J. Braz. Chem. Soc. vol.18 número7
Texto
Imagem
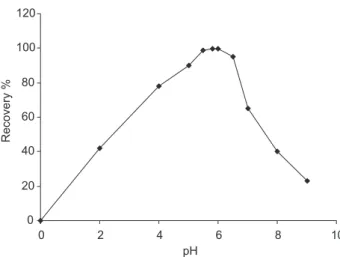
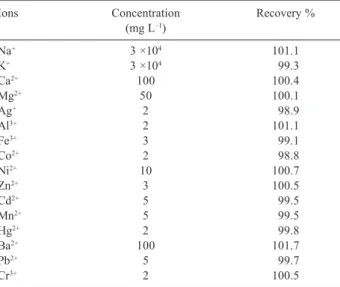
Documentos relacionados
A method for the determination of cadmium by flame atomic absorption spectrometry (FAAS) after its preconcentration onto a column containing silica gel modified with cupferron
A device for direct solid analysis by flame atomic absorption spectrometry (DSA-FAAS) with prior solid phase extraction procedure is proposed for the determination of lead in
Determination of Trace Lead in Waters by Flow Injection Hydride Generation Atomic Absorption Spectrometry..
Immobilized Stearic Acid as a New Sorbent for On-Line Preconcentration and Determination of Lead by Flow Injection Flame Atomic Absorption Spectrometry.. Shayessteh Dadfarnia,*
A cloud point extraction (CPE) procedure was developed for the separation and preconcentration of trace amounts of cobalt prior to its determination by flame atomic
Simple spectrophotometric determination of trace amounts of Ni in complex environmental samples by DMG could be possible after preconcentration and separation of
Determination of Copper, Iron, Lead and Zinc in Gasoline by Sequential Multi- Element Flame Atomic Absorption Spectrometry after Solid Phase Extraction..
In situ surfactant-based solid-phase extraction (ISS-SPE) is proposed as a preconcentration procedure for the determination of cadmium in water and food samples by flame