w w w . e l s e v i e r . c o m / l o c a t e / b j i d
The
Brazilian
Journal
of
INFECTIOUS
DISEASES
Brief
communication
Salivary
anti-PGL-1
IgM
may
indicate
active
transmission
of
Mycobacterium
leprae
among
young
people
under
16
years
of
age
Alexandre
Casimiro
de
Macedo
a,
José
Evandro
Cunha
Jr.
a,
Juliana
Navarro
Ueda
Yaochite
a,
Clodis
Maria
Tavares
b,
Aparecida
Tiemi
Nagao-Dias
a,∗aUniversidadeFederaldoCeará(UFC),FaculdadedeFarmácia,DepartamentodeAnálisesClínicaseToxicológicas,Fortaleza,CE,Brazil
bUniversidadeFederaldeAlagoas,FaculdadedeEnfermagemeFarmácia(ESENFAR),Maceio,AL,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received3January2017
Accepted11May2017
Availableonline9June2017
Keywords:
Leprosy
Salivaryantibodies
Phenolicglycolipid-1antigen
Mycobacteriumleprae
a
b
s
t
r
a
c
t
ConsideringthatthemainrouteofMycobacteriumlepraetransmissionistheupperrespiratory
tract,detectionofsalivaryantibodiescanbeausefultoolfordiagnosingearlyinfection.The
studyaimedtoanalyzesalivaryanti-PGL-1IgAandIgMantibodiesin169childrenaged4–16
yearsold,wholivednearbyorinsidethehouseofmultibacillaryorpaucibacillaryleprosy
patientsintwoendemiccitiesinAlagoasState–Brazil.Salivaryanti-PGL-1antibodieswere
quantifiedbymodifiedELISAmethod.Thefrequencyofcontactandclinicalformofthe
indexcaseweresignificantlyassociatedwithsalivaryantibodylevels.Highfrequencyof
IgMpositivitystronglysuggestsactivetransmissionofM.lepraeinthesecommunities.We
suggestinthepresentworkthatsalivaryanti-PGLIgAandIgMareimportantbiomarkersto
beusedforidentifyingcommunitieswithprobableactivetransmissionofM.leprae.
©2017SociedadeBrasileiradeInfectologia.PublishedbyElsevierEditoraLtda.Thisisan
openaccessarticleundertheCCBY-NC-NDlicense(http://creativecommons.org/licenses/
by-nc-nd/4.0/).
Introduction
Brazil isthe second country withthe highestincidence of
leprosyintheworld.In2015,thecountrypresenteda
detec-tionrateof14.06casesper100,000inhabitants.1Althoughthe
numberofnewcasesseemstodecrease,itmay not
repre-sentthereality.Forinstance,thehighincidenceofthedisease
amongchildrenmeansthatanactivetransmissionoccursin
∗ Correspondingauthor.
E-mailaddress:anagaodias@gmail.com(A.T.Nagao-Dias).
thecommunity.2In2015,thedetectionratesofleprosyamong
people under15 years oldin SantanadoIpanema andRio
Largo,twoBraziliancitieslocatedinAlagoasState,were13.77
and32.81per100,000inhabitants,respectively.3
Asthebacteriaarenotcultivable,secretoryantibodiescan
beausefultooltodetectearlyinfection.Thenasopharynxis
themainportalofentryforMycobacteriumleprae(M.leprae),
andthenasalepithelialcellsareanimportantreservoirofthe
bacteria.4Asmucosalimmuneorgansandtissuescomposean
integratedsystem,salivaisfrequentlyconsideredtobe
repre-sentativeofmucosalhumoralimmuneresponse.Thepurpose
ofthe present work was to evaluatesalivaryanti-phenolic
http://dx.doi.org/10.1016/j.bjid.2017.05.001
1413-8670/©2017SociedadeBrasileiradeInfectologia.PublishedbyElsevierEditoraLtda.ThisisanopenaccessarticleundertheCC
5.0
4.0
3.0
2.0
1.0
0.0
p=0.03 p=0.0019
p=0.028 p=0.055
MB PB MB PB HH PD HH PD
IgM IgA IgM IgA
OD
405
5.0
4.0
3.0
2.0
1.0
0.0
OD
405
A
B
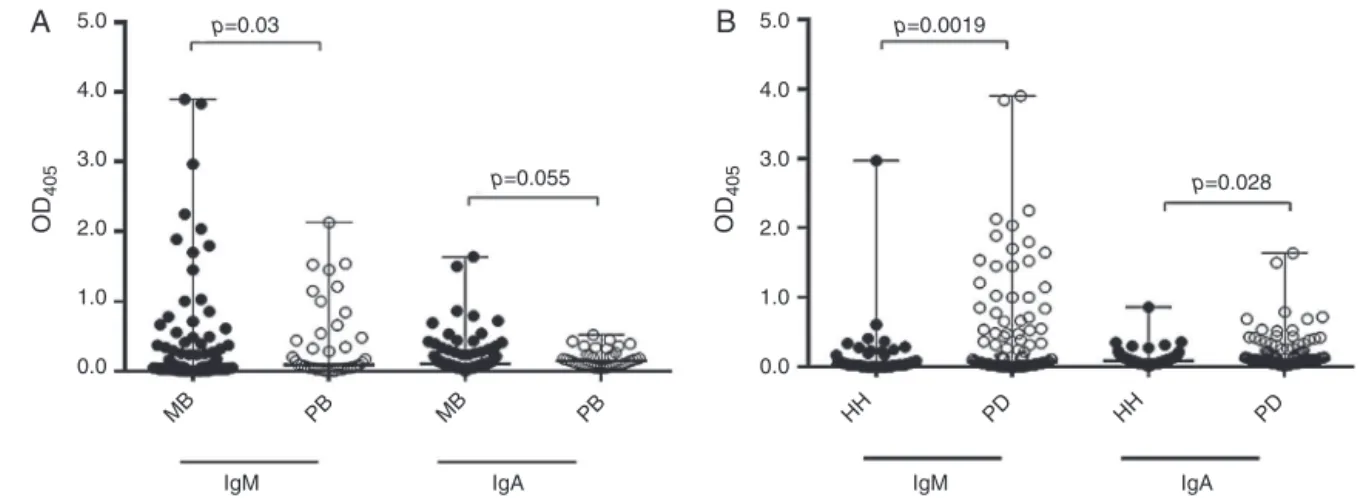
Fig.1–Levelsofsalivaryanti-PGL-1antibodiesin169youngcontactsofleprosypatients.(A)Medianandrangeofsalivary anti-PGL1IgMandIgAincontactsofmultibacillary(MBcontacts,n=115)andpaucibacillary(PBcontacts,n=40)leprosy patients.(B)Medianandrangeofsalivaryanti-PGL1IgMandIgAlevelsinhousehold(HH,n=57)andperidomiciliar(PD, n=112)contacts.Salivaryanti-PGL-1antibodiesweredetectedbymodifiedELISAmethod.
glycolipid1antigen(PGL-1)IgAandIgMisotypesamong169
leprosycontactsaged4–16yearslivinginthemunicipalities
ofSantanadoIpanemaandRioLargo(Alagoasstate,Brazil).
Methods
Subjectsandsamplecollection
Thecontacts(n=169)includedinthestudywereclassifiedas
paucibacillary(PBcontacts,n=40)ormultibacillary(MB
con-tacts,n=115)contacts,accordingtoclinicalformoftheindex
case.Fourteencontactswerenotclassifiedbecausethe
infor-mationwasnotavailableinthepatients’medicalrecords.The
participantswerealsoclassifiedashouseholdcontacts(HH,
n=57)orperidomiciliarcontacts(PD, n=112).Peridomiciliar
contactswerethosewhowererelativesoftheindexcasebut
didnotliveinthesamehouseorthosewholivedclosetothe
indexcase’shouse(uptofivehousesapart).Theprojectwas
approvedbytheNationalCommitteeforEthicsinResearch.
Unstimulatedsalivasampleswerecollectedintotubes,which
weretransportedwithicepackstothelaboratory,wherethey
werekeptat−20◦Cuntiltesting(uptothreeweeksafter
col-lection).Thepresenceoflesionsandnerveenlargementwere
investigatedatthemomentofsamplecollection.Cases
sus-pectedofhavingthedisease werereferredtoadoctorand
excludedfromthestudy.
Detectionofsalivaryanti-PGL-1antibodies
MicroplateswerecoatedwithnativePGL-1at5mg/Lin
abso-lutealcoholfor2hat37◦C(protocol modifiedfromBrito e
Cabraletal.,2013).5Afterblockingwith1%fetalbovineserum
(FBS,LGCBio,Brazil)-Trissolutionfor2hat37◦C,thewells
were incubated with previously cenrifuged saliva samples
(dilutedto1:50with1%FBS-Tris).After18hat4◦Cand
wash-ingwith0.05%FBS-Trissolution,anti-humanIgAoranti-IgM
alkalinephosphatase antibodies (Sigma, USA,1:1000 in1%
FBS-Tris)wereleft on theplates for2h at37◦C.Afternew
incubationfor2hat37◦C,andwashing,thesubstratesolution
(1mg/mL p-nitrophenyl phosphate in 10% diethanolamine
containing 0.5mM MgCl2, pH 9.8) was added to the wells.
After100minatroomtemperature,absorbancereadingswere
recorded at405nm using an ELISA microplate reader. The
resultswereexpressedastheODmeanofthevalues(minus
blank).Thecut-offwasbasedonthe97thpercentileofnormal
controls.6Results30%abovethecut-offvaluewereconsidered
tobepositive.
Analysisofdata
Thedatawereanalyzedusingnonparametrictestsasthedata
didnotfollowaGaussiandistribution(Kolgomorov–Smirnov
test).All statisticalanalysiswasperformedusingGraphPad
Prismversion5.0.Thelevelofstatisticalsignificancewas5%
(p<0.05).
Results
Salivaryanti-PGL-1IgMpresentedgoodcorrelationtosalivary
IgAtiters(Spearmancorrelation,r=0.71,p<0.0001).No
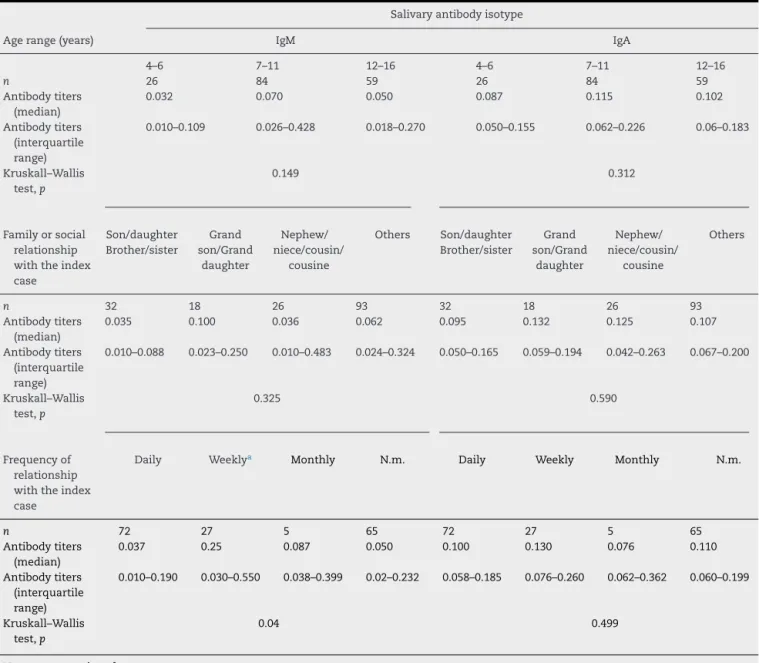
statis-ticalsignificancewasfoundregardingtheagerange,eitherfor
IgMorIgA(Kruskall–Wallistest,p=0.149andp=0.312,
respec-tively,Table1).Nosignificantdifferenceswereeitherfoundin
IgMorIgAtitersinrespecttothedegreeofrelationshipwith
the indexcase(p=0.325andp=0.590,respectively,Table1).
Contactswhoreportedhavingweeklycontactwiththeindex
casehadhigherIgMantibodytitersthanthosewithdaily
con-tact(p=0.04,Table1).MBleprosycontactspresentedhigher
levelsofsalivaryanti-PGL-1IgMandIgA(Mann–Whitneytest,
p=0.03 and p=0.05, respectively)than PB leprosycontacts
(Fig.1A).Interestingly,PDcontactshadhigherlevelsof
sali-varyIgMandIgA(Mann–Whitneytest,p=0.019andp=0.028,
respectively)thantheHHcontacts(Fig.1B).
Discussion
Withtheadventofmultidrugtherapythereportofnewcases
Table1–Titersofsalivaryanti-PGL1IgAandIgMin169youngcontactsofleprosypatientsaccordingtotheagerangeof leprosycontacts,theirdegreeandfrequencyofrelationshipwiththeindexcase.
Salivaryantibodyisotype
Agerange(years) IgM IgA
4–6 7–11 12–16 4–6 7–11 12–16
n 26 84 59 26 84 59
Antibodytiters (median)
0.032 0.070 0.050 0.087 0.115 0.102
Antibodytiters (interquartile range)
0.010–0.109 0.026–0.428 0.018–0.270 0.050–0.155 0.062–0.226 0.06–0.183
Kruskall–Wallis test,p
0.149 0.312
Familyorsocial relationship withtheindex case
Son/daughter Brother/sister
Grand son/Grand
daughter
Nephew/ niece/cousin/
cousine
Others Son/daughter Brother/sister
Grand son/Grand
daughter
Nephew/ niece/cousin/
cousine
Others
n 32 18 26 93 32 18 26 93
Antibodytiters (median)
0.035 0.100 0.036 0.062 0.095 0.132 0.125 0.107
Antibodytiters (interquartile range)
0.010–0.088 0.023–0.250 0.010–0.483 0.024–0.324 0.050–0.165 0.059–0.194 0.042–0.263 0.067–0.200
Kruskall–Wallis test,p
0.325 0.590
Frequencyof relationship withtheindex case
Daily Weeklya Monthly N.m. Daily Weekly Monthly N.m.
n 72 27 5 65 72 27 5 65
Antibodytiters (median)
0.037 0.25 0.087 0.050 0.100 0.130 0.076 0.110
Antibodytiters (interquartile range)
0.010–0.190 0.030–0.550 0.038–0.399 0.02–0.232 0.058–0.185 0.076–0.260 0.062–0.362 0.060–0.199
Kruskall–Wallis test,p
0.04 0.499
N.m.=notmentioned.
a p<0.05inrelationtodailyrelationship.
becomelesssteepinrecentyears;onthecontrary,therehas
beenariseinleprosycasesincludingchildren.6Thismakes
thegoalofeliminatingleprosyimpossibletobeachievedinthe
nextfewyears,2rememberingthattherearepossible
undiag-nosedcasesthatarehiddensourcesofbacterialtransmission.
Inaddition,therearemanyunknownaspectsregardingthe
ecology of M. leprae.6 Strategies are necessary to interrupt
transmission,suchasthedevelopmentofbiomarkersto
iden-tifycontactsand/ortoidentifythoseatriskofdevelopingthe
disease.7
Mucosal immunity in leprosy is poorly understood,
although it is known that the nasal cavity is one of the
firstsitesinfectedbyM.leprae,andthe oralcavitycanalso
be affected, as observed in late-diagnosed patients.8
Sali-varyantibodies ofthe IgAisotypehavebeenconsidered as
biomarkersofinfection,and alsoofimmunity,considering
that they may play a role in inhibiting cell adhesion and
in opsonophagocytosis.9 Smith et al.(2004), in a follow-up
studyofpeopleresidinginendemicregionsforleprosy,found
aninitialpositivityof1.6%forpolymerase-chainreactionof
nasalswaband67.7%forsalivaryanti-M.lepraeIgA.10Avery
interesting aspect observedin the study was that the
fre-quencyofpositivitywashigherincertainseasonalperiods,
especially inthepresenceofhumidity, suggestingthat the
bacillus remains in the community but not necessarily in
theindividual.10Inaccordancewiththishypothesis,Mohanty
andcolleaguesdetectedviablestrainsofM.lepraein
environ-mentalsamplesobtainedfromaroundthehousesofleprosy
patients in Ghatampur (India). The prolonged presence of
bacillicould playanimportantrole inthecontinued
trans-missionofleprosy.11
A very low number ofstudies refer to the presenceof
anti-PGL1IgMinsaliva,5,9,12,13whichpossiblyindicatesrecent
is only five days, and their levels may be correlated with
bacillaryload.12 Abeetal.(1984)foundafrequencyof
pos-itivitycorresponding to4.5%(fiveout of110patients).9 We
foundmuchhigherpositivityofsalivaryanti-PGL1IgM
iso-typeamongleprosycontacts,i.e.17outof47samples(36.1%)
inRioLargo,and15 out of122samples(12.3%)ofchildren
fromSantanadoIpanema.Likewise,inapreviousstudy
car-riedoutinCratoandMaracanaúcities,stateofCeara,Brazil,
13outof135samples(9.6%)turnedoutpositiveforsalivary
anti-PGL1IgM.5 Inthisway,onecouldinferthe magnitude
ofactive transmission in the community. In fact, in 2013,
thecasedetectionrateamongyoungpeopleunder15years
old was 13.25 casesper 100,000 individuals in Santana do
Ipanema,whilenocasewasdetectedinRioLargo.In2014,
nocasewasdetectedinthetwocities.In2015,thecase
detec-tionrateinSantanadoIpanemawas32.81casesper100,000
individuals and 13.77 cases per 100,000 individuals in Rio
Largo.1
Asleprosyinfectionrequiresprolongedcontacttime,those
wholiveinthesamehouse oftheindexcaseisbelievedto
beatriskfordevelopingthedisease;however,recentreports
demonstrated that those who live nearby the index case
shouldalsobeinvestigated.14
Itisanintriguing factfound inourpresent work those
wholived nearbytheindexcasepresentedhigherlevelsof
salivaryantibodiesthan thehouseholdcontacts. The
para-doxtolerance/activationmakesthemucosalimmunesystem
a challenging task. The mucosal immune response may
be affected by various factors,such as soluble or
particu-lateantigens,chemicalnatureandconcentrationofantigen,
frequencyof exposition,gut microbiota composition,
envi-ronmentalantigenicexposure,nutritionalstatus(deficiency
of vitamin A), chronic infections with helminths or other
parasites.15PGL-1,forinstance,facilitatesbacterialadhesion,4
modulatesmacrophagecytokineandchemokineproduction,
andmayleadTcellstoanergy.16Inthisrespect,itis
proba-blethatPGL-1exertssometypeoforaltoleranceonmucosal
immuneresponse.
Natural killer T cells recognize glycolipid antigens
pre-sented byCD1d moleculeand may alsoplayan important
roleinoraltolerancebyinducingtolerogenicdendriticcells
andregulatoryTcells,orbydeletingantigen-specificTcells.17
Thesemechanismscouldpartlyexplainwhatmaybe
occur-ringinchildrenwithprolongedandsustainedcontactwith
theindexcase.
DetectionofpositivesalivaryIgMamongyoungpeople
sug-geststhatM.lepraetransmissionisactiveinthecommunity.
For this reason, a strategy atmunicipal level isextremely
urgentinordertoreducethedisseminationofthebacillus.
Finally,wesuggestinthepresentworkthatsalivaryanti-PGL
IgAand IgMare importantbiomarkerstobeusedfor
iden-tifyingcommunitieswithprobableactivetransmissionofM.
leprae.
Funding
information
This research was financially supported by the
MCTI/CNPq/MS-SCTIE[Process403461/2012-0].
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
Acknowledgements
Wewould liketothankMrs. AnaLúciaCarneiroLeal, Mrs.
GilvâniaFranc¸aVilela,andMrs.AndreaMárciaCostadeFarias
forhelpfulassistance.
r
e
f
e
r
e
n
c
e
s
1.Brasil.SaladeapoioàgestãoestratégicadoMinistérioda
Saúde(SAGE).Indicadoresdemorbidade:hanseníase;2017
[online].Availablefrom:http://sage.saude.gov.br/#/[Accessed
March31,2017].
2.SalgadoCG,BarretoJG,daSilvaMB,FradeMA,SpencerJS. Whatdoweactuallyknowaboutleprosyworldwide?Lancet InfectDis.2016;16:778.
3.Brasil.Diretrizesparavigilância,atenc¸ãoeeliminac¸ãoda
Hanseníasecomoproblemadesaúdepública:manual
técnico-operacional[online].MinistériodaSaúde,Secretaria
deVigilânciaemSaúde,DepartamentodeVigilânciadas
Doenc¸asTransmissíveis–Brasília:MinistériodaSaúde;2016.
Availablefrom:http://portalsaude.saude.gov.br/images/pdf/
2016/fevereiro/04/diretrizes-eliminacao-hanseniase-4fev16-web.pdf
4.SilvaCA,DanelishviliL,McNamaraM,etal.Interactionof
Mycobacteriumlepraewithhumanairwayepithelialcells: adherence,entry,survival,andidentificationofpotential adhesinsbysurfaceproteomeanalysis.InfectImmun. 2013;81:2645–59.
5.BritoeCabralP,JúniorJE,deMacedoAC,etal.Anti-PGL1 salivaryIgA/IgM,serumIgG/IgM,andnasalMycobacterium lepraeDNAinindividualswithhouseholdcontactwith leprosy.IntJInfectDis.2013;17:e1005–10.
6.SmithWC,vanBrakelW,GillisT,SaundersonP,RichardusJH. Themissingmillions:athreattotheeliminationofleprosy. PLoSNeglTropDis.2015;9:e0003658.
7.SmithWC,AertsA.Roleofcontacttracingandprevention strategiesintheinterruptionofleprosytransmission.Lepr Rev.2014;85:2–17.
8.CostaMRSN.Considerac¸õessobreoenvolvimentoda cavidadebucalnahanseníase.HansenInt.2008;33:41–4.
9.AbeM,YoshinoY,MinagawaF,etal.Salivary
immunoglobulinsandantibodyactivitiesinleprosy.IntJLepr. 1984;52:343–50.
10.SmithWC,SmithCM,CreeIA,etal.Anapproachto
understandingthetransmissionofMycobacteriumlepraeusing molecularandimmunologicalmethods:resultsfromthe MILEP2study.IntJLeprOtherMycobactDis.2004;72: 269–77.
11.MohantyPS,NaazF,KataraD,etal.ViabilityofMycobacterium lepraeintheenvironmentanditsroleinleprosy
dissemination.IndianJDermatolVenereolLeprol. 2016;82:23–7.
12.Nagao-DiasAT,AlmeidaTL,OliveiraMeF,SantosRC,LimaAL, BrasilM.Salivaryanti-PGLIgMandIgAtitersandserum antibodyIgGtitersandaviditiesinleprosypatientsandtheir correlationwithtimeofinfectionandantigenexposure.Braz JInfectDis.2007;11:215–9.
séricosesalivaresanti-PGL-1comoparâmetrosdeexposic¸ão ocupacionalaoMycobacteriumleprae.RevPanamInfectol. 2009;11:21–6.
14.BarretoJG,BisanzioD,GuimarãesLdeS,etal.Spatialanalysis spotlightingearlychildhoodleprosytransmissionina hyperendemicmunicipalityoftheBrazilianAmazonregion. PLoSNeglTropDis.2014;8:e2665.
15.LevineMM.Immunogenicityandefficacyoforalvaccinesin developingcountries:lessonsfromalivecholeravaccine. BMCBiol.2010;8:129.
16.DagurPK,SharmaB,UpadhyayR,etal.Phenolic-glycolipid-1 andlipoarabinomannanpreferentiallymodulateTCR-and CD28-triggeredproximalbiochemicalevents,leadingtoT-cell unresponsivenessinmycobacterialdiseases.LipidsHealth Dis.2012;11:119.