Rev. Bras. Reumatol. vol.57 número6
Texto
Imagem
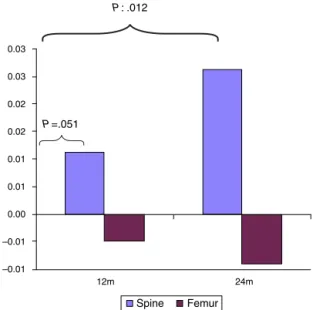
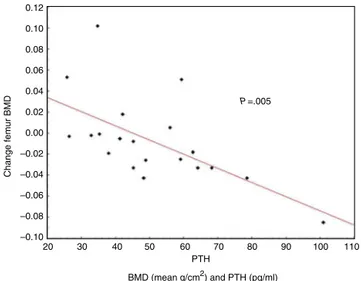
Documentos relacionados
Uma das explicações para a não utilização dos recursos do Fundo foi devido ao processo de reconstrução dos países europeus, e devido ao grande fluxo de capitais no
Neste trabalho o objetivo central foi a ampliação e adequação do procedimento e programa computacional baseado no programa comercial MSC.PATRAN, para a geração automática de modelos
Despercebido: não visto, não notado, não observado, ignorado.. Não me passou despercebido
Peça de mão de alta rotação pneumática com sistema Push Button (botão para remoção de broca), podendo apresentar passagem dupla de ar e acoplamento para engate rápido
The fourth generation of sinkholes is connected with the older Đulin ponor-Medvedica cave system and collects the water which appears deeper in the cave as permanent
financeiras, como ainda por se não ter chegado a concluo entendimento quanto à demolição a Igreja de S. Bento da Ave-Maria, os trabalhos haviam entrado numa fase
Este artigo discute o filme Voar é com os pássaros (1971) do diretor norte-americano Robert Altman fazendo uma reflexão sobre as confluências entre as inovações da geração de
Isto é, o multilateralismo, em nível regional, só pode ser construído a partir de uma agenda dos países latino-americanos que leve em consideração os problemas (mas não as