REVISTA
BRASILEIRA
DE
Entomologia
AJournalonInsectDiversityandEvolution w w w . r b e n t o m o l o g i a . c o mSystematics,
Morphology
and
Biogeography
Mating
behavior
and
description
of
immature
stages
of
Cyclocephala
melanocephala
(Fabricius,
1775)
(Coleoptera:
Scarabaeidae:
Dynastinae),
identification
key
and
remarks
on
known
immatures
of
Cyclocephalini
species
Sérgio
Roberto
Rodrigues
a,∗,
Carlos
Aparecido
Ferreira
Barbosa
a,
Juares
Fuhrmann
b,
Ricardo
Aparecido
Amaro
aaUniversidadeEstadualdeMatoGrossodoSul,Cassilândia,MS,Brazil bUniversidadedeSãoPaulo,MuseudeZoologia,SãoPaulo,SP,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received26February2018 Accepted3July2018 Availableonline12July2018 AssociateEditor:MarcelaMonné
Keywords: Flyingactivity Morphology Scarabaeoidea Taxonomy Whitegrub
a
b
s
t
r
a
c
t
MatingbehavioranddescriptionofimmaturestagesofCyclocephalamelanocephala(Fabricius,1775) (Coleoptera:Scarabaeidae:Dynastinae),identificationkeyandremarksonknownimmaturesof Cyclo-cephalinispecies.SomespeciesofCyclocephalaDejean,1821areregardedasrhizophagouscroppestsand othersasbeneficialspecies.Theobjectiveofthisworkwastoreportthematingbehaviorandtodescribe theimmaturestagesofC.melanocephala.ThestudiesweredevelopedattheUniversidadeEstadualde MatoGrossodoSulinCassilândia,MatoGrossodoSulstate,Brazil.Adultswerecollectedwithalight trapfromSeptembertoDecember2014and2015tocarryoutstudiesofmatingbehavior,breeding,and descriptionsofimmaturestages.Copulationlasted10.4±4.3minandtookplacefrom19:00to24:00h.
Somefemalesrefusedmalesformatingandmovedawayfromthem.Regardingflightperiod,adultswere collectedinlargerquantitiesfrom20:00to23:00h.Identificationkeystoimmaturesofthreegeneraof Cyclocephalini,includingseveralCyclocephalaspeciesarepresented.
PublishedbyElsevierEditoraLtda.onbehalfofSociedadeBrasileiradeEntomologia.Thisisanopen accessarticleundertheCCBY-NC-NDlicense(http://creativecommons.org/licenses/by-nc-nd/4.0/).
IntheCyclocephalinitribe(Coleoptera,Scarabaeidae, Dynasti-nae)about500describedspeciesareknownandofthese,morethan 85%arerepresentedbyCyclocephalaDejean,1821(Ratcliffeetal., 2013).CyclocephalaoccurfromsoutheasternCanadatoArgentina andtheAntilles(Ratcliffe,2003).InBrazil83speciesarerecorded
(Morón,2004).
Adultsfeedonplantsandflowers,thuscontributingto pollina-tion.Someexamplesofthebenefitstoplantsaredescribedtosome Araceae.Maiaetal.(2013)inastudiesconductedinAtlanticForest ofPernambucoState,foundthatflowersofTaccarumuleiEngl.& K.Krause(Araceae)arepollinatedexclusivelybyC.cearaeHöhne, 1923andC.celataDechambre,1980.InflowersofCaladiumbicolor
(Aiton)Vent.(Araceae)observedinAtlanticRainforestof Pernam-buco,Maiaand Schlindwein(2006)foundadultsofC.celata,an
importantpollinator,feedingandmating.Cyclocephalapollinators arealsoimportanttosomeAnnonaceae.InCerradoofGoiásState,
Cavalcanteetal.(2009)foundthatC.atricapillaMannerheim,1829,
∗ Correspondingauthor.
E-mail:sergio@uems.br(S.R.Rodrigues).
C.latericiaHöhne,1923andC.octopunctataBurmeister,1847are
floralvisitorsandpollinatorsofAnnonacrassifloraMart.Moreover,
Costaetal.(2017)conductedstudiesintheCerradoofMatoGrosso
StateandfoundthatC.atricapillaisthemainpollinatorofA.coriacea
Mart.,whileC.octopunctata,C.ohausianaHöhne,1923andC.undata
(Olivier,1789)aresecondarypollinators.InadditiontotheAnnona
L.speciescitedabove,Gottsberger(1989)notedC.atricapillaalso asapollinatorofA.dioicaA.St.-Hil.andA.monticolaMart.,and
C.quatuordecimpunctataMannerheim,1829asapollinator ofA.
cornifoliaA.St.-Hil.andA.tomentosaR.E.Fr.
SpeciesofCyclocephalaarealsofoundinotherplantfamilies, liketheexampleofDieringeretal.(1998,1999)whofoundadults ofC.caelestisDelgado&Ratcliffe,1990feedingandpollinating flow-ersofMagnoliatamaulipanaVázquez(Magnoliaceae)inMexico.In
MatoGrossodoSulState,Brazil,adultsofC.forsteriEndrödi,1963 werefoundfeedingoninflorescencesofAcrocomiaaculeata(Jacq.) Lodd.exMart.(Arecaceae)(OliveiraandÁvila,2011).Inarecent study,MooreandJameson(2013)related80speciesofCyclocephala
associatedwithflowersofvariousspecies.
TheimmaturestagesofCyclocephalaspeciesremaininthesoil andsomespeciesfeedonorganicmatter,asC.flavipennisArrow,
https://doi.org/10.1016/j.rbe.2018.07.001
Figs.1–4.Cyclocephalamelanocephala,adult;1,2,habitus(male,female);3,4,protibiaandtarsus(male,female).Scale:1,2=5mm;3,4=1mm.
1914observedinRioGrandedoSulStatebySalvadoriandPereira
(2006)andC.paraguayensisArrow,1913observedinthe
Pernam-bucobyAlbuquerqueetal.(2014).
However,in some species, larvae canfeed onroots of crop plantsandcausedamage.LarvaeofC.lunulataBurmeister,1847and
C.fulgurataBurmeister,1847appearassociatedwithonioncrops (AlliumfistulosumL.,Amaryllidaceae)andgrasses(Pennisetum clan-destinumHochst.&Chiov.,Poaceae),observationsmadebyVillegas
etal.(2006)inColombia.Whenlarvae,CyclocephalaparallelaCasey,
1915isconsideredanimportantpestofsugarcane(Saccharum offic-inarumL.,Poaceae)inFlorida,UnitedStates(GordonandAnderson,
1981;Cherry,1985),aswellasC.lunulatainMéxico(Aragón-Garcia
andMorón,2000).Also,C.forsteriandC.verticalisBurmeister,1847
werereporteddamagingsugarcanecropsinMatoGrossodoSul
(Coutinho et al.,2011).Cherman etal. (2014)reportedC.
mod-estaBurmeister,1847,C.putridaBurmeister,1847andC.tucumana
Brèthes,1904associatedtotherootsystemofseveralwintercrops inRioGrandedoSul.SantosandÁvila(2007)reportedthe occur-renceoflarvaeofC.forsterifeedingonrootsofsoybean(Glycine max(L.)Merr,Fabaceae)inMatoGrossodoSul.Dependingonthe circumstances,evenspeciesthatarenotusuallyconsidered eco-nomicallyimportantcandamagesomecultures,asC.flavipennis
Arrow,1914(SalvadoriandPereira,2006;Duchinietal.,2017).
Cyclocephalamelanocephala(Fabricius,1775) occurs through-out most of the New World, as United States (Ratcliffe, 1992;
Bauernfeind,2001),Mexico(Ratcliffe,1992)andBrazil(Camargo
andAmabile,2001;Nogueiraetal.,2013;Tairaetal.,2014).Adults
aretypicallyfoundfeedingoninflorescenceofsunflowerplants
(HelianthusannuusL.,Asteraceae)(CamargoandAmabile,2001).
Tairaetal.(2014)foundinMatoGrossodoSul,adultsofthisspecies
causingdamagetoshootsofyoungplantsofrubbertrees(Hevea brasiliensis(Willd.exA.Juss.)Müll.Arg.,Euphorbiaceae). Regard-ingthebiologyofC.melanocephala,Nogueiraetal.(2013)observed thattheegg-adultcyclewascompletedin113daysonaverageand morethanonegenerationispossiblyformedperyear.
DespitethespeciesrichnessofCyclocephala,immature descrip-tionsarescarce(Morónetal.,2014).Thus,studieswereconducted toverifythematingbehavioranddescriptionsofimmatureofC.
melanocephala.
Materialandmethods
Matingbehaviorofadults
Studiesonmatingbehaviorwereconductedonthe experimen-talfarmofUniversidadeEstadualdoMatoGrossodoSul(UEMS)in Cassilândia,MatoGrossodoSulState(MS).Tocollectadults,alight trap(model“LuizdeQueiroz”,SilveiraNeto andSilveira, 1969) wasinstalleddailyalongsidethepasturearea(Urochloadecumbens
Stapfcv.Basilisk,Poaceae)fromOctobertoDecember2014. Adultscollectedweretakentotheentomologylaboratoryof UEMS,separated bysex (firstpairoflegsin malesare dilated)
(Figs.1–4)andseparatedindividuallyin1000mLplasticcontainers
Afterthat,twenty-threecouplesweresortedfromtheadultsthat emergedfromthesoilandflewoff.Eachmaleandfemalewas gath-eredincouplesandputin500mLcontainersformatingbehavior observations.Theobservationroomwaskeptdarkaccordingtothe methodologyfromFacundoetal.(1999).Tovisualizeandrecord thebehaviorofmalesandfemales,aSony®
camcordermodel DCR-SX21STDwasused.
Tostudyadultflighthoursinthefield,alighttrapwasinstalled from18:00huntil06:00hof thenextday,fromOctober 30to November2,2014.At60-minintervals,thetrapwasinspected, andinsectscapturedwerecollected.Theflightscheduledatawere transformedinto√x+1andsubmittedtotheanalysisofvariance
(ANOVA).Themeansweregroupedandcomparedbythe Scott-Knotttest(p<0.05)usingSISVARsoftware(Ferreira,2011).Dataon averagetemperature(◦C),precipitation(mm)andsolarradiation
(kJ/m2)inCassilândia,wereobtainedfromtheInstitutoNational
deMeteorologia(INMET).
Descriptionofimmatures
The described larvae were obtained from adults reared in thelaboratory.FromSeptembertoDecember 2015,adults ofC.
melanocephala were collected at the experimental farm of the UEMS,withlighttrap.Theadultswerecarriedtotheentomology laboratoryandformedcouples,whichremainedin1000mLplastic containerscontainingsoilandBrachiariadecumbensStapf(Poaceae) seedlings,andthecontainerswerecoveredwithvoilfabric.
Thevesselswereinspectedeverydaytofindeggsandtoremove the dead insects. The eggswere kept in Petri dishes, contain-ing sieved and moistened soil, and keptin an air-conditioned roominthelaboratory(26±2◦Candscotophase).ThePetridishes
wereobservedatintervalsof twodays, andthehatchedlarvae weretransferredandindividualizedin500mLplasticcontainers containingsoilandB.decumbensseedlings(26±2◦Cand12h
pho-tophase)(Rodriguesetal.,2014).
FromMaytoAugust2016,thirdinstarlarvaeandpupaewere killedinboilingwaterandpreservedin70%alcohol.The observa-tionsanddrawingsofthemorphologicalaspectsofthelarvaand pupawerecarriedoutinStereomicroscopeMotic,ZeissStemiSV 6stereomicroscopeorZeissAxioscopmicroscope,bothwithlight cameracoupled.Thedetachedstructuresofthelarvalbody(e.g. mouthpartsandlegs)wereslidemountedinHoyer’s(Johnsonand
Triplehorn,2005).TheadultsofC.melanocephalaweredeposited
in the UEMS entomology collection in Cassilândia; the imma-turewasdepositedinthecollectionoftheMuseudeZoologiada UniversidadedeSãoPaulo,SãoPaulo(MZSP).AdobePhotoshop CS6softwarewasusedforimageprocessinganddrawingofthe plates.
The terminology used follows Böving (1936) and Lawrence
(1991)withsomemodificationsbySousaetal.(2018).Theterms
helus(toothor fixedand rigid cuticular process)and phoba(a groupofflexiblefixedcuticularprocesses)wereusedforboth epi-andhypopharynx.Theepipharynxareasubdivisions(corypha, hap-tomerus,paria,pediumandhaptolachus)areinitalictomakeit easiertofind.HeadchaetotaxyfollowsRitcher(1966)andSawada
(1991)assummarizedbySousa etal.(2018).Mandiblesincisor
usuallyhave3definedteeth(S1,S2,S3;distaltoproximal),and S2–S3separationisnotedbytheincisornotch(Figs.17,22).Even whenS2isreduced,thenotchiseasytofindanddefinestheS2and S3area.Besidesthesethreeteeth,aproximalmostincisortooth mayoccur(S4)betweenS3andmolar(onmandibleinnerconcave margin).
Hair-likesetaearetermedas(modificationofˇSípeketal.,2008): minute,when itslengthisat mostthreetimeslongerthanthe diameterofassociatedpuncture(barelydistinctunder magnifica-tionlessthan40×);shortorlongwhenitslengthisatleastfour
timeslongerthanthediameterofassociatedpuncture(easily vis-ibleundermagnificationof20×).Longandshortsetaeareonly
differentiated when conspicuous relative differencesoccur (e.g. rastersetae,Figs.43, 44),otherwise (i.e.wide setallength vari-ation)theoppositionminute/longformerlydefined isused(e.g. cranialsetae,Fig.7).Thelarvaesizecouldaffectthevisibilityof minuteandlongsetae,butdifferencesinrelativesizeandspatial distributionhelptoseparatebothsetaegroup.
Headchaetotaxywaswidelyusedasdiagnosisinscarab imma-tureworks,whilethoracicandabdominalchaetotaxy(exceptfrom raster)werenotorpartiallydescribed(e.g.JamesonandMorón, 2001describedthedorsallobessetation).Toexplorethebodysetae asanidentificationtool,thethoracicandabdominalsetationwere describedandillustrated(Fig.14).Thisuncommonapproachadds newdata andencourage future works toinvestigatetheentire bodychaetotaxy.Chaetotaxyweregiventoeachbodylobe.Terms likescutum,scutellum,presternum,sternum,andderivedterms areavoidedbecausethehomologybetweenlarvallobesandadult sclerites are doubtful,and the ventralbody surface hassternal andpleuralelementsinColeoptera(Kobayashietal.,2013).The resulting lobes terminology is as follows (Fig. 14): thorax dor-sum:anterior,medial,andposteriortergallobes;thoraxlateral: anteriorandposteriorpleurallobes;thoraxventer:anteromedial andposterior ventrallobes; abdomendorsum:anterior,medial, lateralandposteriortergallobes;abdomenlateral:anteriorand posteriortergallobes,andspiraclelobe;abdomenventer:anterior, medial,andposteriorlobes.Whenthislobedivisionisindistinct (e.g.Scarabaeoideapronotumhave1–3lobesandabdomen seg-mentIX usuallyhasanundivideddorsumand asimple pleural lobe,Ritcher,1966),ageneralpositionnameisgiventotheregion ifnecessary.Thechaetotaxytoprothoraciclateralscleriteisalso givenandthelobesnamesarenotappliedtoabdominalsegment X,becauseitsalreadyhasaparticularterminology(rasterandanal lobes).
The proposed identification keys and comparative table
(Table1)includedherein use newdata and information
avail-able in thebibliography (Albuquerque et al., 2014; Branet al.,
2006;Garcíaetal.,2009;Johnson,1941;King,1984;Morelli,1991;
Morelli andAlzugaray, 1994;Morón etal., 2014;Neita-Moreno
andYepes,2011;PereiraandSalvadori,2006;Remedi-de-Gavotto,
1964;Ritcher,1966;Souzaetal.,2014a,b;Vincinietal.,2000).
Results
Cyclocephalamelanocephala(Fabricius,1775)
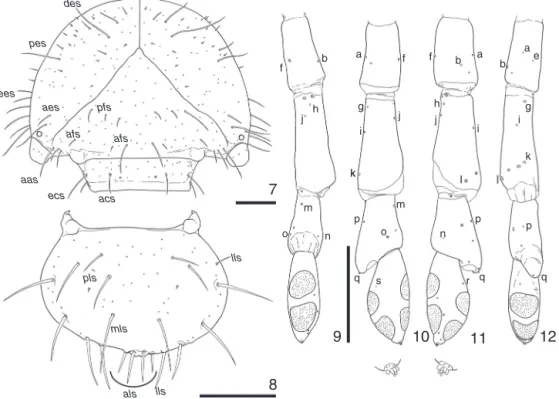
Thirdinstarlarva(Figs.5–44).Body(Fig.5)length:19–26mm; grayishoryellowish white,headand respiratoryplates yellow-ish brown; surface densely setose, setae yellowish brown to brown. Head (Figs. 6, 7, 13) width: 2.8–3mm; epicranial and epistomalsuturesdistinct;stemmataverysmall;antennifer some-what cylindrical and with 3 punctures; cranium, clypeus and labrum(Figs.7,8)withmanyhomogeneouslydistributed punc-tures, except in labralanterior area. Each half of cranium and clypeuswith(Fig.7):4–3longand2–3mindorsoepicranialsetae (des)ina row and1minseta internallypositioned,2 longand numerousminuteposteroepicranialsetae(pes),2–3long anteroep-icranialsetae(aes),5–8longexternoepicranialsetae(ees),2–3long and 1min posterofrontal setae(pfs), 1 long externofrontalseta (efs),2–3longanterofrontalanglesetae(aas),2longanterofrontal setae(afs),2longexternoclypealsetae(ecs),1longanteroclypeal seta (acs). Labrum(Fig. 8): each half with 2–3long and 1min
Table1
ChaetotaxyoftheknownthirdinstarsofCyclocephala.
Species Parietals Frons Clypeus Labrum Raster
des pes aes ees pfs efs aas afs acs ecs pls lls mls als tg* pr* pa al*
C.barrerai 2–3 3 ∼3 ∼4–5 2 0 1 1 0 1 3–4 ∼2 1 – 21–23 u 0 30–32
C.borealis 2–3 – – – 2 1 1–2 1–2 – – – – – – ∼25 u 0 20
C.celata 4 – – – 2 1 2 3 1 2 4–5 4 1 – 14–16 u 0 –
C.comata 2 2 2–3 – 2–3 0 – 0 1 1 2–4? – 1 – 10–12 u 0 48–50
C.disticta 3–4 5–6 – – 4–5 1 2 2 1 1 3–4 ∼4 1 2 7–9 u 0 ∼22
C.fasciolata 3 2 2–3 – 1–2 0 1 0 1 1 0 3 1 – 0 [8–9] u 0 26–28
C.flavipennis – – – – – – – – 1 1 – – – – 13–16 u 0 ∼22
C.fulgurata 3–4 5–6 2–3 ∼8 2 1 1 1 1 1 – ∼3 1 – 12–15 u 0 34–38
C.gregaria 1 1 2–3 ∼3 2 1 1 1 1 1 – ∼2 1 – 19–22 u 0 30–35
C.jalapensis 3 2 2–3 – 1 0 1 0 1 1 0 3 1 – 0 [16] u 0 0 [26]
C.longula(=C.abrupta) 7–8 – – – 3 1 2 2 – – – – – – – u 0 –
C.lunulata 2–4 5–61** 2–3 ∼5 2 1 1 1 1 1 7–8 ∼2–3 1 – 10–1620–28! u 0 26–34∼20!
C.lurida(=C.immaculata) 3 ∼2 ∼3 ∼8 2 1 1 1 1 1 0 ∼4 1 2 13–16 u 0 ∼35 C.melanocepala 3–4 2 2–3 5–8 2–3 1 2–3 2 1 2 2–3 4–5 1 2 8–11 1–2 3–4 28–34
C.modesta – – – – – – – – – – – – – – 11–13 2–3 6–8 ∼25
C.paraguayensis 5 ∼5 ∼1 ∼8 1 1 2 1 1 2 1 ∼2 1 ∼15 4–5 3–4 ∼37
C.pasadenae 3–5 – – – 2–4 1 1 – – – – – – – ∼15 u 0 –
C.putrida – – – – – – – – – – – – – – ∼20 u 0 ∼37
C.signaticollis 3 2 3 – 1 – 3 1 1 1 – – – – 13–16 u 0 20–30
C.sinaloae 3–4 3 ∼3 ∼4–5 2–3 1 1 1–2 1 1–2 2–3 2–3 1 – 14–17 u 0 59–62
C.testacea 4 1 5–6 ∼8 – 1 – 2 1 3 5 – 1 – 28–30 0 ∼33 –
Thechaetotaxyisgivenforonesideofthestructure,exceptforventralanallobe(al).*onlyhamatesetaequantified,ifhamatesetaeabsentthenumberofhair-likesetaeisgiven betweensquarebrackets.“u”unapplieddata(i.e.whenpalidiaisabsent,itisimpossibledefinethepreseptularsetae).“∼”about.Cyclocephalalunulata:generalchaetotaxyby
Branetal.(2006);**redescriptionofMorónetal.(2014)recordedonly1pes;!firstcharacterizationbyKing(1984).aas,anterofrontalanglesetae;acs,anteroclypealsetae; aes,anteroepicranialsetae;afs,anterofrontalsetae;al,ventralanallobesetae;als,anterolabralsetae;des,dorsoepicranialsetae;lls,laterolabralsetae;mls,mediolabral setae;ecs,externoclypealsetae;ees,externoepicranialsetae;efs,externofrontalsetae;pa,palidiumsetae(pali);pes,posteroepicranialsetae;pfs,posterofrontalsetae;pls, posterolabralsetae;pr,tegillumpreseptularsetae;tg,tegillumsetae(includingthepreseptularones).
Figs.5–6.Cyclocephalamelanocephala,thirdinstarlarva;5,lateral;6,head,dorsal.t10,abdominaltergiteX;usb,U-shapedsclerotizedbar.Scale=1mm.
width=2.1)and with5sensilla(1dorsal,1internal,1 external, 2ventral);IIlong(l/w=2.3–2.5)andwith9–10sensilla(3–4 dor-soproximal,2ventroproximal,4ventrodistal);IIIshort(l/w=1.3) andwith10 sensilla(2dorsoproximal, 2internal, 4external, 2 ventral),ventrodistalprocessbearingadorsalsensorialspotand 2distalsensilla;IVlong(l/w=2.8),with6sensilla(4external,2 ventral),2dorsaland2ventralsensorialspots,andadistal sen-sorialareabearingabout9prominentminutesensilla.Epipharynx
(Figs.15,16).Corypha:epizygumdistinctandclythraabsent.
Hap-tomerum:zygumbeak-like,2-toothedandwithabout10sensilla; heliabsent.Paria:acropariaevidentlyseparatedfromchaetoparia and each side withabout 13 setae; each side of acanthoparia with9–11anteriorsetaeand8–9minposteriorsetae;gymnoparia narrow;rightchaetoparia withabout95 setae,left chaetoparia withabout70setae;dexiotormatwicelongthanlaeotorma,left
pterotormarounded,aptormaindistinct,epitormaasanimpressed roundedsulcus;plegmatia,proplegmatiaandphobaabsent.Pedium
des pes ees aes pfs f j h g i k j h j i i k I m k n q s p p p q q r
11
10
9
12
m o n o g ff a ae
b
b a b
afs pls mls als lls lls
8
7
acs ecs aas afsFigs.7–12.Cyclocephalamelanocephala,thirdinstarlarva;7,craniumandclypeus,dorsal;8,labrum,dorsal(epipharyngealventralsetaeomitted);9–12,leftantenna(dorsal, internal,external,ventral;externalandinternalsideswithapexdetail;somesensillanumberedtoeasilythecorrespondence;sensillumhcanbeabsent).Chaetotaxy(italic) onthetext.Scale=0.5mm(antennaldetailswithmagnificationfourtimesbiggerthanantennae).
setae.Leftmolarwith9dorsoproximalsetaeinarow,3 ventro-proximalsetaeinatuft;2anteriorchisel-liketeethtransversally positionedtoeachother,adorsalandaventraltooth,2 transver-salshallowcarinaebetweenthedorsalandventralteeth;aciawith apexroundedandbearingabout6setae;calxsemicircular;brustia with12setae.Maxillae(Figs.23–25).Galeaandlaciniaseparated bysuture;galeawithanuncus;laciniawith3unci;mala with-outconspicuoussetaerow.Stipewithstridulatoryareabearing13 obtuseteethandadistaltruncateprocess.Palpwith4palpomeres: Iwithanexternoproximalsensillumandaminuteexternodistal seta;IIwithanexternodorsalsensillumand3ventralsensilla;III withanexternalseta,aventralsetaand2ventralsensilla;IVwith 3externalsensillaandaninternodistalminuteseta,distal senso-rialareabearingabout13sensilla.Hypopharynx(Figs.16,24)with asymmetricalsclerite,rightandleftlateralwithabout14setae(7 long,7min),lateromedialleftareawitharowofabout22stout setae,lateromedialrightareawithagroupofphobae;right ante-riorareawithaprominenttooth,posteriorareawithaprominent semisphericalprocess.Posteriorpreoralarea:eachsideofdorsal area(posteriortoepipharynx,Fig.15)withasensillum;eachside ofventralarea(posteriortohypopharynx,Fig.24)with2sensilla, rightareawithananteriorstoutseta,leftareawitharowofabout23 stoutsetae.Labium(Figs.16,24,25).Submentumwithaposterior scleritebearing1setaeand4–5sensillaoneachside,anteriorarea with2setaeandasensillumoneachside.Mentumwithaglabrous scleritemediallyinterrupted,anteriorareawith7–8setaeand1–4 sensillaoneachside.Prementumwithascleritebearing6–8setae distributedaroundpalpiinsertion;ligula(Figs.16,24)with3large medialsetaeoneachside,16smallposteriortooth-likesetae,a smallmedialtubercle-likeprocess,aposteriortransversalsclerite, posteriorareabearingasperites.Palpwith2palpomeres:Iwitha minuteventroproximalseta;IIwithaventrodistalsensillum,distal sensoryareawithabout13sensilla.Thorax(Figs.5,14). Protho-raxwithatergallobebearing40–50thinsetae(setaesimilarto
Fig.44d),anteriorpleurallobewith4–5thinsetae,posterior pleu-rallobewith9–17,prothoraciclateralscleritewith6–8thinsetae, anteromedialventrallobewith46–54,posteriorventrallobebare. Meso-andmetathoraxwithanteriortergallobebearing15–19thin setae,medialtergallobewith44–60thinsetae,posteriortergallobe with16–20thinsetae,anteriorpleurallobewith4–7thinsetae, posteriorpleurallobewith2–4thinsetae,anteromedialventral lobewith34–40thin setae,posterior ventrallobewith2setae.
Legs(Figs.26–28).Pro-,meso-andmetafemurinternodistalarea
withasmallandacutetuberclebearingadistalseta,meso-and metafemurwithanexternodorsalmacula;pretarsuswith2 lat-eroventralsetaeandanacuminateapex,propretarsuslongerthan meso-andmeso-longerthanthemetapretarsus(Figs.29–31). Tho-racicspiracle(Figs.32–34)with11–14perforationsindorsalradius (DR),6–8inlateralradius(LR),13–16perforationsinventralradius (VR);perforationsoblongorslightlyameboidshaped;bullaslightly largerthanthedistancebetweenrespiratoryplatearms.Abdomen
(Figs.5,14):AbdominalsegmentIwithanteriortergallobebearing
hyr
ant mda
mdc
hyt tnt mlf th1
th2
th3
ab1
esl pal
ppl
ab7
ab8 ab7
ab8
ab9
ab10
ab10
usb ab9
14
tpl tml tll val
vml vpl ab2
ab1 th3
th2 th1
tal
13
Figs.13–14. Cyclocephalamelanocephala,thirdinstarlarva;13,head,ventral(leftsidewithsubmentumandcardo);14,distendedbodytegument(hair-likesetaerepresented bytheirpuncture,abdominalsegments3–7omitted).ab1–10,abdominalsegment1–10;ant,antennifer;esl,spiraclelobe;hyr,hypostomalrod;mda,cranio-mandibular acetabulum;mdc,cranio-mandibularcondyle;mlf,maxillolabialcomplexforamen;pal,pleuralanteriorlobe;ppl,pleuralposteriorlobe;tal,tergalanteriorlobe;tll,tergal laterallobe;tpl,tergalposteriorlobe;tnt,tentorium;th1–3,pro-,meso-andmetathoracic;usb,U-shapedsclerotizedbar;val,ventralanteriorlobe;vml,ventralmediallobe; vpl,ventralposteriorlobe.Scale=0.5mm.
medialventrallobewith5–7thinsetae,posteriorventrallobebare. AbdominalsegmentVIIIsimilartoVII,buttergallobewith25–28 thinsetae.AbdominalsegmentIXwithatergallobewithan ante-riorgroupof10–15thinsetaeandaposterolateralgroupof21–24 thinsetae,apleurallobewith14–17thinsetae,anteriorventral lobebare,posteriorventrallobewith4medialthinsetaeand2–4 lateralthinsetae.SegmentX(Figs.5,14,43)withacurvedanal opening,tergitewithananteriorU-shapedsclerotizedthinbar,a groupofabout170–180anteriorthinsetaeandaposteriorgroup of46–61stoutsetae;ventralanallobewith10–15thinsetaeand 28–34hamatesetae.Spiracles(Figs.35–42):Ismallerthanother onesandwith6–8perforationsinDRandLR,and13–15inVR;II–VI
acr
aca
crp
ppa
crp
ppa
lip
15
16
Figs.15–16.Cyclocephalamelanocephala,thirdinstarlarva;15,epipharynx;16,cibarium.aca,anteriormostacanthopariaseta;acr,lateroposterioracropariaseta;crp,right partofcrepis;lip,ligulartubercle-likeprocess;ppa,posteriorpreoralarea.Scale=0.5mm.
S1 S2
inn
S3
S2
17
18
19
20
21
22
24
25
23
ppa
Figs.23–25. Cyclocephalamelanocephala,thirdinstarlarva;23,maxilla,internal;24,maxilla,hypopharynx,ligula,dorsal;25,maxilla,labium,ventral.ppa,posteriorpreoral area.Scale=0.5mm.
Remarks.LarvaeofC.melanocephalaandC.paraguayensisare easilydistinguishedfromotherknownCyclocephalinilarvaeby thepresenceofpalidiawithbifurcatesetae(Fig.44a).Thebody chaetotaxy(Table1)isusefulassupplementarydatatothespecies identification.
ForC.modestaandC.putrida,onlytherasterisknown.Morelli
(1991)providedimagesofthesespecies,rasterandattributedthe
originaldata to anunpublished thesis: “L. Alvarado. 1980. Sis-temáticay bionomía decoleópteros que enestados inmaduros vivenenel suelo.UniversidadNacional delaPlata.Facultad de CienciasNaturalesyMuseo,Argentina”.LarvaeofC.modestahavea peculiarraster(notseeninotherCyclocephalini)withpalidialong andposteriorlydivergent.Otherwise,larvaeofC.putridacannot bedistinguishedfromotherCyclocephala larvaeastherasterdo
notshowanyspecifiedpattern.Moredataaboutbothspeciesare neededtoclarifythelarvaetaxonomy.
ThirdinstarlarvaeofC.flavipennisandC.signaticollis Burmeis-ter,1847areeasilydifferentiatedfromotherCyclocephalinilarvae by ventral anal lobe ornamentation, both have posteromedial hamatesetaedistinctlybiggerthananterolateralsetae.Immature ofC.signaticolliswasdescribedbyRemedi-de-Gavotto(1964)and redescribedbyMorelli(1991),andlarvaeofC.flavipenniswerefirst timecharacterizedbyPereiraandSalvadori(2006).Untilnow,itis impossibletoseparatebothspeciesandmorestudiesareneeded tosolvethisproblem.
34
35
36
37
38
39
40
41
42
33
30
29
32
26
27
28
31
Figs.26–42.Cyclocephalamelanocephala,thirdinstarlarva;26–28,rightlegsandpleurites(anterior,medial,posterior);29–31,rightpro-,meso-andmetapretarsus,dorsal; 32–33,detailofperforationsofmesothoracicspiracle(dorsalarm,medialarea);34–42,mesothoracicspiracleandabdominalspiraclesI–VIII.Scale,Fig.26=0.3mm;Figs.29, 34=0.1mm;Fig.32=0.05mm.
a
b
d
b
c
c
d
a
43
44
(Scarabaeinae;PaulianandLumaret,1972).However,the macu-laeofB.bubalushasaminutesetaandtwoslightlyglobosedark spots(maculaesmoothinC.melanocephala).
Material examined. Brazil, Mato Grosso do Sul, Cassilândia, experimentalfarmofUEMS,20.v.2016,leg.R.A.Amaro,6larvae (MZSP–10.356).
KeytoknownthirdinstarlarvaeofknownCyclocephalini(minutesetaeareomittedinthekey)
1–AntennomereIVwithoneormorethan2dorsalsensorialspots;zygumasacross-barorbeak-likewith1ormorethan2teeth;abdominaltergitesVIII–IXwithor withoutnumeroussmallstoutsetae...DynastinaeotherthanCyclocephalini 1′–AntennomereIVwith2dorsalsensorialspots;zygumbeak-likewith0or2teeth;abdominaltergitesVIII–IXwithoutsmallstoutsetae...Cyclocephalini.......2
2(1)–Headwithoutanterofrontalsetae(afs);zygumtoothlessandwithposteriormargincrenulateorstraight;leftmandiblewithfourthscissortooth(S4 present)...3 2′–Eachheadsidewith0–2anterofrontalsetae(afs);zygum2-toothed;leftmandiblewithorwithoutS4....CyclocephalaDejean,1821(part).........................7
3(2)–Headwithoutposterofrontalsetae(pfs);perforationsofthoracicspiracleameboidandbearingmorethan4sinuosities...AncognathamancaLeConte,1866 3′–Eachheadsidewith1–2posterofrontalsetae(pfs);perforationsofthoracicspiracleoblong,ifperforationsslightlyirregularshaped,thenthemwithlessthan4
sinuosities...4 4(3)–Eachheadsidewith1anterofrontalangleseta(aas)...C.fasciolataBates,1888
4′–Eachheadsidewith2–4anterofrontalanglesetae(aas)...DyscinetusHarold,1869..................................................................................5
5(4)–Eachheadsidewith4–5dorsoepicranialsetae(des)...D.morator(Fabricius,1798) 5′–Eachheadsidewith2dorsoepicranialsetae(des).......................................................................................................................6
6(5)–Eachsidewith3–4anterofrontalangleseta(aas);haptolachusleftsidewithabout18setae...D.dubius(Olivier,1789) 6′–Eachheadsidewith2anterofrontalangleseta(aas);haptolachusleftsidewithlessthan10setae.................................. D.rugifrons(Burmeister,1847)
7(2)–Palidiapresent,sometimespaliirregularlydistributedandseptulabarelydistinct,butpalieveneasilydifferentiatedfromtegillarsetae...8 7′–Palidiaabsent............................................................................................................................................................11
8(7)–Eachpalidiumwithmorethan30setae...C.testaceaBurmeister,1847 8′–Eachpalidiumwithlessthan10setae...................................................................................................................................9
9(8)–Eachpalidiumwith6–8acutesetae...C.modestaBurmeister,1847 9′–Eachpalidiumwith3–4bifurcatesetae(Fig.44a)......................................................................................................................10
10(9)–Eachheadsidewith2–3posterofrontalsetae(pfs),2anterofrontalsetae(afs),2–3posterolabralsetae(pls)...C.melanocephala(Fabricius,1775) 10′–Eachheadsidewith1posterofrontalseta(pfs),1anterofrontalseta(afs),1posterolabralseta(pls)...................................C.paraguayensisArrow,1913
11(7)–LeftmandiblewithS4...C.jalapensisCasey,1915 11′–LeftmandiblewithoutS4...............................................................................................................................................12
12(11)–Ventralanallobewith7–9posteromedialhamatesetaetwicelargerthanotherhamatesetae(cf.Remedi-de-Gavotto,1964:Fig.8;PereiraandSalvadori, 2006:Fig.6b)... C.flavipennisArrow,1914andC.signaticollisBurmeister,1847(seeremarks) 12′–VentralanallobewithmedialsetaenotorslightlybiggerthanlateralsetaeORsetaeprogressivelylargertomedialarea,butsetaeenlargementneverabrupt...13
13(12)–AbdominalspiracleIevidentlysmallerthanII–V...14 13′–AbdominalspiracleI–Vwithsimilarsize..............................................................................................................................17
14(13)–Eachheadsidewithmorethan6dorsoepicranialsetae(des)...C.longulaLeConte,1863 14′–Eachheadsidewithlessthan5dorsoepicranialsetae(des)...........................................................................................................15
15(14)–Eachheadsidewith1dorsoepicranialseta(des)and1posteroepicranialseta(pes)...C.gregariaHeyne&Taschenberg,1908 15′–Eachheadsidewith2–4dorsoepicranialsetae(des)and5–6posteroepicranialsetae(pes).........................................................................16
16(15)–Maxillarystridulatoryareawith7teethandananteriortruncateprocess...C.lunulataBurmeister,1847 16′–Maxillarystridulatoryareawith9–10teethandananteriortruncateprocess..........................................................C.fulgurataBurmeister,1847
17(13)–Ventralanallobewithmorethan45hamatesetae...18 17′–Ventralanallobewithlessthan40hamatesetae......................................................................................................................19
18(17)–Eachheadsidewith2dorsoepicranialsetae(des),2posteroepicranial(pes)andwithoutanterofrontalsetae(afs)...C.comataBates,1888 18′–Eachheadsidewith3–4dorsoepicranialsetae(des),3posteroepicranial(pes)and1–2anterofrontalsetae(afs)...C.sinaloaeHowden&Endrödi,1966
19(17)–Eachsideofrasterwith7–9hamatesetae...C.distinctaBurmeister,1847 19′–Eachsideofrasterwithmorethan10hamatesetae...................................................................................................................20
20(19)–Eachsideofrasterwithlessthan17hamatesetae...21 20′–Eachsideofrasterwithmorethan18hamatesetae...................................................................................................................23
21(20)–Maxillarystridulatoryareawith4teethandananteriortruncateprocess...C.celataDechambre,1980 21′–Maxillarystridulatoryareawithmorethan6teethandananteriortruncateprocess...............................................................................22
22(21)–AbdominaltergiteXwithaU-shapedthinsclerotizedbar(similartoFigs.5,14),areaanteriortoscleromewithatransversalsetaegroup...C.lurida Bland,1863
22′–AbdominaltergiteXwithaU-shapedthinsclerotizedbar,areaanteriortoscleromebare................................................C.pasadenae(Casey,1915)
23(20)–Ventralanallobewithabout20hamatesetae... C.barreraiMartínez,1969 23′–Ventralanallobewithabout30hamatesetae...................................................................................................C.borealisArrow,1911
Pupa(Figs.45–51).Body(Figs. 45–47)length15.2–15.4mm;
thoraxwidth 7.1–7.5mm; whitish, integument macroscopically smoothandglabrousbutcoveredbyathinandshortmicroscopic pubescence,whichgivesavelvetyappearancetothesurface,this pubescenceslightly longer in abdomenlaterals. Head(Fig.48). Vertex hidden under pronotum from dorsal view. Epistomal suturedistinct laterally and indistinct medially. Canthussmall. Labrumshortandtransversal,positionedbetweenthemandibles. Maxillary palps prominent. Labium slightly rounded. Antenna withtwodefinedregions:scape-pedicelandfunicle-clava.Thorax. Pronotumwiderthanlong,greaterwidthattheposteriormargin, lateralmarginsrounded.Prosternumwithvisibleposteriorprocess betweenpro-andmesocoxae,processacuteinmales(Fig.49)and
roundedinfemales(Fig.50);precoxalareahiddenbytheheadin ventralview.Mesonotumas longaspronotumandlongerthan metanotum.Elytracurvedventrallyaroundthebodyandalmost smooth. Pro-, meso- and metacoxa contiguous; profemur-tibia slightlyexposed indorsalview;mesofemur-tibiasuperposedto wingsin ventralviewand hiddenin dorsalview;protibia with
45
46
47
Figs.45–47. Cyclocephalamelanocephala,femalepupa(dorsal,ventral,lateral).Scale=5mm.
48
49
50
51
posterior genital ampullae larger than long and with a distal impressedline;sterniteXslightlyexposed.
Remarks.PupaeofC.melanocephalaandC.paraguayensisare easilydistinguishedfromotherknownCyclocephalinipupae(see thekey below)by thepresence of 5 dioneiform organs, other cyclocephalinepupaehave4(C.signaticollis)or6organs.Morón
etal.(2014)characterizedpupaeofC.jalapensiswith6dioneiform
organs,moredataareneededtoseparatethisspeciesfromothers. ThelarvaeandpupaeofC.borealisArrow,1911werefirsttime describedandillustratedbyJohnson(1941),butmore morpholog-icaldataareneededtoseparateitspupaefromothers.
BesidestheCyclocephalaspecies,othertwospeciesof Dyscine-tusHarold,1869havetheirpupaedescribed.Dyscinetusrugifrons
(Burmeister,1847) pupawasdescribed byVincini etal.(2000), andD.dubiuspupa(Olivier,1789)wasdescribedbyNeita-Moreno
andYepes(2011).Apparently,pupaeofCyclocephalahavetergite
IXventralfoldhiddenmostofsterniteIXinlateralview(Fig.47), whileDyscinetuspupaehaveashorttergiteIXventralfoldandmost ofsterniteIXisexposedinlateralview(cf.Neita-MorenoandYepes, 2011:Fig.h).Moredataareneededtoconfirmthesedifferencesand toproposeanadequatediagnosis.
Material examined. Brazil, Mato Grosso do Sul, Cassilândia, experimentalfarmofUEMS,31.viii.2016,leg.R.A.Amaro,4females, 1male(MZSP–10.356).
KeytoknownpupaeofCyclocephala
1–Abdomenwith4dioneiformorgansbetweenIII–IV,IV–V,V–VI,VI–VII...C.signaticollisBurmeister,1847 1′–Abdomenwithmorethan4dioneiformorgans,organspresentbetweenI–IIandII–III................................................................................2
2(1)–Abdomenwith5dioneiformorgansbetweenI–II,II–III,III–IV,IV–V,V–VI...3 2′–Abdomenwith6dioneiformorgansbetweenI–II,II–III,III–IV,IV–V,V–VI,VI–VII......................................................................................4
3(2)–AbdominaltergiteVIIanteriormarginabout2.5timeswiderthantheposteriormostdioneiformorgan(betweenVI–VII)...C.melanocephala (Fabricius,1775)
3′–AbdominaltergiteVIIanteriormarginabout1.75timeswiderthantheposteriormostdioneiformorgan(betweenVI–VII)...C.paraguayensisArrow,1913
4(2)–AbdominaltergiteXIventralfoldformingaposteriortubercle-likeprocessbearingsomerelativelylongsetae(cf.Souzaetal.,2014a:
Figs.16–18)...C.testaceaBurmeister,1847 4′–AbdominaltergiteXIventralfoldposteriorlyobtuseoracute,butneverwithatubercle-likeprominence............................................................5
5(4)–AbdominaltergiteXIventralfoldposteriorlyacute...C.fulgurataBurmeister,1847 5′–AbdominaltergiteXIventralfoldposteriorlyobtuse.....................................................................................................................6
6(5)–Profemur-tibiaarticulationareahiddenindorsalview...C.testaceaBurmeister,1847 6′–Profemur-tibiaarticulationareavisibleindorsalviewasasmallareanexttopronotumposteriorangleandelytralhumeralarea(similartoFig.45)...7
7(6)–PubescenceofabdominaltergiteXIventralfoldalmosthiddenindorsalview,maleanteriorgenitalampullaslightlyconstrictedmedially...C.gregaria Heyne&Taschenberg,1908
7′–PubescenceofabdominaltergiteXIventralfoldevidentindorsalview,maleanteriorgenitalampullastronglyconstrictedmedially...C.lunulata
Burmeister,1847
Matingbehaviorofadults
Severalstagesofmatingbehaviorwereobservedforthe23 cou-plesmatedinthelaboratory.Adultsremainedinthesoilduring theday.Atnightfall,males(n=18)andfemales(n=13)exposeda portionoftheclypeusnearthesoilsurfaceandmovedtheantennal lamellaeindifferentdirectionsfor5.2±2.1min(range3–11)before
theyleftthesoilandstartedtheflight.However,somemales(n=5) andfemales(n=10)arrivednearthesoilsurface,leftquickly,and begantofly(Fig.52).
Whentheyleft the soil, adults flewactively onaverage for 8±3.5min(range4–14)andthenbegantowalkonthesoilwith
theantennaeerectedandthelamellaeopen.Fourteenoutofthe couplesmatedshowedmultiplestagesofmatingbehavior,while ninecouplesshowednomatingbehavior.Atthefirststageof mat-ing,themaleapproachesthefemalefrombehind(n=9)touching theendoftheelytraorpygidiumofthefemalewiththeantennae andprotarsiortouchesthefemaleonherside(n=5).Whenthe maletouchesthefemale,possiblychemicalrecognitionbetween bothisoccurring;however,evidenceofapheromonereleasewas notobserved.Atthenextstage,themaleclimbedonthefemale (n=14),holdingherwithhisclawsandremainedinthatpositionfor
2.3±1min(range1–4).Sometimes,thefemalesrefusedthemales
formating(n=4),thosewalkedawayfromthemaleand began tofly.
Whenthemalewasacceptedbythefemale(n=10),itplaced itsbodytoreachthefemale’spygidium.Next,rhythmical move-mentsofthemaleabdomenwereobservedwhileitexposedthe aedeagus andbegan the copulation.Copulationlasted on aver-age10.4±4.3min(range5–16).Aftercopulation,themale(n=10)
retractedtheaedeagusin8sandremainedonthefemalean aver-ageof18.0±6min(range10–28);afterwardthemaleclimbedoff
thefemaleandtheyseparatedfromeachother.Femalescopulated inthelaboratoryonlybetween19:00and00:00h;two copula-tionsfrom19:00to20:00h,fourfrom20:00to21:00h,twofrom 21:00to22:00h,onefrom22:00to23:00h,andonefrom23:00to 24:00h.
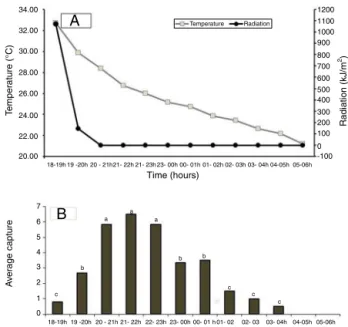
Adultswerecollectedwithalighttrapfrom18:00to04:00h thenextday(Fig.3).After18:00h,brightnessof1072kJ/m2began
todecreaseandat20:00h,brightnesswas0kJ/m2,coincidingwith
thebeginningoftheflightactivity.Adultswerecollectedingreater quantityfrom20:00to23:00h.Theaveragetemperatureduringthe flightobservationsrangedfrom32.7◦Cto21.1◦Cbetween18:00
and06:00h,respectively(Fig.53).
Discussion
ThestepsrelatedtothematingbehaviorofC.melanocephala
aredescribedforthefirsttime.Someinformationisknownabout matingbehaviorofsomeCyclocephalaspecies.Souzaetal.(2014a)
studiedthebiologicalaspectsofC.distinctaBurmeister,1847and
foundthatcopulationoccurredfrom18:00to20:00h.ForC.celata,
Souza et al. (2014b) reported that adults mate day and night.
AdultsofC.verticaliskeptinthelaboratorymatewithan aver-agedurationofabout12–19.2min(BarbosaandRodrigues,2016;
Rodriguesetal.,2010).ForC.lunulata,copulationlasted15–20min,
andthefemalesmaymatemorethanonce(Stechauner-Rohrínger
andPardo-Locarno,2010).
Eventhoughtherewasacontactwiththeantennaeandfirst pairoflegswhenmalesgetclosertofemales,noevidenceof sex-ualpheromonereleasehasbeenobservedforthem.Inadultsof
Exomalaorientalis(Waterhouse,1875)(Rutelinae),Facundoetal.
(1999)foundthatfemalesreleasesexualpheromoneandattract
Female and male n = 23
Leave the soil and fly actively
Land and form a couple
Land and do not form a couple
Male touches the front part of female
Male climbs on female
Female refuses male and detaches Female accepts male for
copulation
Male bends the abdomen and expose edeagous
Copulation starts with introduction of edeagous
Female remains still
Copulation finishes and male retracts edeagous
Male remains on female after copulation
Male and female detach from each other Male touches the back
part of female
23
14
14
10 4
5
9
9
10
10
10
10
10
10
Fig.52.EthogramofmatingbehaviorofCyclocephalamelanocephala(n=23couples)inthelaboratory.
demonstratedthatthenon-acceptanceofCanthoncyanellus cyanel-lusLeConte,1859(Scarabaeinae)femalesbysomemalesformating wasrelatedtodifferencesinsexualmaturityofbothsexes.Such behaviorwasalsoobservedinAnomalatestaceipennisBlanchard, 1851(Rutelinae)byRodriguesetal.(2014).
Aftercopulation,malesofC.melanocephalaremainedonfemales probablytopreventanothermalefromcopulatingwithher.Arakaki
etal.(2004)observedthatincopulationsofDasylepidaishigakiensis
(Niijima&Kinoshita,1927)(Melolonthinae)themaleremainedon
guardoverthefemaleaftercopulationtoavoidtheapproachof othermales.InLiogenysbidenticepsMoser,1919(Melolonthinae), malesremainedfor4honaverageonthefemaleaftercopulation, asaprotectionagainstothermales(Rodriguesetal.,2014).
Conflictsofinterest