w w w . r e u m a t o l o g i a . c o m . b r
REVISTA
BRASILEIRA
DE
REUMATOLOGIA
Review
article
What
rheumatologists
should
know
about
orofacial
manifestations
of
autoimmune
rheumatic
diseases
Aline
Lauria
Pires
Abrão
a,∗,
Caroline
Menezes
Santana
b,
Ana
Cristina
Barreto
Bezerra
a,
Rivadávio
Fernandes
Batista
de
Amorim
b,
Mariana
Branco
da
Silva
c,
Licia
Maria
Henrique
da
Mota
d,
Denise
Pinheiro
Falcão
baProgramadePós-Graduac¸ãoemCiênciasdaSaúde,FaculdadedeCiênciasdaSaúde,UniversidadedeBrasília(UnB),Brasília,DF,Brazil bProgramadePós-Graduac¸ãoemCiênciasMédicas,FaculdadedeMedicina,UniversidadedeBrasília(UnB),Brasília,DF,Brazil
cFaculdadedeCiênciasdaSaúde,UniversidadedeBrasília(UnB),Brasília,DF,Brazil dServic¸odeReumatologia,HospitalUniversitáriodeBrasília(UnB),Brasília,DF,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received4February2015 Accepted28August2015 Availableonline16March2016
Keywords:
Autoimmunerheumaticdiseases Orofacialmanifestations Saliva
Orallesions Periodontaldisease
a
b
s
t
r
a
c
t
Orofacial manifestationsoccur frequently inrheumatic diseases andusually represent earlysignsofdiseaseorofitsactivitythatarestillneglectedinclinicalpractice.Among theautoimmunerheumaticdiseaseswithpotentialfororalmanifestations,rheumatoid arthritis(RA),inflammatorymyopathies(IM),systemicsclerosis(SSc),systemiclupus ery-thematosus(SLE),relapsingpolychondritis(RP)andSjögren’ssyndrome(SS)canbecited. Signsandsymptomssuchasoralhyposalivation,xerostomia,temporomandibularjoint dis-orders,lesionsoftheoralmucosa,periodontaldisease,dysphagia,anddysphoniamaybe thefirstexpressionoftheserheumaticdiseases.Thisarticlereviewsthemainorofacial man-ifestationsofrheumaticdiseasesthatmaybeofinteresttotherheumatologistfordiagnosis andmonitoringofautoimmunerheumaticdiseases.
©2016ElsevierEditoraLtda.ThisisanopenaccessarticleundertheCCBY-NC-ND license(http://creativecommons.org/licenses/by-nc-nd/4.0/).
O
que
o
reumatologista
deve
saber
sobre
as
manifestac¸ões
orofaciais
das
doenc¸as
reumáticas
autoimunes
Palavras-chave:
Doenc¸asreumáticasautoimunes Manifestac¸õesorofaciais Saliva
Lesõesbucais Doenc¸aperiodontal
r
e
s
u
m
o
Manifestac¸õesorofaciaisocorremcomfrequêncianasdoenc¸asreumáticase,comumente, representamsinaisiniciais oude atividadeda doenc¸aqueaindasãonegligenciadosna práticaclínica.Entreasdoenc¸asreumáticasautoimunescompossíveismanifestac¸õesorais, incluem-se a artrite reumatoide(AR),miopatiasinflamatórias (MI), esclerosesistêmica (ES), lúpuseritematososistêmico(LES), policondriterecidivante(PR)esíndromede Sjö-gren (SS). Sinais e sintomas orofaciais como hipossalivac¸ão, xerostomia, disfunc¸ões
∗ Correspondingauthor.
E-mail:[email protected](A.L.Abrão).
http://dx.doi.org/10.1016/j.rbre.2016.02.006
temporomandibulares, lesões na mucosa bucal, doenc¸a periodontal, disfagia e disfo-nia podem ser a primeira expressão dessasdoenc¸as reumáticas.Esse artigo revisaas principais manifestac¸ões orofaciais das doenc¸as reumáticasque podem ser de inter-esse doreumatologista, para diagnóstico e acompanhamentodas doenc¸as reumáticas autoimunes.
©2016ElsevierEditoraLtda.Este ´eumartigoOpenAccesssobumalicenc¸aCC BY-NC-ND(http://creativecommons.org/licenses/by-nc-nd/4.0/).
Introduction
Autoimmunerheumaticdiseasesconstituteaheterogeneous group of conditions characterized by immune tolerance breakdownandproductionofautoantibodiesandofa num-ber of substances responsible for lesions in several body structures.Inthiscategory,rheumatoidarthritis(RA), inflam-matory myopathies (IM), systemic sclerosis (SSc), systemic lupuserythematosus(SLE)andSjögren’ssyndrome(SS)can beincluded.1
Somerheumaticdiseasesshowmucocutaneous manifes-tations.Generally,thechangesareconsequencesofsystemic disordersandmanifestthemselvesinsidiously,showingsigns andsymptomsintheoralcavity(Table1).However,inthe con-textofautoimmune diseases,the oralapproachappearsto havenotyetarousedscientificinterest.Inthispaper,some dentalclinicalfindingsoftenfoundinpatientstreatedatthe RheumatologyOutpatientClinicofBrasília,Hospital Univer-sitáriodeBrasilia (HUB)–UNBwillbediscussed,basedon a narrativeliteraturereview.Forthisreview,thefollowingterms wereenteredinPubMeddatabase(AutoimmuneRheumatic Disease[allfields])AND“dentistry”[allfields],limitedtothose studiesconductedonhumansubjects.Itwasfoundthatare onlysixty-eightstudieswere publisheduntilJune21,2015. Somestudiespointtoepidemiologicaldata ofmedicaland dentalinterest.Inthiscontext,clearlyonerealizesthelimited approachtothesubject.However,thepaperschosen demon-stratethatthedentistcanandshouldactintheearlydiagnosis andmanagementofthesedisorders,sincethesepatientshave specificneeds.
Thus,thisnarrativereviewaimstoaddressthemain orof-acialmanifestationsinautoimmunerheumaticdiseasesthat maybeofinterest totherheumatologist fordiagnosis and clinicalfollow-up.
Literature
review
Rheumatoidarthritis
Rheumatoidarthritis(RA)isachronicautoimmune inflam-matorydiseaseofunknownetiology.2Theclassicfeaturesof
thisdiseasearechronic,bilateralandsymmetric polyarthri-tis,jointpainandinflammationthatcanresultindeformity, instability and destruction of synovial joints.3,4 RA affects
more often the synovial membrane of small joints of the extremities,resultinginswelling,edemaandpain,andcan leadtoboneandcartilagedestruction,severedisabilityand untimelymortality.3
ThemostcommonoralmanifestationsinpatientswithRA are:
Temporomandibulardisorder(TMD)
The temporomandibularjoint (TMJ) isasynovial joint and can be affected bydisorders innon-articular tissues, with manifestationsofmusclespasm,fibromyalgia,andmyotonic dystrophy,amongothers.However,TMJjointtissuesmayalso beaffectedbymechanicaltrauma,infection,iatrogenic disor-ders,andgout,aswellasbyautoimmunerheumaticdiseases suchasRAandpsoriasis.5Onecanobservethepresenceof
typical inflammatorymediators ofosteoarthritis, including tumornecrosisfactor(TNF)-␣,interleukin(IL)-1,IL-6and IL-8.Thesefindingsmaintaincorrelationwiththeextentofthe disease,i.e.,clinicalsymptoms,numberofjointeffusionsor morphologicalchanges.6,7
TMDsareconsideredtobethemostcommonconditions causingorofacialpainofnon-dentalorigin,and thedentist isthe professional responsibleforthe clinicalexamination ofTMJ andforrequestingimagingexams ofthis anatomi-calregion.ATMDcanmanifestsymptomssuchasearpain, headache,non-specificnervepain,andtoothache.Its diagno-sisrequiresamedicalanddentalapproach,whichmakesthe evaluationoftheprevalenceofTMDacomplexissue,andits studyisoftenoverlookedintheclinicalpracticeofrheumatic autoimmunediseases.8,9
TMD can occur bothin adultsand –more commonly– in children with RA. A study that evaluated 223 children with juvenile idiopathic arthritis revealed that 38.6% had some TMJinvolvement (pain, swelling and/orlimitation in range of motion).10 When TMJ involvement is manifested
during a child’s development, there may be a mandibu-lar growth restriction, resulting in micrognatia and/or ankylosis.11
Inadults,studiesontheprevalenceofTMDinRApatients resultedinextremelyvariedvalues(5–86%),dependingonthe populationstudied,diagnosticcriteriaused,andassessment methods.4,5TMDisthemostcommonorofacialmanifestation
inRApatients.Thepatientmayshowabilateral,profound and pervasiveacute pain, which isexacerbated duringthe function.Theclinicalexaminationmayreveal:malocclusion, sensitivity andinflammationofpre-auricular regions, joint stiffnessuponwaking,limitationofjawmovement, intracap-sularcrepitusorclickingandpaininmasticatoryand/orneck muscles.4,12Imagingstudiesmayshowbonestructurelossat
Table1–Oralmanifestationsofautoimmunerheumaticdiseasesandtheirclinicalimplications.
Oralevents Autoimmunerheumaticdiseases
RA IM SSc SLE SS Clinicalimplications
PM DM
Periodontaldisease X X X •Worseningfactorfordiabetesandrheumatic
andheartdiseases
Dentalcaries X X X •Dependingontheextentoftheinjury,pain,
chewinginvolvement,andfociofinfectioncan occur,likelyworseningdiabetesandrheumatic andheartdiseases
Candidiasis X X •Itchingand/orburninginthemucosa
•Riskofesophagealinfection •Inappetence
Hyposalivation X X X X •Dysphonia
•Dysphagia
•Thrushandulcersintheoralmucosa •Greatertendencytooralandoropharyngeal
infections30
•Recurrentesophagitis
•Sleepinterruptedforwaterintakeand urination
Xerostomia X X X X X •Decreaseinqualityoflife
Halitosis X •Decreaseinqualityoflife
Mouthburning X X X X •Dysgeusia
•Eatingdifficulty •Cancerphobia
Oralulcers X X X •Pain
•Difficultyinfeedingandoralhygiene
TMD X X X X •Headache
•Otalgiaand/ortinnitus •Afeelingofeartamponade •Anirradiatingcervicalpain •Chronicheadache •Limitedmouthopening •Difficultytochewandspeak12
Microstomia X •Limitedmouthopening
•Difficultyineatingandtohaveagoodoral hygiene46
Regionalresorptionof jawbone/TMJ
X •Limitedmouthopening
Dysphagia X X X •Dehydration
•Malnutrition
Aspirationofsecretionsand/orfoodtothe lung–aspirationpneumonia
Dysphonia X X X X •Decreaseinqualityoflife
Changesinlanguage X X X •Difficultytoperceivefood,speechand
swallowing.
Anglecheilitis X •Painandlimitedmouthopening
Alterationsintooth morphology
X •Facialestheticsandmasticatoryfunction
changes
Pathologicalchangesin salivaryglands
X X •Hyposalivation
Changesinmimicand chewingmusclesand inthepharynx
X •Dysphagia
•Dysphonia •Difficultytochew
Trigeminalneuralgia •Episodesofintensepainintheeyes,lips,nose,
scalp,foreheadand/orjaw
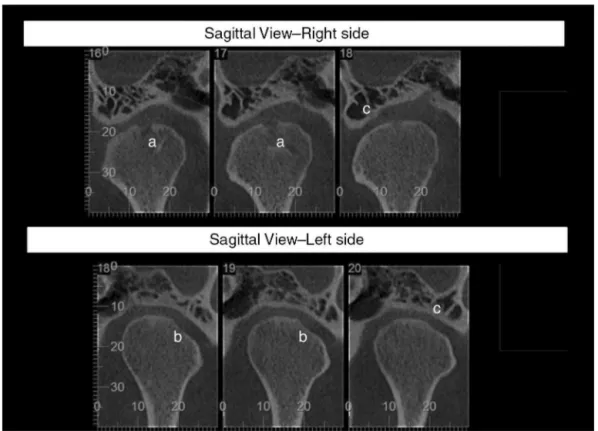
Fig.1–Computedtomographyoftemporomandibularjointofapatientwithrheumatoidarthritisandcomplaintofclicking whilechewing.Presenceofsubchondralcystintheupperportionoftherightmandibularhead(a),erosionoflateralportion oftheleftmandibularhead(b)andflatteningofarticulareminences(c).
Periodontaldisease(PD)
PDisachronicinfectiousdiseasecausedbyGram-negative anaerobic bacteria, affectingthe tissues of protection and supportof the tooth, suchas gums, periodontal ligament, cementum and alveolar bone. Under PD designation, both reversible(gingivitis)andirreversible(periodontitis)processes areincluded.Whenundiagnosedanduntreated,PDcancause progressivedestructionofalveolarbone,causingtooth mobil-ity and subsequent dental loss.14 According to the World
Health Organization, periodontal disease affects approxi-mately10–15% ofthe world population.14 Brazilian official
datashowthat19.4%ofadultsaged35–44yearsarecarriers ofthisdisease.15
Somerecentstudiesalsosuggestasignificantassociation betweenRAandPD.13,16–18TherelationshipbetweenRAand
progressionofinflammatoryconditions(p.ex.,periodontitis) isnotclear.Themainreasonforthisscenarioisthelackof uniformityintheclassificationofthevariousformsofboth diseases.19ItisestimatedthattheprevalenceofPDincreases
twiceinRApatientscomparedtothegeneralpopulation.17
Thus,thepresenceofamoderate-to-severeRAalsoincreases morethan twicetheriskofdevelopingformsof moderate-to-severeperiodontitiscomparedtoindividualswithoutthe disease.17–19
Furthermore,thereisevidenceofsimilarityinthe patho-genesisofRAandPD.MicroorganismssuchasPorphyromonas gingivalismayplayaroleinbothconditions,16beingableto
invadeisolatedhumanchondrocytesinthekneejoint, inter-feringwithcellcycle andinducingthesecells’apoptosis.20
AnotherimportantfactorwouldbethatP.gingivalisexpresses the peptide argininedeiminase (PAD),which converts argi-nine to citrulline by a citrullination process. Thisprocess, which is common to some human proteins, is associated withthepathophysiologyofRA.Ithasalowimmune toler-ancetocitrullinatedproteinsinsynovialfluid,whichtriggers thedevelopmentofimmunoglobulinsagainsttheseproteins, presentinjointsandtendons.21,22 Inaddition,studieshave
demonstratedthepresenceofantibodiesinresponsetooral anaerobicbacteriainthesynovialtissueandserum.Others authorsalsofoundthepresenceoforalbacterialDNAinthe synovialfluidofRApatients.18Infact,RAandPDhavea
vari-etyofmarkedlysimilarclinicalandpathophysiologicfeatures (Table2).23,24
Table2–Pathophysiologicalsimilaritiesinthe characteristicsofrheumatoidarthritisandperiodontal disease.23,24
Pathophysiological characteristics
RA PD
Cellinfiltrate Macrophages,T lymphocytes, plasmacellsand PMN
Equivalent
Immune phenomenon
Immunecomplex deposition, complement fixation
Equivalent
Cytokines IL-1␣,IL-1,IL-6, IL-8,TNF-␣and TGF-
Equivalent
Localcellsaffected Chondrocytesand synoviocytes
Gingival fibroblast, osteoblast,and keratinocytes
Inductionofbone resorption
PGE2,TNF-␣,IL-1 LPS,PGE2, TNF-␣,IL-1
TissueDestruction Metalloproteinase, phospholipase andelastase
Equivalent
Granulationtissue Presentin cartilage/bone interface
Presentinthe cement/bone interface
IL-1␣, interleukin-1 alpha; IL-1, interleukin-1 beta; IL-6, interleukin-6; IL-8, interleukin-8; LPS, lipopolysaccharide; PD, periodontaldisease;PGE2,prostaglandinE2;PMN, polymorphonu-clearleukocytes;RA,rheumatoidarthritis;TNF-␣,tumornecrosis factoralpha;TGF-,growthtransformingfactorbeta.
improvementinendothelialfunction,withadecreaseinlocal andsystemicinflammatoryprocesses.25
Hyposalivation/xerostomia
Amongoral changes,it turns out that hyposalivation (low salivaryflow) and xerostomia (dry mouth) are common in autoimmunerheumaticdiseases,andxerostomiaaffects1% ofRApatients.26 About one thirdofRApatientshave
sec-ondary SS.27 A study including 604 RA patients showed a
decrease in salivary flow in43% ofsubjects.28 The risk of
developinghyposalivationincreaseswiththeseverityofthe disease.Itisworthmentioningthatanotherstudyconducted in483hospitalizedpatientsduetocomplicationsofarthritis foundthatonly17.7%ofxerostomia-positivepatientswere treatedforxerostomia.Incontrast,84.8%ofpatientstreated forxerophthalmiaweretreatedforthiscondition.Itwasalso observed that the therapeutic modalities administered for xerostomia were not effectiveand alsowere notin accor-dancewithcurrentrecommendationsfoundinthemedical literature.29
Therefore,atimelydiagnosisandpropermonitoringofSS associatedwithRAareimportantstepstopromotegainsin thequalityoflifeofthesepatients(aswillbediscussedinthe
SSsection),takingintoaccountthatsalivaperformsfunctions ofsystemicinterest,forinstance,thesenseoftaste, epithe-lialrepairoforopharynxandesophagus,andesophagealacid contentbuffering,amongotherfunctions.30
Inflammatorymyopathies
Polymyositis(PM)anddermatomyositis(DM)areautoimmune diseases classified as idiopathic inflammatory myopathies, beingcharacterizedbymusculoskeletalinflammation.31
PMisasystemicconnectivetissuedisease,characterizedby bilateral,symmetrical,proximalmuscleweakness.Itaffects muscles of the shoulder and pelvic girdle and progresses towardproximalmusclesofthelimbs.Itsonsetisfrequently gradualandprogressive.PMexhibitsageographicallyvariable incidence,withaboutonecaseforevery100,000inhabitants, predominantlyaffectingfemales.32
Impairment of skeletal muscles ofposterior pharyngeal wall and proximal third of the esophagus can lead to oropharyngeal dysphagia, with aspiration and dysphonia. Consequently, thepatient cancomplain ofhypersalivation. Thiscomplication,however,willbeduetoanimpaired func-tionalactivityofswallowingmusclesinassociationwiththe salivary reflexcaused byreflux.Two thirds ofthe patients present involvementoftheneckflexormuscles, whichcan cause difficulty in neck support. Constitutional symptoms includefatigue,low-gradefever,weightloss,andarthralgia orarthritisofsmallandmediumjoints.33
Somerarecasereportsrelatepresenceofulcerationsonthe entiretongue,ofalinearaspectandwithawhitesecretionon theedges,andalsotongueatrophy,inwhichonecanobserve areddenedmucosa.34
DMisanautoimmunediseaseofunknownetiologythat is characterized by a systemic small-vessel vasculopathy predominantlyinvolvingmusclesandskin.Besidesthe cuta-neousinvolvement,thecharacterizationofDMisbasedinthe patternofmuscleinvolvement,presenceofassociatedclinical manifestations,andhistopathologicalchanges.35
The prevalenceoforal involvementinDM is unknown. Mostoftheinformationavailablecomesfromindividualcase reportsorsmallcaseseries,andsomeearlyreportsofcases didnotclearlyseparateMSfromPM.36
Aninvolvementofmimicmusclesmayoccur,whichleads toadecreaseinfacialexpression.Similarly,theinvolvement ofthemasticatoryandpharyngealmusclesmayresultin dys-phagia,dysphonia,andhypersalivation.Theinvolvementof striatedmuscleofthepharynxoresophagusalsocontributes totheoccurrenceofdysphagia.Inpatientswithdysphagia, DM reaches18–50%ofpatientsand correlateswithdisease severity.36,37Inaddition,thepresenceofdysphagiaincreases
theriskofaspirationpneumonia.Mortalityratesrangefrom1 to5years,reaching31%ofpatientswithDManddysphagia.37
However, the occurrence of hypersalivation is not always attributable toan excess insaliva production, but may be causedbyaninabilitytoretainsalivaandswallowingit,dueto theweaknessofperioralmuscletone,orbecauseofdysphagia. Theinvolvementoftonguemuscle resultsinmacroglossia, inadditionofhypotonia,whichcanalsomakeitmore diffi-cult chewing,swallowingand speech.38 Involvementofthe
Mucosaledema, erythemaand telangiectasia are the com-monestoralchanges.38
Although27.5%ofpatientswithDMalsosufferarthritis, TMJinvolvementisrare,withonlyonecasereportedinthe literature.Insomereports,thepresenceofprominentblood vesselsthroughouttheoralmucosaandaphthousstomatitis/ ulcer-likelesionsweredescribed.40About10–46%ofpatients
developpainfuloralandgingivalulcers.41Theteethhaveshort
andbulgingroots,withobliterationofrootcanalsaswellas pulpchambercalcification.Xerostomiaisalsoseenasa com-moncomplaint.42
Systemicsclerosis
Systemic sclerosis (SSc)isan autoimmune disease charac-terizedbyinflammationandhyper-reactivity ofmicro-and macrovascularcirculationassociatedwithexcessivecollagen deposition intissues, withsubsequent fibrosis ofthe skin and/orinternalorgans.43SSchasapredilectionforfemales,
withanincidenceof2–10/onemillioninhabitantsinthe gen-eralpopulation.44Inaddition,thereisaconsensusaboutan
increaseinmorbidityandmortality,withanestimated66% survivalat10years.45
The oral manifestationsare scarcely studied and often neglectedbyclinicians,althoughleadingtomajorfunctional disability.Microstomiaisthemostcommonoralfindingand developsduetocollagendepositioninperioraltissues, caus-inglimitation ofmouth opening,perioralgroovewrinkling, and soft palate, larynx and oral mucosa stiffness.46
Fur-thermore, hyposalivation and/or dry mouth are secondary manifestationsofthedisease.TMDcanalsooccur,with vary-ingdegreesofsubsequentresorptionofmandibularbranch, coronoidprocess,menton and condyle.5 Itisbelieved that
these areas are reabsorbed due to the chronic collagen deposition.Tonguecancerhasasignificantlyincreased fre-quencyinpatientswithSScthat presentamouth opening <30mm.47
Theresorptionofsometeethhasalsobeenreportedwith somefrequencyinthesepatients.There maybean abnor-malincrease in the frequency ofdecayed teethand of an
atypical tooth eruption. Apparently there is also a predis-position forthe occurrenceofPD,due toincreasedplaque buildup.Thisproblemarisesfromthedifficultyofcleaningthe mouth(causedbyasmallermouthopening)andintheuse ofthedentalbrush.Thislattercomplicationisdueto scle-roticchangesinfingersandhands.Furthermore,theuseof systemiccorticosteroidsforlongperiodsinfluencein reduc-ingtheperiodontalinflammatoryresponse,thusmakingthis processaprogressiveandofteninsidiousone.48
Systemiclupuserythematous
SLE isan autoimmune disease ofunknown etiology, influ-enced by environmental and genetic factors, and which mainly affects women inthe second and thirddecades of life.49 The prevalence of oral lesions in patients with SLE
variesbetween6.5%and21%.SLEaffects primarilytongue, oralmucosa,lipsandpalate.Forthisreason,oralulcersare considered primaryevents,thatare includedinthe follow-ingactivityindexesofthisdisease:BILAG(BritishIslesLupus AssessmentGroup),50SLEDAI(SystemicLupusErythematosus
Disease Activity Index),51 SELENA-SLEDAI (Safety of
Estro-gens in LupusErythematosus National Assessment),SLAM (SystemicLupusActivityMeasure),52 andECLAM(European
ConsensusLupusActivityMeasurement).53
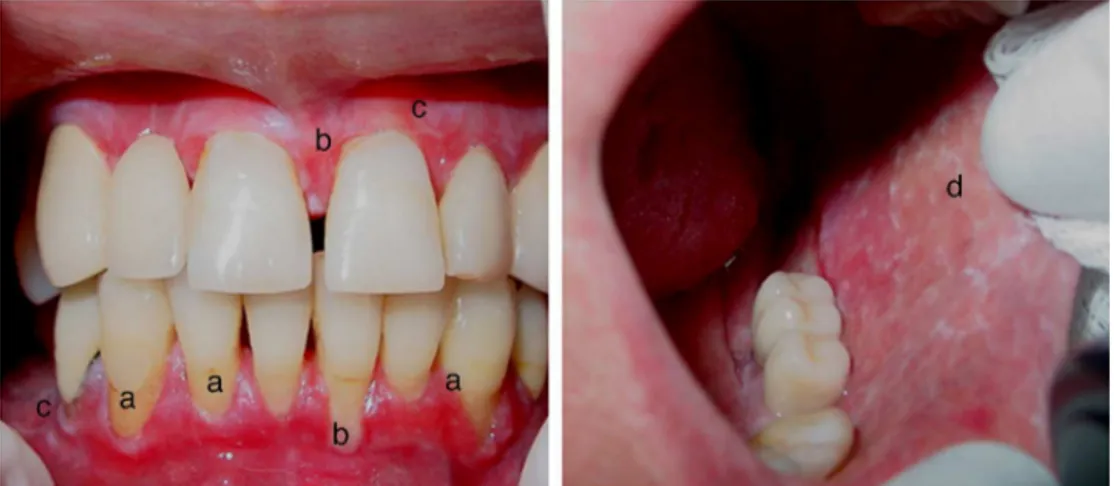
Thelesionsappearindifferentways,suchasblemishes and plaques on the mucosa. The lesions may be erythe-matous, ulcerated,ofa recurrentaphthousstomatitis, and lichenplanus-orleukoplakia-likelesions(Fig.2).Thesizeof these lesionsisalsovariable,from asmall surface erosion to ulcers covering a wide and extensive area.54,55 The few
studiesonorallesionsinpatientswithSLEshow, microscop-ically,parakeratosisororthokeratosis,acanthosis,epithelial atrophy,vacuolardegenerationofthebasalmembranewith necrosisofbasalkeratinocytes,basementmembrane thick-ening,lichenoidmononuclearinfiltrate,anddeepconnective tissuevasculitis.Injuriesinthevermilionborderoflips (espe-cially in the lower lip), deserve special attention,as these lesions may be related to lupus cheilitis, with or without epithelialdysplasia.54,56
Othersecondaryorofacialsigns/symptomsinclude:mouth burning,hyposalivation,xerostomia, salivary glanddisease (suchasfocal necrosisofthe parotidgland),TMD, desqua-mative gingivitis and PD.54 Hyposalivation can lead to an
increasedoccurrenceofdentalcariesandtoapredisposition tocandidiasis,especiallyifimmunosuppressiveagentssuch ascorticosteroidsarebeingused.56
Sjögren’ssyndrome(SS)
SSisaninflammatoryautoimmunediseasepresentinga fre-quentchroniccourse,inwhich thelymphocyticinfiltration ofexocrineglands,particularlylacrimalandsalivaryglands, impairs its secretory function.55 Simultaneously, systemic
manifestationsofcutaneous,respiratory,renal,hepatic, neu-rologicand vascular naturecan occur. SS hastwo distinct forms:primarySS–notassociatedwithanotherdisorder;and secondarySS–inwhichthepatientexpressesthissyndrome inassociationwithotherautoimmunediseases.57,58
ItisestimatedthatSSaffects0.2%oftheworld popula-tion,mainlywomen,inaratioof9:1.57,58InBrazil,duetothe
absenceofofficialestimatesorscientificallyconfirmeddata onitsincidence,nooneknowstheexactnumberofpatients withthissyndrome.However,itwasstatedthatthemajorityof diagnosedcasesarerelatedtomenopausal,orolder,women.59
SSfollowsavariablecourseandexhibitsawidespectrum ofclinicalmanifestations.Inaddition,manyofitssymptoms arenon-specific,makingdifficultanddelayingthediagnosis. EightypercentofpatientswithSSexhibitaninsidiousonsetof symptomsofdrynessthatdevelopoveraperiodfromseveral monthstoyears.58
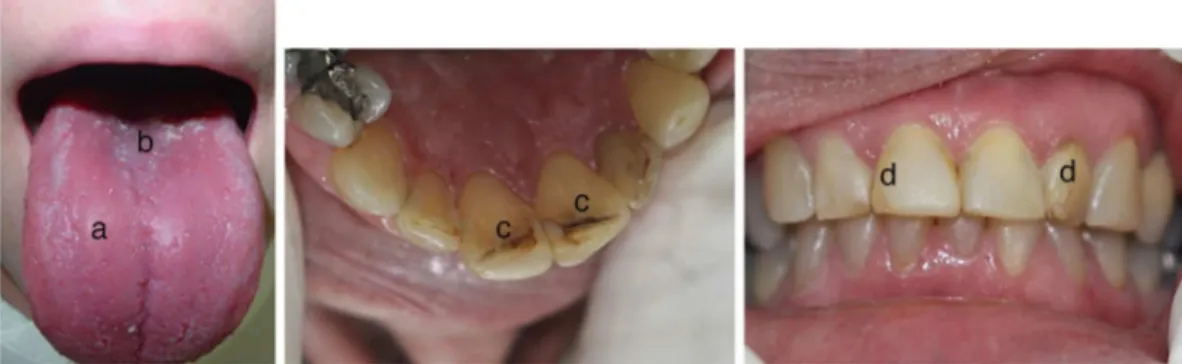
TheoralmanifestationsobservedinpatientswithSSare attributedtotheinvolvementofsalivaryglands,whichleads tolesssalivarysecretion.Inconsequence,theworse lubrica-tionandlossofbufferingandantimicrobialactionofsalivary secretion increase the incidence of oral/dental infections, mucosal friability, and symptomsof irritationand burning mouth(Fig.3).57Ontheotherhand,somepatientscomplain
ofxerostomia,whichmaynotbeaccompaniedbyadecrease insalivary secretion.30 However,in the initialstage ofthe
disease, when the diagnosis has not yet been well estab-lished,patientsmaycomplainofxerostomiaduetochangesin
salivarycomposition,ortoareductionofsalivarysecretion fromthesmallersalivaryglands(fromlipmucosaandpalate). Thus,sialometrymay revealthatthe patienthasanormal salivaryflow;however,salivarycompositiontestswillindicate qualitativechanges.60
Usually,dentalcariesandfungalinfectionsareobservedin mucousmembranes(especiallycandidiasis)thatcanmanifest aspseudomembranousorerythematouslesions.Thefriability of the mucosa in patients with SS often leads tosoft tis-sueinjuries.Suchsignsincludedryandcrackedlips,median rhomboidglossitisorafissuredtongue,lossoflingual papil-lae,stomatitis,angularcheilitis,aphthousinjury,lipmucosal ulcers,difficultyinswallowingsolids,andodynophagia.57
SS patients oftendisplay voice disorders andcorrelated symptomsthatareassociatedwithadecreaseintheirquality oflife.Itisknownthatthelubricationofthevocalcordsis carriedoutbysaliva.61Thus,thisbiologicalfluidisimportant
foraproperphonation.
Another relevantpoint referstothe drop inthe quality oflifeofpatientswithSS,becauseoftheirchangingeating habits,causedbydrymouth.62ThereisaStrongcorrelation
amongoraldrynessandfatigue,pain,psychologicaldistress, andaworsequalityofsleep;andthatitisconsideredasa car-diovascularriskfactor.63Inthisstudy,theauthorsconcluded
thatamultidisciplinarytherapeuticapproachmaybethebest waytominimizedrymouthanditsconsequencesinpatients withprimarySS.63
Finally,anothercommonoralmanifestationisan asymp-tomaticandself-limitingincreasesofparotidglandsorother majorsalivaryglands,55whichmaybepointingtotheearly
stageofSS.
Therefore,theestablishmentofanearlydiagnosisofSSis essentialforthechoiceofthecorrecttreatment,which con-sistsinrelievingthesignsandsymptomsinordertominimize oravoidsequelsthatcanimpactonthehealthandqualityof lifeofpatients.64
Gustatory,mechanicalandchemicalsialogogueshavebeen usedtostimulatesalivaproduction.However,the effective-ness of these resources is low, because they provide only temporaryrelief,requiringfrequentapplications.65Many
top-icaltreatmentssuchassprays,lozenges,mouthwashes,gels, oilsortoothpasteshavebeenevaluated,butthereisnostrong
evidencethatanyofthesetopicaltreatmentsiseffectiveto alleviatethepatientwithdrymouth.66 Oxygenatedtri-ester
glycerol-basedlubricantsaremoreeffectivethanwater-based electrolytesprays.Chewinggumincreasessalivaproduction, butthereisnoevidencethattheseproductsarebetterorworse than saliva substitutes. However, acidic secretagogues and those containingsugar shouldbe avoided,66 because these
productsloweroralpH,promotegreatertooth demineraliza-tionandirritateamucousalreadyverysensitive.Oneshould optfortheuseofsugar-freechewinggum,butcontaining flu-orideandbicarbonateinitscomposition.Thesecomponents increasesalivarypHandassistinpreventingtoothdecay.67,68
Chemical sialagogues, such as pilocarpine and cevime-line,areeffectiveinrelievinghyposalivation,butmaycause adverseeffects.65Electricalstimulationappliedtothe
affer-entpathways(throughtheoralmucosaorskin)inareasof salivaryglands,showedincreasedsalivaproductionandrelief ofdrymouthinpatientswithSS65andinpatientsundergoing
cervical-brainradiotherapy.69
Asystematicreviewofrandomizedcontrolledtrialswas conductedtogatherevidenceondrugtherapyinprimarySS. Theauthorssuggestedthatsalivasubstitutesandsugar-free chewinggumsmaybeeffectiveincasesofmild-to-moderate drymouth.Consumptionofalcoholandsmokingshouldbe avoided, and it is a critical factor the establishment of a thoroughoralhygiene.Thetreatmentofchoiceforpatients withresidual functionofsalivaryglandsiscevimelineand oralpilocarpine. However,nostudywas published compar-ing theefficacy ofthese twodrugs. Thedoses which have shown better effects in terms ofefficacy and safety were: pilocarpine5mgevery 6h; andcevimeline 30mgevery 8h. N-acetylcysteinecouldbeanalternativeinpatientswith con-traindicationsorintolerancetomuscarinicagonists.70
Conclusion
Orofacial manifestations in patients with autoimmune rheumaticdiseasesarecommonproblems,butstillsparsely addressed by rheumatologists in their everyday clinical practice.Thisarticleproducedasummaryofthemain mani-festationsobserved,inordertofamiliarizetheseprofessionals withtheirdiagnoses,underlyingthepossibleneedforanearly referraltothedentist.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
Acknowledgements
TheauthorswouldliketoacknowledgeNathalyaLopesSilva, RafaellyStavale,TalithaGiovannadaSilvaandFrancisca Ires-daniaAlvesMacedofortheircollaboration.Thesecondand seventhauthorsarealsogratefulforthefinancialsupportof CAPES–Coordenac¸ãodeAperfeic¸oamentodePessoaldeNível Superior.
r
e
f
e
r
e
n
c
e
s
1.MoscaM,ChiaraT,RosariaT,StefanoB.Undifferentiated connectivetissuediseases(UCTD):simplifiedsystemic autoimmunediseases.AutoimmunRev.2011;10:256–8.
2.HelmickCG,FelsonDT,LawrenceRC,GabrielS,HirschR, KwohCK,etal.Estimatesoftheprevalenceofarthritisand otherrheumaticconditionsintheUnitedStates:PartI. ArthritisRheum.2008;58:15–25.
3.AletahaD,NeogiT,SilmanAJ,FunovitsJ,FelsonDT,Birnbaum NS,etal.2010rheumatoidarthritisclassificationcriteria:an AmericanCollegeofRheumatology/EuropeanLeagueAgainst Rheumatismcollaborativeinitiative.ArthritisRheum. 2010;62:2569–81.
4.SidebottomA,SalhaR.Managementofthe
temporomandibularjointinrheumatoiddisorders.BrJOral MaxillofacSurg.2013;51:191–8.
5.AlikoA,CiancagliniR,AlushiA,TafajA,RuciD. Temporomandibularjointinvolvementinrheumatoid arthritis:systemiclupuserythematosusandsystemic sclerosis.IntJOralMaxillofacSurg.2011;40:704–9.
6.TakahashiT,KondohT,FukudaM,YamazakiY,ToyosakiT, SuzukiR.Proinflammatorycytokinesdetectableinsynovial fluidsfrompatientswithtemporomandibulardisorders.Oral SurgOralMedOralPatholOralRadiolEndodontol.
1998;85:135–41.
7.KaneyamaK,SegamiN,NishimuraM,SuzukiT,SatoJ. Importanceofproinflammatorycytokinesinsynovialfluid from121jointswithtemporomandibulardisorders.BrJOral MaxillofacSurg.2002;40:418–23.
8.MelchiorreD,CalderazziA,BongiSM,CristofaniR,BazzichiL, EligiC,etal.Acomparisonofultrasonographyandmagnetic resonanceimagingintheevaluationoftemporomandibular jointinvolvementinrheumatoidarthritisandpsoriatic arthritis.Rheumatology.2003;42:673–6.
9.ManfrediniD,Guarda-NardiniL,WinocurE,PiccottiF,Ahlberg J,LobbezooF.Researchdiagnosticcriteriafor
temporomandibulardisorders:asystematicreviewofaxisI epidemiologicfindings.OralSurgOralMedOralPatholOral RadiolEndodontol.2011;112:453–62.
10.CannizzaroE,SchroederS,MullerLM,KellenbergerCJ, SaurenmannRK.Temporomandibularjointinvolvementin childrenwithjuvenileidiopathicarthritis.JRheumatol. 2011;38:510–5.
11.ScrivaniSJ,KeithDA,KabanLB.Temporomandibular disorders.NEnglJMed.2008;359:2693–705.
12.Roldán-BarrazaC,JankoS,VillanuevaJ,ArayaI,LauerHC.A systematicreviewandmeta-analysisofusualtreatment versuspsychosocialinterventionsinthetreatmentof myofascialtemporomandibulardisorderpain.JOralFacial PainHeadache.2013;28:205–22.
13.KlasserGD,BalasubramaniamR,EpsteinJ.Topicalreview– connectivetissuediseases:orofacialmanifestations includingpain.JOrofacPain.2007;21:171–84.
14.PetersenPE,OgawaH.Strengtheningthepreventionof periodontaldisease:theWHOapproach.JPeriodontol. 2005;76:2187–93.
15.Bascones-MartinezA,Matesanz-PerezP,Escribano-Bermejo M,González-MolesM-Á,Bascones-IlubdainJ,MeumanJ-H, etal.Periodontaldiseaseanddiabetes–reviewofthe literature.MedOralPatolOralCirBucal.2011;16: e722–9.
rheumatoidarthritisandtheirrelatives.JRheumatol. 2010;37:1105–12.
17.BerthelotJM,GoffBL.Rheumatoidarthritisandperiodontal disease.JointBoneSpine.2010;77:537–41.
18.OgrendikM,KokinoS,OzdemirF,BirdPS.Serumantibodies tooralanaerobicbacteriainpatientswithrheumatoid arthritis.MedscapeGenMed.2005;7:2.
19.MercadoF,MarshallRI,KlestovAC,BartoldPM.Relationship betweenrheumatoidarthritisandperiodontitis.JPeriodontol. 2001;72:779–87.
20.PischonN,RoehnerE,HockeA,GuessanPN,MüllerHC, MatziolisG,etal.EffectsofPorphyromonasgingivalisoncell cycleprogressionandapoptosisofprimaryhuman
chondrocytes.AnnRheumDis.2009;68:1902–7.
21.DetertJ,PischonN,BurmesterGR,ButtgereitF.The associationbetweenrheumatoidarthritisandperiodontal disease.ArthritisResTher.2010;12:218.
22.DeSmitMJ,BrouwerE,VissinkA,vanWinkelhoffAJ. Rheumatoidarthritisandperiodontitis;apossiblelinkvia citrullination.Anaerobe.2011;17:196–200.
23.PreshawPM,TaylorJJ.Howhasresearchintocytokine interactionsandtheirroleindrivingimmuneresponses impactedourunderstandingofperiodontitis?JClin Periodontol.2011;38:60–84.
24.LundbergK,WegnerN,Yucel-LindbergT,VenablesPJ. PeriodontitisinRA—thecitrullinatedenolaseconnection. NatRevRheumatol.2010;6:727–30.
25.TonettiMS,D’AiutoF,NibaliL,DonaldA,StorryC,ParkarM, etal.Treatmentofperiodontitisandendothelialfunction.N EnglJMed.2007;356:911–20.
26.TuressonC,O’FallonWM,CrowsonCS,GabrielSE,Matteson EL.Occurrenceofextraarticulardiseasemanifestationsis associatedwithexcessmortalityinacommunitybased cohortofpatientswithrheumatoidarthritis.JRheumatol. 2002;29:62–7.
27.AndonopoulosA,DrososAA,SkopouliFN,MoutsopoulosHM. Sjogren’ssyndromeinrheumatoidarthritisandprogressive systemicsclerosis.Acomparativestudy.ClinExpRheumatol. 1988;7:203–5.
28.RussellSL,ReisineS.Investigationofxerostomiainpatients withrheumatoidarthritis.JAmDentalAssoc(1939). 1998;129:733–9.
29.GuobisZ,BasevicieneN,PaipalieneP.Aspectsofxerostomia prevalenceandtreatmentamongrheumaticinpatients. Medicina(Kaunas,Lithuania).2007;44:960–8.
30.FalcãoDP,MotaLMHD,PiresAL,BezerraACB.Sialometry: aspectsofclinicalinterest.RevBrasilReumatol.
2013;53:525–31.
31.ScolaR,WerneckL,PrevedelloD.Polimiositee dermatomiosite.Dendrito,Curitiba.1999;4:77–82.
32.YaziciY,KagenLJ.Clinicalpresentationoftheidiopathic inflammatorymyopathies.RheumDisClinNAm. 2002;28:823–32.
33.LundbergIE,DastmalchiM.Possiblepathogenicmechanisms ininflammatorymyopathies.RheumDisClinNAm. 2002;28:799–822.
34.GibsonJ,LameyPJ,ZomaA,BallantyneJ.Tongueatrophyin mixedconnectivetissuedisease.OralSurgOralMedOral Pathol.1991;71:294–6.
35.MastagliaFL,OjedaVJ.Inflammatorymyopathies:part1.Ann Neurol.1985;17:215–27.
36.TanakaTI,GeistSMRY.Dermatomyositis:acontemporary reviewfororalhealthcareproviders.OralSurgOralMedOral PatholOralRadiol.2012;114:e1–8.
37.OhTHE,BrumfieldKA,HoskinTL,StolpKA,MurrayJA, BasfordJR.Dysphagiaininflammatorymyopathy:clinical characteristics,treatmentstrategies,andoutcomein62 patients.MayoClinProc.2007;82:441–7.
38.TanakaTI,GeistSRY.Dermatomyositis:acontemporary reviewfororalhealthcareproviders.OralSurgOralMedOral PatholOralRadiol.2012;114:e1–8.
39.MártonK,HermannP,DankóK,FejérdyP,MadlénaM,NagyG. Evaluationoforalmanifestationsandmasticatoryforcein patientswithpolymyositisanddermatomyositis.JOral PatholMed.2005;34:164–9.
40.BrennanMT,PatronasNJ,BrahimJS.Bilateralcondylar resorptionindermatomyositis:acasereport.OralSurgOral MedOralPatholOralRadiolEndodontol.1999;87:446–51.
41.HuberAM,DuganEM,LachenbruckPA,FeldmanBM,Perez MD,ZemelLS,etal.Preliminaryvalidationandclinical meaningoftheCutaneousAssessmentToolinjuvenile dermatomyositis.ArthritisCareRes.2008;59:214–21.
42.AkdisCA,AkdisM,BieberT,Bindslev-JensenC,Boguniewicz M,EigenmannP,etal.Diagnosisandtreatmentofatopic dermatitisinchildrenandadults:EuropeanAcademyof AllergologyandClinicalImmunology/AmericanAcademyof Allergy,AsthmaandImmunology/PRACTALLConsensus Report.Allergy.2006;61:969–87.
43.TambyMC,ChanseaudY,GuillevinL,MouthonL.New insightsintothepathogenesisofsystemicsclerosis. AutoimmunRev.2003;2:152–7.
44.KatsambasA,StefanakiC.Life-threateningdermatosesdue toconnectivetissuedisorders.ClinDermatol.2005;23:238–48.
45.SilmanA.Scleroderma–demographicsandsurvival.J Rheumatol.1997;Suppl48:58–61.
46.YuenHK,MarlowNM,ReedSG,MahoneyS,SummerlinLM, LeiteR,etal.Effectoforofacialexercisesonoralaperturein adultswithsystemicsclerosis.DisabilRehabil.2012;34: 84–9.
47.AlantaA,CabaneJ,HachullaE,PrincG,GinistyD,HassinM, etal.Recommendationsforthecareoforalinvolvementin patientswithsystemicsclerosis.ArthritisCareRes. 2011;63:1126–33.
48.NagyG,KovácsJ,ZeherM,CzirjákL.Analysisoftheoral manifestationsofsystemicsclerosis.OralSurgOralMedOral Pathol.1994;77:141–6.
49.Al-RayesH,Al-SwailemR,ArfinM,SobkiS,RizviS,TariqM. Lupusaroundtheworldsystemiclupuserythematosusand infections:aretrospectivestudyinSaudis.Lupus.
2007;16:755–63.
50.HayEM,BaconPA,GordonC,IsenbergDA.TheBILAGindex:a reliableandvalidinstrumentformeasuringclinicaldisease activityinsystemiclupuserythematosus.QJM.
1993;86:447–58.
51.BombardierC,GladmanDD,UrowitzMB,CaronD,ChangCH, AustinA,etal.DerivationoftheSLEDAI.Adiseaseactivity indexforlupuspatients.ArthritisRheum.1992;35:630–40.
52.LiangMH,SocherSA,LarsonMG,SchurPH.Reliabilityand validityofsixsystemsfortheclinicalassessmentofdisease activityinsystemiclupuserythematosus.ArthritisRheum. 1989;32:1107–18.
53.VitaliC,BencivelliW,IsenbergDA,SmolenJS,SnaithML, SciutoM,etal.Diseaseactivityinsystemiclupus
erythematosus:reportoftheConsensusStudyGroupofthe EuropeanWorkshopforRheumatologyResearch.II. Identificationofthevariablesindicativeofdiseaseactivity andtheiruseinthedevelopmentofanactivityscore.The EuropeanConsensusStudyGroupforDiseaseActivityinSLE. ClinExpRheumatol.1991;10:541–7.
54.BrennanMT,ValerinMA,Nape ˜nasJJ,LockhartPB.Oral manifestationsofpatientswithlupuserythematosus.Dent ClinNAm.2005;49:127–41.
56.AlbiliaJB,LamDK,ClokieCM,SándorGK.Systemiclupus erythematosus:areviewfordentists.JCanDentAssoc. 2007;73:823–30.
57.KassanSS,MoutsopoulosHM.Clinicalmanifestationsand earlydiagnosisofSjogrensyndrome.ArchInternMed. 2004;164:1275.
58.DawsonLJ,SmithPM,MootsRJ,FieldES.Sjogren’ssyndrome– timeforanewapproach.Rheumatology(Oxford).
2000;39:234–7.
59.BarbieriR,ChiereghinA.SíndromedeSjögren.Temas ReumatolClín.2009;10:88–93.
60.FalcãoDP,LealSC,VieiraCN,WolffA,AlmeidaTF,NunesFP, etal.Sialometryofupperlabialminorglands:aclinical approachbytheuseofweighingmethodSchirmer’stest stripspaper.SciWorldJ.2014:268634.
61.TannerK,PierceJL,MerrillRM,MillerKL,KendallKA,RoyN. Thequalityoflifeburdenassociatedwithvoicedisordersin Sjögren’ssyndrome.AnnOtolRhinolLaryngol.
2015;124:721–7.
62.LanfranchiH,AnsolaM.Drymouthandnutritionqualityof lifeinpatientswithSjögrensyndrome.OralSurgOralMed OralPatholOralRadiol.2013;116:e499.
63.GandíaM,Morales-EspinozaEM,Martín-GonzálezRM, RetamozoS,KostovB,Belenguer-PrietoR,etal.Factors influencingdrymouthinpatientswithprimarySjögren syndrome:usefulnessoftheESSPRIindex.OralHealthDent Manage.2014;13:402–7.
64.MiedanyYE,AhmedI,MouradHG,MehannaAN,AtySA, GamalHM,etal.Quantitativeultrasonographyandmagnetic resonanceimagingoftheparotidgland:cantheyreplacethe histopathologicstudiesinpatientswithSjogren’ssyndrome? JointBoneSpine.2004;71:29–38.
65.StrietzelFP,LafaurieGI,MendozaGR,AlajbegI,PejdaS, VuleticL,etal.Efficacyandsafetyofanintraoral
electrostimulationdeviceforxerostomiarelief:amulticenter, randomizedtrial.ArthritisRheum.2011;63:180–90.
66.FurnessS,WorthingtonHV,BryanG,BirchenoughS,
McMillianR.Interventionsforthemanagementofdrymouth: topicaltherapies.CochraneDatabaseSystRev.2011;
7:CD008934.
67.AndersonLA,OrchardsonR.Theeffectofchewing
bicarbonate-containinggumonsalivaryflowrateandpHin humans.ArchOralBiol.2003;48:201–4.
68.BijellaMF,BrighentiFL,BuzalafiMAR.Fluoridekineticsin salivaaftertheuseofafluoride-containingchewinggum. BrazOralRes.2005;19:256–60.
69.LakshmanAR,BabuGS,RaoS.Evaluationofeffectof transcutaneouselectricalnervestimulationonsalivaryflow rateinradiationinducedxerostomiapatients:apilotstudy. JCancerResTher.2015;11:229.