorous pair-wi se spec ies had the ir hi ghest rate of capture in lhe beginning of lhe rainy season fro m October to December wi lh neg li gible captures in lhe las! half of the rainy season (Fig. 3). The fr ugivoro us
Temporal distribution of five bat species ... 953
1 2 3 '" 5 6 7 8 SI 10 11 12 13
hours after sunset
15 1
.Gs 15
"
1 2 3 4 5 6 7 6 9 1011 12 13
hours after sunset 2
1: :
I
DAc
!
1:
J
セ@
o
II
.tl
I,
01,..,
セ オLN L@
..,Il'r-,-JJOL,-, u,.D -,-01Ju.,セ@
o .;u>,.o.La,WI..,-J.llI,LJ.a.,UJl.,la,nhj j a s o n d j f m a m
months 3
j j a s o n d
mon ...
j f m a m
4
Figs 1-4. Hourly activity 01 Glassophaga soricina and Anoura caudifer (1) and Carollia perspicillata, Sturnira lilium and Platyrhinus lineatus (2); and Irequency of captures on mist nets of Glossophaga soricina and Anoura caudifer(3) and Carollia perspicillata, Sturnira lilium
and Platyrhinus lineatus (4) in the Panga Reserve.
species also exhibited their highest rates of capture in the I Si half ofthe rainy season
with a dramatic decrease in January before a return to hi gher frequencies for the last three months ofthe rainy season (Fig. 4). They also have a modest increase in acti vity near the middle of the dry season around June. There was no signifi cant difference for the nectarivorous pair-wise species Clossophaga soricina-Anoura caudifer lhroughout the year (Kolmogorov-Smirnov test, D = 0, 154; P > 0,05), or for lhe frugivorous pair-wise species Carollia perspicillata-Stumira liliul11 (i n the dry season Ko lmogorov-Smirnov test, D = 0, 154; P > 0,05; in the rainy season Kolmogorov-Smirnov test, D = 0,077; p > 0,05; and throughout the year Kolmogo-rov-Smirnov test, D = 0,077; p > 0,05).
The monthly activity patterns of these five Phyllostomidae species showed an increase in the rainy months when there was more food availability which bring out the opportunistic character of the forag ing behavior of these bats . Monthly temporal segregation was not supported by the present data (Figs 3 and 4). The frequency of caplure of frugivorous bats was, in general, hi ghest during the rainy season (Fig. 4) suggesting the possibility of mi gration fo r at least portions of these populations in the study area, and their associations to the avai labi lity of food resources, notably Cecropia pachystachya Trec. fnlits for P. lineatus .
ACKNOWLEDGEMENTS . We are graleful to Burton K. Lim for com ments and suggeslions on the manuscript. two anonymous reviewers, Paulo De Marco Jr. for help \Vith statistics, 10 Carlos A.K. Komeno for help in lhe field. and 10 CN Pq, CAPES and FAPESP who provided support through grants. and FUNDUNESP \Vho provided fin anci ai support for research mate-riais.
954 Pedro & Taddei
REFERENCES
BROWN, J.H. 1968. Activity patterns of some neotropi ca l bals. Jour. Mammal. 49 : 754-757 . FISC HER, E.A. 1992. Foraging of Nectarivorous Bats on 8 auhinia ungulata. B iotropica 24: 579-582. FlscHER, E.A. ; F.A. JI MENEZ; M. SAZIM A. 1992. Po lini zação por morcegos e m duas espécies de
Bombacaceae na Estação Eco lógica de Juréia, São Paul o. Rev. bras. BoI. 15 : 67-72.
HEITHAUS. E.R. ; T.H . FLEMI NG; P.A. OPLER. 1975. Foraging patterns and resource uli lization in seven species of bats in a seasonal tropi cal fores!. Ecology 56: 841-854
KUNZ, T.H. 1973. Resource utilization : tempora l and spatial co mpone nts ofbat aClivity in Central lowa. Jour. Mantmal. 54 : 14-32.
LEM KE, T.O. 1984. Foraging eco logy of the long-nosed bat, Glossophaga sorióna , with respecI 10
resource availability. Ecology 65 : 538-548.
MAR INHO-FI LHO, J.S. & I. SAZIMA. 1989. Activity patterns ofsix phyll ostomid bat species in Southeastem Brazil. Revista Brasileira de Biologia 49: 777-782.
MARSHALL, A. G. & A.N . MCWILLlAM. 1982. Eco logical observations on epomophorine fruit bats (Megachiroptera) in West African savanna woodland. Jour. Zoo1.198 : 53-67 .
OBSERVATÓR IO NACIONAL 00 RIO DE JANEIRO. 1990. Efeméridcs astronôm icas. Rio de Janeiro, Secretaria Especial da Ciência e Tecnologia, CNPQ, 574p.
199 1. Efcmérides astronômicas. Rio de Janeiro, Secretaria Espec ial da Ciê ncia e Tecnologia, CN Pq, 574p.
PEDRO, W.A . & V.A. TADDEI. 1997. Taxonomic assemblage ofbats from Panga Reserve, southeastern Brazil: abundance patterns and trophi c re lations in lhe Phyllostomidae. BoI. Mus. Biol. Mello Leitão, n. sér., Santa Teresa 6: 3-2 1.
PIANKA, E.C. 1973 . The structure of lizard communities. Ann. Rev. Ecol. Syst. 4 : 53-74.
SAZIMA. M .: M.E. FABIAN & l. SAZIMA. 1982. Polini zação de Lueh.ea spec iosa (Tili aceae) por
GIIIssllphaga soricilla (Chiroptera - Phyll ostomidae). Rev. Brasil. Biol. 42 : 505-5 13.
SAZIMA, M. & I. SAZIMA. 1978. Bat Pollination of lhe Pass ion Flower, Passiflo ra Inu cronala, in Southeastern Brazil. Biotr opica 10: 100- 109.
ZAR, J.H. 1984. Biostatistical Analysis. New Jersey, Prenli ce Halll nc., 2nd ed., 7 18p.
ZARET, T.M . & A.S. RAND. 197 1.Competition in tropi cal stream fi shes: sUppOI1 for the co mpetiti ve excl usion principie. Ecology 52: 336-342.
Received in 15.VI.2000: accepted in 04.IX.2002.