J. Evid. Based Med. Healthc., pISSN- 2349-2562, eISSN- 2349-2570/ Vol. 3/Issue 85/Oct. 24, 2016 Page 4627
REVIEW OF DENGUE DEATHS: ACUTE LIVER FAILURE AS A MAJOR CAUSE OF MORTALITY
N. K Thulaseedharan 1, K.G Sajeeth Kumar2, E.T. Arun Thomas3, Bhagya Shaji4, Nevin Sadeep51Professor and HOD, Department of General Medicine, Government Medical College, Kozhikode. 2AdditionalProfessor, Department of General Medicine, Government Medical College, Kozhikode. 3Junior Resident, Department of General Medicine, Government Medical College, Kozhikode. 4Junior Resident, Department of General Medicine, Government Medical College, Kozhikode. 5Junior Resident, Department of General Medicine, Government Medical College, Kozhikode.
ABSTRACT
BACKGROUND
Dengue fever is a mosquito-borne viral disease endemic in many tropical and sub-tropical countries. The annual average number of dengue fever cases reported has increased dramatically in recent years. The analysis of deaths due to dengue fever will help in sensitizing treating physicians to reduce mortality in future. Aim of the study to determine the causes of mortality in dengue fever.
MATERIALS AND METHODS
Retrospective analysis of deaths due to dengue fever was carried out in a tertiary care centre in North Kerala. The study period was from 1st June to 31st December, 2015, during a major dengue epidemic. The data was collected from the hospital records
using a pre-designed proforma.
RESULTS
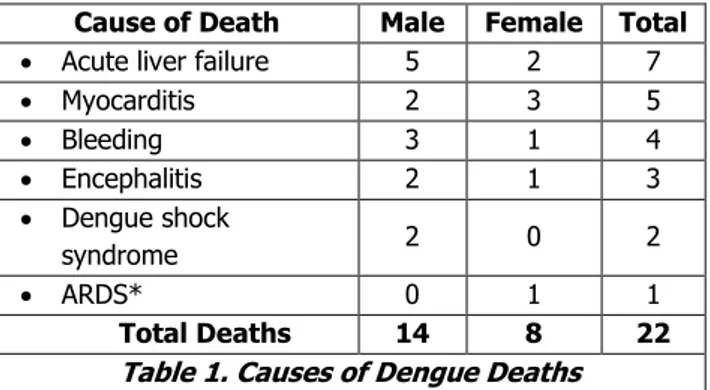
There were 22 deaths among a total of 2525 confirmed cases of dengue fever. Acute liver failure (ALF) was the major cause of death, observed in 7 patients. All the patients with ALF had serum transaminase levels more than 30 times the upper limit of normal with mean AST to ALT ratio of 1.6:1. Five patients with acute liver failure had hepatomegaly. Five deaths occurred due to myocarditis, which was the second major cause identified. Other causes were major bleeding (4 cases), encephalitis (3 cases), dengue shock syndrome (2 cases) and acute respiratory distress syndrome (1 case).
CONCLUSIONS
Acute liver failure was the leading cause of mortality in dengue fever. Hepatic sinusoidal endothelial damage resulting in sinusoidal obstruction is a possible mechanism; supported by the observation that majority of patients with ALF had hepatomegaly. Dengue virus infection should always be considered as a differential diagnosis of acute liver failure in endemic areas. Other fatal complications of dengue fever were myocarditis and major bleeding.
KEYWORDS
Dengue fever, Acute liver failure, Myocarditis, Encephalitis, Thrombocytopenia, Dengue Shock Syndrome, Bleeding.
HOW TO CITE THIS ARTICLE: Thulaseedharan NK, Kumar KGS, Thomas ETA, et al. Review of dengue deaths: acute liver failure as a major cause of mortality. J. Evid. Based Med. Healthc. 2016; 3(85), 4627-4631. DOI: 10.18410/jebmh/2016/976
BACKGROUND
Dengue fever is a mosquito-borne viral disease endemic in many tropical and sub-tropical countries causing major epidemics that have a significant economic and health toll. Dengue virus, an arbovirus of the flaviviridae family comprises of four antigenically related but distinct serotypes (DEN-1, DEN-2, DEN-3 and DEN-4) and is transmitted through the bite of the day feeding mosquito, Aedes aegypti. Dengue virus infection usually presents as a short febrile
illness associated with arthralgia, low back pain, myalgia and retro orbital pain.
The first infection with dengue virus is usually mild, whereas subsequent infections with the other serotypes cause severe disease. This is because of cross-reactive but non-neutralizing antibodies from a previous infection that can bind to the newly infecting serotype facilitating the entry and thereby replication of these viruses in host cells. The resulting higher peak viral titres causes a more severe disease, possibly because of enhanced activation of cytokine cascades and the complement system as well as increased endothelial dysfunction and platelet destruction.[1]
Thrombocytopenia and plasma leakage are the usually observed complications. Rarely, acute liver failure (ALF) or central nervous system dysfunction dominates the clinical picture, occurrence of which is associated with high mortality rates.[2-6] Dengue virus infection is also known to produce
myocarditis.[7,8] The annual average number of dengue fever
cases reported has increased dramatically in recent years Financial or Other, Competing Interest: None.
Submission 31-09-2016, Peer Review 06-09-2016, Acceptance 14-10-2016, Published 21-10-2016. Corresponding Author:
Dr. N. K. Thulaseedharan, Professor and HOD,
J. Evid. Based Med. Healthc., pISSN- 2349-2562, eISSN- 2349-2570/ Vol. 3/Issue 85/Oct. 24, 2016 Page 4628 with unusual complications being reported more frequently.
The analysis of deaths due to dengue fever is therefore imperative to determine the fatal complications which will help in sensitizing treating physicians to reduce mortality in future.
AIM OF THE STUDY
To determine the causes of mortality in dengue fever.
METHODS
Our study was an observational study conducted at Calicut Medical College from 1st June to 31st December, 2015.
Calicut Medical College is a 3025-bed tertiary care hospital serving Northern parts of Kerala, India - an area of high dengue transmission where a major epidemic occurred during the study period. A retrospective analysis of all dengue deaths in our centre during this period was performed. Cases of dengue deaths were identified using the hospital inpatient data base. A case of dengue fever was defined as: a compatible clinical illness with either (i) positive serology (IgM dengue antibody) or (ii) positive NS1 antigen. Dengue deaths were defined as deaths occurring as a direct consequence of acute dengue virus infection. Patients aged less than 12 years were excluded from the study. Hospital records were examined in detail to note the clinical and laboratory data, course of the disease and the treatment provided to the patient. The data was collected with the help of a pre-designed proforma.
Acute liver failure was defined in our study as evidence of coagulation failure defined by International Normalized Ratio (INR) ≥2.0, and any degree of mental alteration (encephalopathy) in a patient with no underlying chronic liver disease. Patients with seropositivity for Hepatitis B, C and E, other agents known to cause hepatitis, peripheral smear showing malaria parasite, or positive rapid malarial antigen test and recent history of exposure to any hepatotoxic drugs or toxins were not considered as ALF due to dengue virus infection. Myocarditis was diagnosed when any two of the following were present- cardiac symptoms, elevated biomarker of cardiac injury (Troponin I), ECG findings suggestive of acute injury, suggestive abnormality on echocardiography. Dengue encephalitis was diagnosed in
the setting of altered sensorium with normal metabolic parameters and either MRI changes or positive CSF IgM dengue antibodies.
RESULTS
Twenty-two deaths due to dengue fever satisfied the inclusion and exclusion criteria, among the 2525 dengue fever cases admitted in our centre during the study period. The mortality rate among hospitalised patients with dengue virus infection was 0.9%. Their ages ranged from 14 years to 64 years with a mean age of 38 years. Acute liver failure was the major cause of death, observed in 7 patients. Five deaths occurred due to myocarditis, which was the second major cause identified. Other causes were major bleeding in 4 cases, encephalitis in 3 cases, dengue shock syndrome in 2 cases and acute respiratory distress syndrome in 1 case (Table 1).
Cause of Death Male Female Total
Acute liver failure 5 2 7
Myocarditis 2 3 5
Bleeding 3 1 4
Encephalitis 2 1 3
Dengue shock
syndrome 2 0 2
ARDS* 0 1 1
Total Deaths 14 8 22
Table 1. Causes of Dengue Deaths
* Acute respiratory distress syndrome
Acute Liver Failure
ALF resulted in 7 out of 22 deaths (31.8%). Hepatomegaly was present in 5 out of the 7 patients. Peak total bilirubin level ranged from 1.6-6.9 mg/dl. All the patients had AST and ALT levels 30 times above the upper normal limit. Six patients had AST level greater than ALT. Mean AST to ALT ratio was 1.6. None of the patients with acute liver failure had documented hypotension during any period to suggest ischemic hepatitis (Table 2). All patients were treated with intensive supportive care.
Patient Characteristic Case 1 Case 2 Case 3 Case 4 Case 5 Case 6 Case 7
Age Range: 14-46 years
Mean: 32.4 years
27 44 24 26 14 46 46
Sex
(Males 71.4%, Females 28.6%) F M M M M M F
Fever at the onset of encephalopathy No No No Yes No No Yes
Interval between fever and onset of
encephalopathy 8 days 6 days 5 days 4 days 7 days 6 days 5 days
Hepatomegaly Yes No Yes Yes Yes Yes No
Lowest platelet count (per mm3)
Range: 4,000-60,000 Mean: 23,000
47,000 8,000 19,000 9,000 60,000 4,000 19,000
Lowest WBC count (per mm3)
Range: 2,200-8,300 Mean: 4,700
J. Evid. Based Med. Healthc., pISSN- 2349-2562, eISSN- 2349-2570/ Vol. 3/Issue 85/Oct. 24, 2016 Page 4629 Peak Total Bilirubin (in mg/dl)
Range: 1.6-6.9 Mean: 3.9
5.2 6.9 3.3 3.2 2.4 1.6 4.5
Peak AST* (in IU/L)
Range: 2,040-7,792 Mean: 4,843
3,088 6,520 4,282 7,792 2,040 6,520 3,660
Peak ALT** (in IU/L)
Range: 1,364-4,230 Mean: 3,022
2,060 3,280 3,222 3,248 3,752 4,230 1,364
AKI Stage*** 0 I 0 II I 0 II
Table 2. Acute Liver Failure
*Aspartate aminotransferase **Alanine aminotransferase
***Acute Kidney Injury, KDIGO staging
Myocarditis
Myocarditis resulted in 5 out of 22 deaths. ST segment and T wave abnormalities in electrocardiogram were seen in 4 of these patients. All the patients were hemodynamically stable at the time of hospital admission. Sudden hemodynamic decompensation upon mild physical activity led to the death of 4 of these patients (Table.3).
Patient Characteristic Case 1 Case 1 Case 3 Case 4 Case 5
Age Range: 21-58 years
Mean: 37 years
48 22 36 58 21
Sex
(Males 40%, Females 60%) F F F M M
Fever at the onset of myocarditis Yes Yes No Yes No
Interval between fever and onset of
myocarditis 5 days 4 days 6 days 3 days 8 days
Lowest platelet count(per mm3)
Range: 8,000-81,000 Mean: 38,000
22,000 60,000 19,000 8,000 81,000
Lowest WBC count (per mm3)
Range: 1,600-8,700 Mean: 4,200
4,000 3,500 8,700 3,400 1,600
Blood pressure at the time of hospital
admission (in mm of Hg) 130/80 90/60 106/70 90/60 100/70
Electrocardiogram
T wave inversion
V3-V6
ST segment elevation
V1-V4
Biphasic T waves V3
-V5
Diffuse T wave inversions
Sinus tachycardia
Troponin I Elevated Elevated Not elevated Elevated Elevated
Table 3. Myocarditis
Major Bleeding
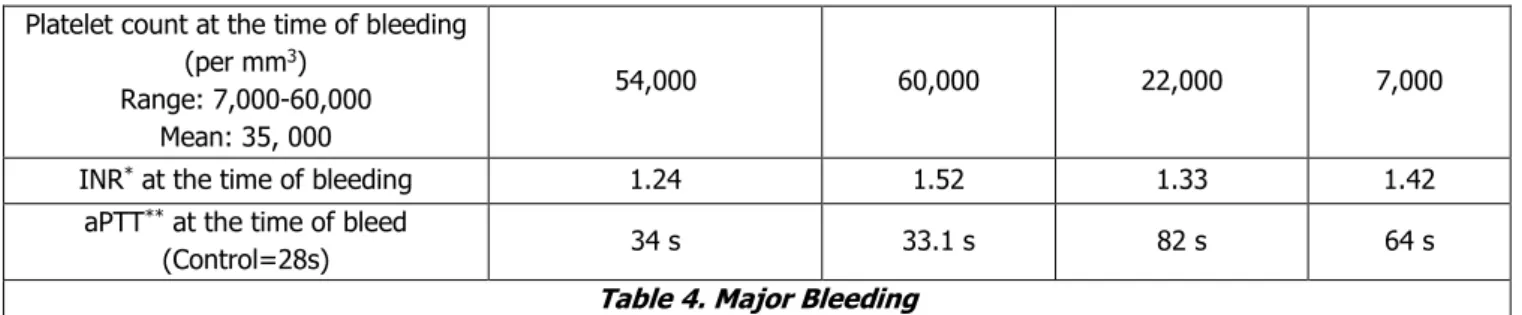
Major bleeding resulted in 4 deaths (out of total 22 deaths). Platelet count at the time of bleeding ranged from 7,000 to 60,000 per mm3. All these patients had mild prolongation of INR and aPTT (table.4).
Patient Characteristics Case 1 Case 1 Case 3 Case 4
Age Range: 21-58 years
Mean: 37 years
56 62 30 64
Sex M M M F
Fever at the onset of bleeding No No Yes No
Interval between fever and onset of
bleeding 6 days 6 days 5 days 5 days
Site of bleeding Upper GI bleed, haematemesis
Alveolar
haemorrhage Haemoperitoneum
J. Evid. Based Med. Healthc., pISSN- 2349-2562, eISSN- 2349-2570/ Vol. 3/Issue 85/Oct. 24, 2016 Page 4630 Platelet count at the time of bleeding
(per mm3)
Range: 7,000-60,000 Mean: 35, 000
54,000 60,000 22,000 7,000
INR* at the time of bleeding 1.24 1.52 1.33 1.42
aPTT** at the time of bleed
(Control=28s) 34 s 33.1 s 82 s 64 s
Table 4. Major Bleeding
*International normalised ratio
**Activated partial thromboplastin time.
Dengue Encephalitis
Three deaths were due to encephalitis, confirmed by CSF IgM dengue antibodies. All of them had altered sensorium and two had seizures. CT brain and routine CSF study was normal in all patients. MRI was done in 2 patients, which showed T2 hyper intensity of bilateral thalami.
Dengue Shock Syndrome
Two patients died due to dengue shock syndrome. Both of them had hypotension, pleural effusion, ascites, haematocrit more than 50, hypoalbuminemia and 4+ albuminuria. They failed to respond to fluid resuscitation and colloids. Their clinical course was further complicated by acute tubular necrosis and ischemic hepatitis.
DISCUSSION
Dengue virus infection has emerged as one of the most prevalent infectious diseases in the tropical and sub-tropical countries. Unusual complications, notably acute liver failure is being recognised more frequently.[9] We retrospectively
reviewed 22 deaths due to dengue fever in a tertiary care centre during a recent epidemic to determine the exact causes of mortality. In our study, acute liver failure emerged as the leading cause of death. The exact mechanism of hepatic failure in dengue fever is unknown. One postulated mechanism is the hepatic sinusoidal endothelial damage resulting in sinusoidal obstruction. This leads to ischemic hepatitis even in the absence of systemic hypotension, as the portal blood constitutes more than 75% of blood flow to liver.[10] This hypothesis is supported by our observation that
majority of patients with ALF had hepatomegaly. This is unusual in other causes of ALF but is characteristically observed in hepatic sinusoidal obstruction syndrome.[11]
Severe plasma leak and hypotension due to myocarditis could also contribute to the ischemic hepatitis but none of our patients with ALF had these complications. Our patients had markedly elevated serum transaminases level with higher AST levels. This observation was similar to the findings observed in various case reports on dengue associated ALF.[12-20]
Myocarditis was the second leading cause of death due to dengue virus infection. Dengue virus infection is known to produce myocarditis but the exact frequency of occurrence is unknown [7, 8]. One important observation made in our study was that all the patients were hemodynamically stable at the time of hospital admission,
but mild physical exertion led to sudden decompensation and death, signifying the importance of strict bed rest in severe dengue fever.
Dengue encephalitis though considered rare, was observed in 3 of our patients confirmed by CSF dengue antibodies. Thalamic involvement was seen in the MRI studies of 2 patients. Kamble et al has described a similar case of dengue encephalitis featuring JE-like thalamic involvement visualized on CT brain.[21]
Contrary to popular belief, major haemorrhage and plasma leakage were down in the list of the causes of death due to dengue fever. Platelet counts at the time of major haemorrhage ranged between 7,000 and 60,000 per mm3.
This indicates that absolute platelet count is an unreliable predictor for major haemorrhages. All patients had mild prolongation of INR and aPTT at the time of bleeding, indicating that coagulopathy is also a contributing factor for bleeding. Mild prolongation of prothrombin and partial thromboplastin time and reduced fibrinogen levels have been noted in several studies. However, levels of fibrin degradation products were not elevated to a degree consistent with classical disseminated intravascular coagulation.[22-24]
Our study could analyse all the deaths among a large group of patients with dengue fever, and was appropriately centred in an area most significantly affected by the epidemic. However, analysis of the non-fatal cases, if done, would have increased the strength of the study. Mortality studies of dengue fever epidemics can bring to light the major epidemiological changes in the pattern of fatal complications. This can sensitise practising physicians in early recognition and treatment of the same. Larger multi-centric studies are indispensable in tackling this emerging disease.
CONCLUSION
Acute liver failure was the leading cause of mortality in dengue fever. Hepatic sinusoidal endothelial damage resulting in sinusoidal obstruction is a possible mechanism; supported by the observation that majority of patients with ALF had hepatomegaly. Dengue virus infection should always be considered as a differential diagnosis of acute liver failure in endemic areas. Other fatal complications of dengue fever were myocarditis and major bleeding.
REFERENCES
J. Evid. Based Med. Healthc., pISSN- 2349-2562, eISSN- 2349-2570/ Vol. 3/Issue 85/Oct. 24, 2016 Page 4631 2. Nimmannitya S, Thisyakorn U, Hemsrichart V. Dengue
haemorrhagic fever with unusual manifestations. Southeast Asian J Trop Med Public Health 1987;18(3):398-406.
3. Chhour YM, Ruble G, Hong R, et al. Hospital-based diagnosis of hemorrhagic fever, encephalitis, and hepatitis in Cambodian children. Emerg Infect Dis 2002;8(5):485-489.
4. Solomon T, Dung NM, Vaughn DW, et al. Neurological manifestations of dengue infection. Lancet 2000;355(9209):1053-1059.
5. Patey O, Ollivaud L, Breuil J, et al. Unusual neurologic manifestations occurring during dengue fever infection. Am J Trop Med Hyg 1993;48(6):793-802.
6. Gubler DJ. Dengue and dengue hemorrhagic fever. Clin Microbiol Rev 1998;11(3):480-496.
7. Obeyesekere I, Hermon Y. Myocarditis and cardiomyopathy after arbovirus infections (dengue and Chikungunya fever). British Heart Journal I972;34:821-827.
8. Kabra SK, Juneja R, Madhulika, et al. Myocardial dysfunction in children with dengue haemorrhagic fever. Natl Med J India 1998;11(2):59-61.
9. Deepak NA, Patel ND. Differential diagnosis of acute liver failure in India. Ann Hepatol 2006;5(3):150-156. 10. Wachsberg RH, Bahramipour P, Sofocleous CT, et
al. Hepatofugal flow in the portal venous system: pathophysiology, imaging findings, and diagnostic pitfalls. Radiographics 2002;22(1):123-140.
11. Helmy A. Review article: updates in the pathogenesis and therapy of hepatic sinusoidal obstruction syndrome. Aliment Pharmacol Ther 2006;23(1):11-25. 12. Gasperino J, Yunen J, Guh A, et al. Fulminant liver
failure secondary to haemorrhagic dengue in an international traveller. Liver Int 2007;27(8):1148-1151. 13. Ling LM, Wilder-Smith A, Leo YS. Fulminant hepatitis in dengue haemorrhagic fever. J Clin Virol 2007;38(3):265-268.
14. Penafiel A, Devanand A, Tan HK, et al. Use of molecular adsorbent recirculating system in acute liver failure attributable to dengue hemorrhagic fever. J Intensive Care Med 2006;21(6):369-371.
15. Viswanathan S, Iqbal N, Anemon PP, et al. Fatal fulminant hepatic failure in a diabetic with primary dengue. J Trop Med 2010;2010:413561.
16. Subramanian V, Shenoy S, Joseph AJ. Dengue hemorrhagic fever and fulminant hepatic failure. Dig Dis Sci 2005;50(6):1146-1147.
17. Souza LJ, Coelho JM, Silva EJ, et al. Acute hepatitis due to dengue virus in a chronic hepatitis patient. Braz J Infect Dis 2008;12(5):456-459.
18. Giri S, Agarwal MP, Sharma V, et al. Acute hepatic failure due to dengue: a case report. Cases J 2008;1:204.
19. Lawn SD, Tilley R, Lloyd G, et al. Dengue hemorrhagic fever with fulminant hepatic failure in an immigrant returning to Bangladesh. Clin Infect Dis 2003;37(1):e1– e4.
20. Tan SS, Bujang MA. The clinical features and outcomes of acute liver failure associated with dengue infection in adults: a case series. Braz J Infect Dis 2013;17(2):164-169.
21. Kamble R, Peruvamba JN, Kovoor J, et al. Bilateral thalamic involvement in dengue infection. Neurol India 2007;55(4):418-419.
22. Mitrakul C, Poshyachinda M, Futrakul P, et al. Hemostatic and platelet kinetic studies in dengue hemorrhagic fever. Am J Trop Med Hyg 1977;26(5 Pt 1):975-984.
23. Isarangkura PB, Pongpanich B, Pintadit P, et al. Hemostatic derangement in dengue haemorrhagic fever. Southeast Asian J Trop Med Public Health 1987;18(3):331-339.