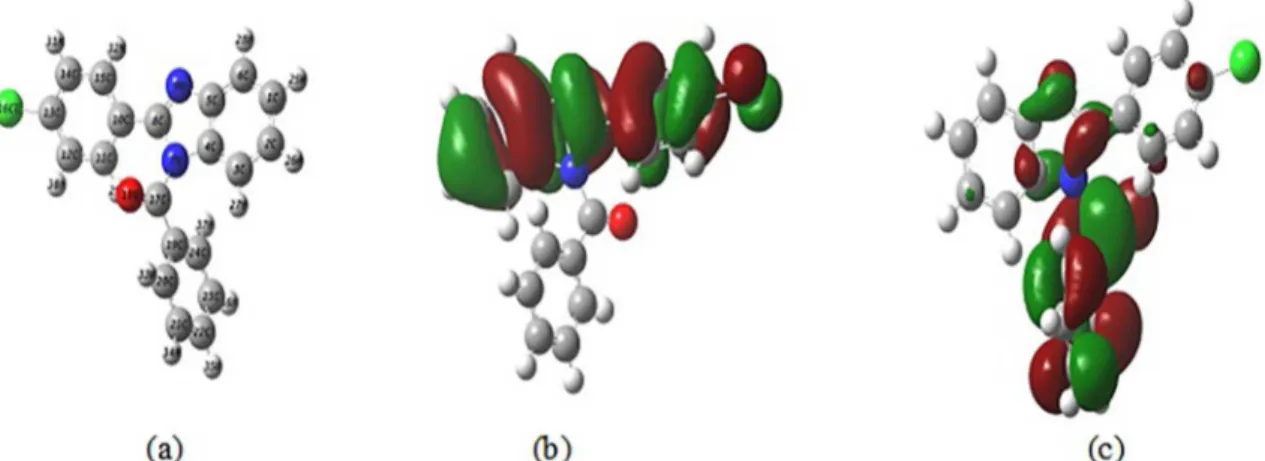
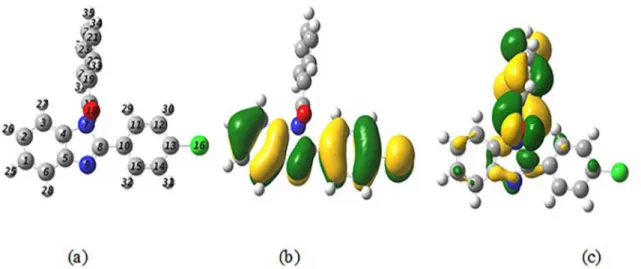
Experimental and Theoretical Study of Corrosion Inhibition of Mild Steel in 1.0 M HCl Medium by 2(-4( hloro phenyl-1H- benzo[d]imidazol)-1-yl)phenyl)methanone
Texto
Imagem

Documentos relacionados
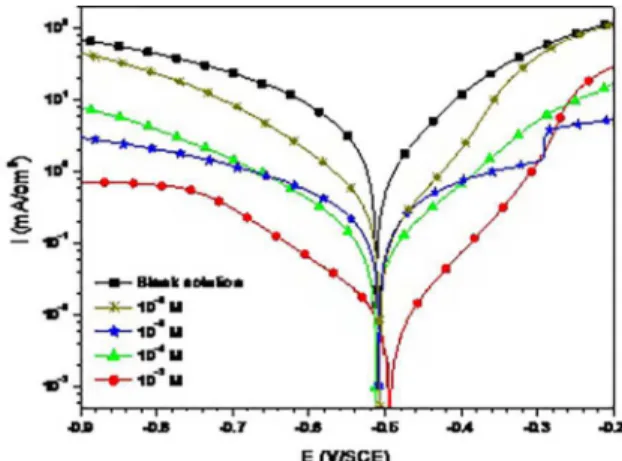
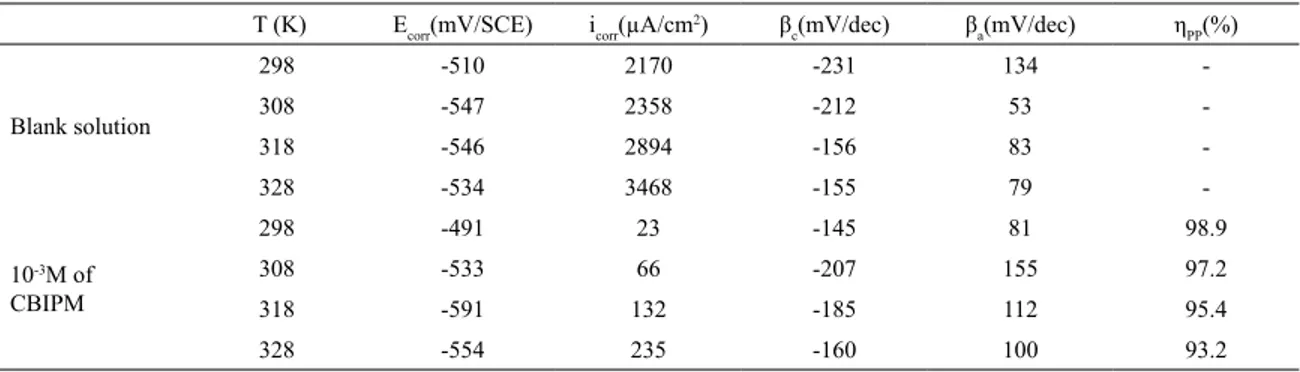
This behavior could be related to a change in the anodic reaction mechanism (iron dissolution) which is corroborated by a decrease in the anodic Tafel slope with
In this research, the effect of mesoporous silica loaded by inhibitor was examined on the corrosion behavior of mild steel in the epoxy coatings.. To prove
In this work, the effect of heat treatments on the microstructure and corrosion resistance of an AISI 430 steel was studied by means of double loop electrochemical
The corrosion inhibition of mild steel in 20% formic acid at 30-60 °C in absence and presence of different concentrations of gemini surfactants was studied using weight
The corrosion behavior of both sintered 316L and HA-316L composites was evaluated by electrochemical techniques in simulated body luid (Ringer’s solution) and in 0.1M HCl solution
The behavior of roasted cofee extract and its isolated high molecular weight fraction have been investigated as carbon steel corrosion inhibitors in HCl solution by weight
5: Variation of inhibition efficiency (%) with inhibitor concentrations (M) for mild steel coupons in 2M HCl solution containing pyridoxol hydrochloride at different
Bitter orange, Citrus Aurantium (CA), extract and one of its chemical constituents, Linalool, have been evaluated as a corrosion inhibitor for mild steel in 0.5 mol L -1