H
UMAN
M
UTATION
M
UTATION IN
B
RIEF
Received 27 January 2010; accepted revised manuscript 8 July 2010. © 2010 WILEY-LISS, INC.
DOI: 10.1002/humu.21336
Genetic Screening of LCA in Belgium: Predominance
of CEP290 and Identification of Potential Modifier
Alleles in AHI1 of CEP290-related Phenotypes
Frauke Coppieters1, Ingele Casteels2, Françoise Meire3, Sarah De Jaegere1, Sally Hooghe1,
Nicole van Regemorter4, Hilde Van Esch5, Aušra Matulevičienė6, Luis Nunes7, Valérie Meersschaut8,
Sophie Walraedt9,10, Lieve Standaert10, Paul Coucke1, Heidi Hoeben11, Hester Y. Kroes12, Johan Vande Walle13,
Thomy de Ravel5, Bart P. Leroy1,9,#, and Elfride De Baere1,#
1Center for Medical Genetics Ghent, Ghent University Hospital, Ghent, Belgium; 2Department of Ophthalmology, Leuven University Hospitals, Leuven, Belgium; 3Hôpital Des Enfants Reine Fabiola, Brussels, Belgium; 4Centre de Génétique de Bruxelles, Free University of Brussels, Brussels, Belgium; 5Centre for Human Genetics, Leuven University Hospitals, Leuven, Belgium; 6Department of Human and Medical Genetics, Faculty of Medicine, Vilnius University, Vilnius, Lithuania; 7Service of Medical Genetics, Hospital Dona Estefânia Rua Jacinta Marto, Lisboa, Portugal; 8Department of Radiology, Ghent University Hospital, Ghent, Belgium; 9Department of Ophthalmology, Ghent University Hospital, Ghent, Belgium; 10Revalidation Center Spermalie, Bruges, Belgium; 11Department of Nephrology, Middelheim Hospital, Antwerp, Belgium; 12Department of Medical Genetics, University Medical Center Utrecht, Utrecht, The Netherlands; 13Department of Pediatrics, Ghent University Hospital, Ghent, Belgium; #Equal contribution
*Correspondence to Elfride De Baere, MD, PhD, Phone: +32-9-3325186. Fax: +32-9-3326549. E-mail: [email protected]
Communicated by Stylianos E. Antonarakis
ABSTRACT: Leber Congenital Amaurosis (LCA), the most severe inherited retinal dystrophy, is genetically heterogeneous, with 14 genes accounting for 70% of patients. Here, 91 LCA probands underwent LCA chip analysis and subsequent sequencing of 6 genes (CEP290, CRB1, RPE65, GUCY2D, AIPL1 and CRX), revealing mutations in 69% of the cohort, with major involvement of CEP290 (30%). In addition, 11 patients with early-onset retinal dystrophy (EORD) and 13 patients with Senior-Loken syndrome (SLS), LCA-Joubert syndrome (LCA-JS) or cerebello-oculo-renal syndrome (CORS) were included. Exhaustive re-inspection of the overall phenotypes in our LCA cohort revealed novel insights mainly regarding the CEP290-related phenotype. The AHI1 gene was screened as a candidate modifier gene in three patients with the same CEP290 genotype but different neurological involvement. Interestingly, a heterozygous novel AHI1 mutation, p.Asn811Lys, was found in the most severely affected patient. Moreover, AHI1 screening in five other patients with CEP290-related disease and neurological involvement revealed a second novel missense variant, p.His758Pro, in one LCA patient with mild mental retardation and autism. These two AHI1 mutations might thus represent neurological modifiers of CEP290-related disease. ©2010 Wiley-Liss, Inc.
KEY WORDS: LCA, CEP290, AHI1, modifier, genotype-phenotype correlation
OFFICIAL JOURNAL
INTRODUCTION
Leber Congenital Amaurosis (LCA; MIM# 204000) was first described as a congenital type of retinitis pigmentosa (RP). Approximately 20% of all blind children are thought to suffer from this disease. Phenotypic features include a congenital onset, severely reduced or absent electroretinogram (ERG), nystagmus, the oculo-digital sign and a fundus aspect varying from normal to severely atrophic. Two main types of LCA have been reported, based on the presence or absence of photophobia, night blindness, hyperopia, macular/peripheral retinal abnormalities and measurable visual acuity (Hanein et al., 2004; Hanein et al., 2006). LCA displays variable expression, and seems to represent the extreme and severe end of a spectrum of inherited retinal disease.
LCA is predominantly inherited in an autosomal recessive manner. So far, one locus - LCA9 (Keen et al., 2003) - and the following 14 genes have been identified: GUCY2D (Perrault et al., 1996), RPE65 (Marlhens et al., 1997), CRX (Freund et al., 1998), AIPL1 (Sohocki et al., 2000a), RPGRIP1 (Dryja et al., 2001), CRB1 (den Hollander et al., 2001), RDH12 (Perrault et al., 2004), IMPDH1 (Bowne et al., 2006), CEP290 (den Hollander et al., 2006), RD3 (Friedman et al., 2006), LCA5 (den Hollander et al., 2007) and SPATA7 (Wang et al., 2009), with the involvement of TULP1 (Hagstrom et al., 1998) and LRAT (Thompson et al., 2001) under debate. Mutations in these genes account for ~70% of all LCA cases. Several of them are also implicated in other retinal dystrophies: CRB1, RPE65, RDH12 and SPATA7 are associated with both LCA and early-onset retinal dystrophy (EORD), which often overlap (Gu et al., 1997; den Hollander et al., 1999; Janecke et al., 2004; Wang et al., 2009).
Several subtypes of LCA can be considered part of the ciliopathies, as four disease genes – TULP1, RPGRIP1, CEP290 and LCA5 – encode ciliary proteins. Since cilia are present throughout the whole body, mutations in ciliary genes may cause a broad phenotypic spectrum. One of the best examples is CEP290, the most frequently mutated gene in the western European LCA population. In addition to LCA, CEP290 is associated with Joubert syndrome (JS; MIM# 213300), Senior-Loken syndrome (SLS; MIM# 266900), Meckel-Grüber syndrome (MKS; MIM# 249000) and Bardet-Biedl syndrome (BBS; MIM# 209900); a range of clinically and genetically heterogeneous ciliopathies (Sayer et al., 2006; Valente et al., 2006b; Baala et al., 2007; Brancati et al., 2007; Helou et al., 2007; Leitch et al., 2008). Recent studies suggest that modifiers may play a role in the pathogenesis of ciliopathies (Leitch et al., 2008; Khanna et al., 2009; Louie et al., 2010).
Establishing a molecular diagnosis for LCA is not only important in the context of genetic counselling and clinical prognosis, but is also essential in view of future gene therapy. Recent Phase I clinical trials for RPE65 gene replacement therapy provide hopeful prospects for the treatment of inherited retinal dystrophies (Bainbridge et al., 2008; Hauswirth et al., 2008; Maguire et al., 2008; Cideciyan et al., 2009; Maguire et al., 2009). As such therapies are likely to be gene-specific, the development of robust clinical testing and efforts toward gene identification are of utmost importance.
Current diagnostic testing for LCA generally involves chip analysis that contains known mutations in all known LCA and EORD genes (Asper Ophthalmics, Estonia). Depending on the population, causal mutations are found in approximately 55% of all cases (Yzer et al., 2006). Only a limited number of laboratories subsequently screen an additional number of genes (Stone, 2007; den Hollander et al., 2008).
This study includes an extensive genetic survey in order to identify the molecular cause in 91 LCA probands mainly of Belgian origin, using LCA chip analysis for 8 to 13 genes and subsequent sequencing of the following genes: CEP290 (MIM# 610142), CRB1 (MIM# 604210), RPE65 (MIM# 180069), GUCY2D (MIM# 600179), AIPL1 (MIM# 604392) and CRX (MIM# 602225). In addition, exhaustive phenotyping was performed in all patients carrying mutation(s), and the AHI1 gene was screened for modifier alleles of CEP290-related disease.
MATERIALS AND METHODS
Patients
Ninety-one consenting subjects initially diagnosed with LCA were referred for molecular testing by an ophthalmologist and/or geneticist, mainly associated with the University Hospitals of Ghent, Leuven or Brussels. Eleven probands are born from a consanguineous marriage. The inclusion criteria for LCA were bilateral visual loss before the age of 6 months accompanied by nystagmus and an undetectable or significantly reduced ERG. Twelve patients presented with additional mental retardation and/or autistic behaviour. For 18 patients with an available Magnetic Resonance Imaging (MRI), the absence of a molar tooth sign (MTS) excluded the diagnosis of
JS. In addition, genotyping was performed on 11 probands with EORD (disease diagnosed beyond the first six months of life but before the age of three) and 13 with a retinal dystrophy in the context of JS (LCA-JS), SLS or cerebello-oculo-renal syndrome (CORS). These patients were not included during calculations of gene-specific contributions in isolated LCA. Genomic DNA and RNA were extracted from leukocytes using the Puregene DNA isolation kit (Gentra) and the RNeasy Mini kit (Qiagen) respectively, followed by cDNA synthesis with the iScript cDNA Synthesis kit (Bio-Rad). If available, parents and/or siblings were also genotyped. Seven of the patients were reported previously (Yzer et al., 2006; Brancati et al., 2007; Perrault et al., 2007). Patient notation was performed according to their clinical diagnosis (prefixes LCA, SLS, LCA-JS, CORS and EORD), with consecutive numbering in the order of the genes involved.
Genotyping
As a pre-screening method, all patients with either isolated LCA or EORD were analysed with a microarray containing 344 to 641 mutations in 8 (GUCY2D, CRX, RPE65, CRB1, RPGRIP1, AIPL1, LRAT and MERTK) to 13 (addition of TULP1, LCA5, RDH12, CEP290 and SPATA7) LCA and EORD genes (LCA chip Versions 2004-2009; Asper Ophthalmics, Estonia) (http://www.asperbio.com) (Zernant et al., 2005). Each of the mutations found by the LCA chip was subsequently confirmed through Sanger sequencing. In case of a heterozygous mutation, the coding exons and intron-exon boundaries of the involved gene were sequenced.
Patients in whom no mutations were identified after LCA chip analysis were analysed through sequencing of all coding exons and intron-exon boundaries of CEP290, CRB1, RPE65, GUCY2D, AIPL1 and CRX, the first five genes being the most frequently mutated in LCA. At the time the LCA chip did not yet include CEP290 variants, stepwise targeted mutation analysis was performed prior to sequencing of the total coding region. We initially screened for the frequent c.2991+1655A>G mutation followed by four additional mutations: c.4723A>T (p.Lys1575X), c.5587-1G>C (splice site), c.5163del (p.Thr1722GlnfsX2) and c.3310-1_3310delinsAA (splice site). The first three mutations occurred multiple times in a previous study (Perrault et al., 2007); the latter was found in three patients with a heterozygous c.2991+1655A>G mutation in our population. CEP290 was also screened at cDNA level in patients with only a single mutation in CEP290. To this end, cDNA screening using 16 overlapping primer sets was optimized. Four patients with CEP290-related LCA who presented with mental retardation, two patients with SLS, one patient with CORS and one patient with LCA-JS underwent sequencing of the AHI1 gene. For 13 patients with SLS/LCA-JS/CORS, molecular testing of CEP290 was requested. Supp. Table S1 includes all primer sequences used in this study.
Mutation nomenclature
Mutation nomenclature uses numbering with the A of the initiation codon ATG as +1 (www.hgvs.org/mutnomen), based on the following RefSeqs: NM_201253.1 (CRB1), NM_000329.2 (RPE65), NM_000180.3 (GUCY2D), NM_014336.3 (AIPL1), NM_000554.4 (CRX), NM_025114.3 (CEP290), NM_152443.2 (RDH12), NM_020366.3 (RPGRIP1) and NM_001134831.1 (AHI1) (http://www.ncbi.nlm.nih.gov/nuccore). All mutations and variants found in CEP290 were submitted to the locus-specific mutation database CEP290base (http://medgen.ugent.be/cep290base) (Coppieters et al., 2010).
Evaluation of sequence changes
The presence of all mutations was confirmed on a second PCR product. Segregation analysis of disease alleles was performed if possible. Genomic DNA obtained from > 340 unrelated ethnically matched healthy individuals was used as a control panel. Thorough bio-informatic evaluation of novel variants was done using Alamut software (v.1.5). Variants were designated as “unclassified variant (UV)” if no consensus was seen in all prediction programs used. The Alamut output for missense changes is listed in Supp. Table S2.
Clinical evaluation of patients
After identification of the molecular cause, clinical records were revisited, based on a clinical checklist comprising data on visual function, retinal appearance and associated (extra-) ocular features. When possible, ERG, fundus pictures, autofluorescence (AF) images and optical coherence tomography (OCT) were obtained. In case of CEP290-related LCA, neurological (MRI) and nephrological data (kidney ultrasound [US], urinary and blood parameters) were evaluated.
RESULTS
Mutation screening strategy of known LCA genes
As a first step, 102 probands were subjected to LCA chip analysis (91 LCA and 11 EORD). In total, 30 sequence changes assigned as mutations by Asper Ophthalmics were identified in 47 individuals. Homozygous and compound heterozygous variants in one gene were each found in 13 patients; a single heterozygous variant was identified in 17 individuals. In addition, variants within two distinct genes were found in four patients. The zygosity of p.Glu1330X (CRB1) could not be determined in LCA-36. Confirmation of each mutation through direct sequencing identified two inconsistencies. At first, LCA-58 was genotyped heterozygously for the AIPL1 mutation p.Trp278X by chip, while she was in fact homozygous. Secondly, a heterozygous p.Arg38AlafsX3 mutation in AIPL1 (LCA chip version 2006) could not be confirmed in LCA-23. Instead, a heterozygous c.111C>T (p.=) variant was identified on the same nucleotide position. This miscall has previously been described (Henderson et al., 2007). In addition, subsequent sequencing of GUCY2D in LCA-51 revealed a heterozygous c.389del mutation that was not detected on the LCA chip. The variants c.2101C>T (p.Pro701Ser) (GUCY2D), c.3341A>G (p.Asp1114Gly) (RPGRIP1) (Vallespin et al., 2007a), c.286G>A p.Val96Ile (AIPL1) (Yzer et al., 2006) and c.1301C>T (p.Ala434Val) (RPE65) (Morimura et al., 1998) have already been reported as polymorphisms and were therefore discarded as mutations. Moreover, identification of the GUCY2D p.Pro701Ser variant in a homozygous state in both healthy parents from an LCA patient further supported its non-pathogenic nature. After the exclusion of these polymorphisms, variants were assigned to be mutations in 45 patients (39 LCA and 6 EORD).
Secondly, all patients with a heterozygous mutation identified through chip analysis were subjected to screening of the relevant gene. In addition, all patients with negative chip results underwent sequencing of 6 LCA genes. In the following sections, the molecular results are discussed in detail for each of the genes.
CEP290
CEP290 was found to be the most frequently mutated gene in our cohort, accounting for 30% (27/91) of cases with isolated LCA (Table 1). Since the LCA chip did not contain CEP290 variants at the onset of this study, only a fraction of currently known mutations were detected using this technique. The c.2991+1655A>G, c.4723A>T (p.Lys1575X) and c.3310-1_3310delinsAA mutations were the most recurrent, with gene-specific allele frequencies of 49%, 11% and 6%, respectively. Similar to previous studies, most of the mutations are either nonsense, frameshift or splice site mutations. Only two missense variants were identified, of which the pathogenic effect is currently uncertain (p.Ala1566Pro and p.Leu1694Pro) (Supp. Table S2). Overall, 13 novel CEP290 mutations were identified to cause LCA. The complex allele c.3310-1_3310delinsAA has a predicted effect on splicing, which was confirmed by cDNA analysis (data not shown). The silent c.1824G>A change affects the last nucleotide of exon 18 and was also predicted to alter splicing (data not shown).
Since CEP290 mutations may cause a phenotypic spectrum ranging from isolated LCA to more complex disorders, we analysed 13 additional probands suffering from LCA-JS, SLS or CORS. CEP290 harbored mutations in seven of them (Table 1). Six probands carried known mutations, whereas a novel p.Thr2457AlafsX27 mutation segregated in family LCA-JS-2.
Sequencing of the entire coding region did not reveal a second mutation in LCA-27, while the pathogenic effect of one variant was uncertain in LCA-25 and LCA-26. Subsequent cDNA screening in LCA-25 and LCA-27 was normal, thereby making deep intronic splice site mutations or large exon deletions/duplications very unlikely. No RNA was available for LCA-26.
CRB1
Mutations in CRB1 were found in 15 families with LCA (16%) and 5 families with EORD (Table 1). The LCA chip allowed the identification of a homozygous or compound heterozygous CRB1 mutation in 15 probands (12 LCA and 3 EORD), and a heterozygous CRB1 mutation in 3 patients. Sequencing of the whole coding region of CRB1 in the latter revealed a known and novel mutation, respectively (LCA-29, p.Gln362X and LCA-34, c.4006-1G>T), and a novel unclassified variant (EORD-3, p.Asp491Val) on the second allele (Supp. Table S2).
Sequencing of the total coding region identified compound heterozygous mutations in two additional probands. In one of them, CRB1 screening was exceptionally performed without prior LCA chip analysis, given clear clinical indications for a CRB1-related phenotype (EORD-2). Indeed, this patient was compound heterozygous for the known p.Cys948Tyr mutation, and the novel p.Cys310Tyr variant, which is predicted to disrupt a disulfide bridge (Supp. Table S2). In addition, two sisters with LCA carried two novel frameshift mutations (LCA-41). As previously described, the p.Lys801X and p.Cys948Tyr mutations were most frequent, showing gene-specific allele frequencies in the LCA cohort of 27% and 23%, respectively.
RPE65
Eight cases with LCA showed mutations in RPE65 (9%) (Table 1). LCA chip analysis identified a homozygous RPE65 mutation in LCA-43 and LCA-47 and two compound heterozygous mutations in LCA-49. In addition, a heterozygous mutation was detected in four patients through LCA chip analysis. Sequencing of RPE65 in these individuals identified three novel mutations and the known p.Phe530LeufsX40 mutation which was not yet present on the LCA chip at the time (LCA-48). Although the evidence for a pathogenic nature of the novel mutation p.Trp331dup is not conclusive, segregation in patient LCA45a, her affected aunt LCA45b and her (healthy) parents sustains a causal role. The two other novel mutations result in a frameshift (LCA-46 and LCA-50). Following sequencing of the total coding region, one additional proband was found to be homozygous for the novel RPE65 mutation p.Pro181Leu (LCA-44) (Supp. Table S2). Interestingly, segregation analysis of the mutations found in LCA-46 could only confirm segregation of p.Leu341Ser in the mother, suggesting that p.Ser121LeufsX6 occurred de novo (paternity confirmed).
GUCY2D
Mutations in GUCY2D were found in seven probands with LCA (8%) (Table 1). Five of them were identified with GUCY2D mutations using the LCA chip. One was homozygous for p.Phe565Ser (LCA-52), while three others carried p.Arg768Trp. One of the latter was homozygous (LCA-53); the other two were compound heterozygous for p.Phe565Ser (LCA-54) and the novel missense change p.Lys866Asn (LCA-55), respectively (Supp. Table S2). In addition, LCA-57 was heterozygous for the p.Pro575Leu variant that was previously identified in the mother of an LCA patient (Koenekoop et al., 2002). However, no second mutation was found. LCA-56 was compound heterozygous for the novel missense changes p.Glu196Val and p.Pro711Leu (Supp. Table S2). In addition, a novel splice site mutation was identified in LCA-51 (c.2577-2A>C).
AIPL1
Only two distinct AIPL1 variants were detected through LCA chip analysis in five LCA patients (5%) (Table 1). The p.Trp278X mutation occurred homozygously in four probands (LCA-58 to LCA-61). In addition, a heterozygous p.Thr114Ile variant was found in proband LCA-62. Direct sequencing of the AIPL1 gene identified the known variant p.Pro376Ser (missing signal on LCA chip). Segregation analysis in the parents, however, revealed a cis-allelic inheritance from the mother. No further mutations were detected following additional sequencing of AIPL1 in other patients.
CRX
LCA chip analysis identified 2 CRX missense variants in two LCA patients (Table 1). The p.Tyr142Cys variant was previously described as a mutation (Vallespin et al., 2007a). Stone and colleagues, however, considered this variant as a polymorphism based on the estimate of pathogenic probability and the identification of this variant in a patient with two disease-causing alleles in another LCA gene (Stone, 2007) (LCA-63). The pathogenicity of the second variant p.Val242Met also remains unclear (LCA-64) (Swain et al., 1997; Rivolta et al., 2001; Chen et al., 2002). Given their uncertain pathogenic potential, both variants were discarded as mutations for further calculations.
RDH12
Mutation screening of RDH12 was performed downstream of LCA chip results involving this gene. A heterozygous p.Ala269GlyfsX2 mutation was identified in two probands with EORD. Subsequent sequencing of RDH12 identified an additional missense change in both patients, p.Val233Asp (EORD-6) and p.Ser175Leu (EORD-7) respectively (Supp. Table S2). According to UniProt, the Ser175 residue might be a substrate binding site (http://www.uniprot.org/uniprot/Q96NR8). A known mutation located in the same codon, p.Ser175Pro, lacks the ability to catalyze the reduction of retinaldehyde to retinol in vitro (Lee et al., 2007). In addition, a homozygous p.Ala269GlyfsX2 mutation was identified in patient EORD-8, for which RDH12 sequencing was performed prior to LCA-chip analysis (upon request).
RPGRIP1
Similarly, screening of RPGRIP1 was performed in the context of LCA chip analysis. In one LCA patient, a heterozygous mutation was identified. Sequencing of RPGRIP1, however, did not identify a second mutation (LCA-65).
Mutations in multiple LCA genes
For the assessment of the potential involvement of a second gene in LCA, only variants with significant pathogenic potential were taken into account (see above). Two patients with CEP290-related LCA displayed a heterozygous mutation in another LCA gene: LCA-16 was heterozygous for the known p.Arg85Cys mutation in RPE65, while LCA-20 carried the common p.Lys801X mutation in CRB1.
Identification of potential modifier alleles in the AHI1 gene
The AHI1 gene was sequenced as a candidate modifier gene in eight patients with CEP290-related LCA who presented with mental retardation. Four of them were diagnosed with LCA (3, 20, 23 and LCA-24); two patients also suffered from NPHP (SLS-2 and SLS-3) and in two other cases, the LCA phenotype was part of a JS diagnosis (CORS-1 and LCA-JS-3). A MTS was absent on brain imaging in two isolated patients with LCA (no data were available for LCA-20, LCA-24, SLS-2 and SLS-3).
A heterozygous novel AHI1 p.Asn811Lys mutation was found in the most severely affected patient CORS-1, out of three patients with the same CEP290 genotype but different neurological involvement (SLS-2, SLS-3 and CORS-1). Moreover, AHI1 screening in the five remaining patients revealed a second heterozygous missense variant, p.His758Pro, in LCA-3. Conservation and in silico predictions for both changes suggest a possible effect on protein structure/function (Supp. Table S2). Interestingly, exonic splicing enhancer (ESE) predictions point to a change in ESEs for both variants (data not shown). Moreover, both changes are located in a conserved WD-40 repeat (http://www.uniprot.org/uniprot/Q8N157) and were absent in > 340 Belgian control individuals.
In addition, SLS-2 was found to be heterozygous for the known p.Ser1123Phe change. Although it concerns a potentially pathogenic variant that affects a phosphorylation site and is located in a highly conserved region (Dephoure et al., 2008), this change was considered a polymorphism because of its frequency in the Dutch population and the observation that it did not segregate in a family with JS (Valente et al., 2006a; Kroes et al., 2008).
Table 1. Mutations identified in 80 unrelated patients with LCA/EORD, using LCA chip analysis and direct sequencing of CEP290, CRB1, RPE65, AIPL1, GUCY2D and CRX
Patient Origin Par cons Segr
Allele 1 Allele 2 Reference
Intron/ exon Nucleotide change Amino acid change Intron/ exon Nucleotide change Amino acid change CEP290
LCA-1 Belgium - X I26 c.2991+1655A>G* p.Cys998X* I26 c.2991+1655A>G* p.Cys998X* (den Hollander et al.,
2006)
LCA-2 Belgium - X I26 c.2991+1655A>G p.Cys998X I26 c.2991+1655A>G p.Cys998X (den Hollander et al.,
2006)
LCA-3+ Belgium - X I26 c.2991+1655A>G* p.Cys998X* E6 c.322C>T p.Arg108X (den Hollander et al.,
2006)
LCA-4 Belgium - NA I26 c.2991+1655A>G p.Cys998X E25 c.2695C>T p.Gln899X (den Hollander et al., 2006)
LCA-5 Belgium - NA I26 c.2991+1655A>G p.Cys998X E34 c.4393C>T p.Arg1465X
(den Hollander et al., 2006), (Brancati et al., 2007) (CORS)
LCA-6 Belgium - X I26 c.2991+1655A>G p.Cys998X E36 c.4723A>T p.Lys1575X
(den Hollander et al., 2006), (Brancati et al., 2007; Perrault et al., 2007)
LCA-7F Belgium - NA I26 c.2991+1655A>G p.Cys998X E36 c.4723A>T p.Lys1575X
(den Hollander et al., 2006), (Brancati et al., 2007; Perrault et al., 2007)
LCA-8 Belgium - NA I26 c.2991+1655A>G p.Cys998X E36 c.4723A>T p.Lys1575X
(den Hollander et al., 2006), (Brancati et al., 2007; Perrault et al., 2007)
LCA-9 Belgium - X I26 c.2991+1655A>G p.Cys998X E39 c.5344C>T p.Arg1782X (den Hollander et al., 2006)
LCA-10 Lithuania - X I26 c.2991+1655A>G p.Cys998X E6 c.384_385del p.Asp128GlufsX17 (den Hollander et al., 2006)
LCA-11 Belgium - X I26 c.2991+1655A>G p.Cys998X E6 c.437del p.Glu146GlyfsX17 (den Hollander et al.,
2006) LCA-12
The Netherlan ds
- NA I26 c.2991+1655A>G* p.Cys998X* E19 c.1859_1862del p.Arg621IlefsX2
(den Hollander et al., 2006), (Perrault et al., 2007)
LCA-13 Belgium - NA I26 c.2991+1655A>G p.Cys998X E29 c.3422dup p.Leu1141PhefsX5 (den Hollander et al., 2006)
LCA-14 Belgium/
Morocco - X I26 c.2991+1655A>G* p.Cys998X* E31 c.4001del p.Thr1334IlefsX2
(den Hollander et al., 2006)
LCA-15 (Perrault et al., 2007)
Belgium - NA I26 c.2991+1655A>G p.Cys998X E37 c.4962_4963del p.Glu1656AsnfsX3
(den Hollander et al., 2006), (Perrault et al., 2007)
LCA-16+ Belgium/
Greece - X I26 c.2991+1655A>G p.Cys998X E40 c.5493del p.Ala1832ProfsX19
(den Hollander et al., 2006), (Brancati et al., 2007; Frank et al., 2008) (CORS)
LCA-17 Belgium - X I26 c.2991+1655A>G p.Cys998X E40 c.5519_5537del p.Lys1840ArgfsX5 (den Hollander et al., 2006)
LCA-18 Belgium - X I26 c.2991+1655A>G p.Cys998X E43 c.5865_5867delins
GG p.Glu1956GlyfsX9
(den Hollander et al., 2006)
LCA-19 Belgium - X I26 c.2991+1655A>G p.Cys998X I13 c.1189+1G>A Splice defect (den Hollander et al.,
2006) LCA-20+
(Yzer et al., 2006)
Belgium - NA I26 c.2991+1655A>G p.Cys998X I21 c.2218-2A>C Splice defect (den Hollander et al., 2006)
LCA-21 Belgium - X I26 c.2991+1655A>G p.Cys998X I28-E29
c.3310-1_3310delinsAA Splice defect
(den Hollander et al., 2006)
LCA-22 Belgium - NA I26 c.2991+1655A>G p.Cys998X I28-E29
c.3310-1_3310delinsAA Splice defect
(den Hollander et al., 2006)
LCA-23 Belgium - NA I26 c.2991+1655A>G p.Cys998X I28-E29
c.3310-1_3310delinsAA Splice defect
(den Hollander et al., 2006)
Patient Origin Par cons Segr
Allele 1 Allele 2 Reference
Intron/ exon Nucleotide change Amino acid change Intron/ exon Nucleotide change Amino acid change
LCA-25F Belgium - X E36 c.4723A>T p.Lys1575X E35 c.4696G>C p.Ala1566Pro UV (Perrault et al., 2007)
LCA-26 Belgium X E18 c.1824G>A p.=, splice site E38 c.5081T>C p.Leu1694Pro UV
LCA-27 Belgium - NA I26 c.2991+1655A>G p.Cys998X ? ? ? (den Hollander et al.,
2006)
SLS-1 Pakistan FC X E2 c.21G>T p.Trp7Cys E2 c.21G>T p.Trp7Cys (Valente et al., 2006b)
(CORS)
SLS-2 Belgium - NA E36 c.4723A>T p.Lys1575X E34 c.4393C>T p.Arg1465X
(Perrault et al., 2007), (Brancati et al., 2007) (CORS)
SLS-3 Belgium - NA E36 c.4723A>T p.Lys1575X E34 c.4393C>T p.Arg1465X
(Perrault et al., 2007), (Brancati et al., 2007) (CORS) CORS-1+ (Brancati et al., 2007)
Belgium SD NA E36 c.4723A>T p.Lys1575X E34 c.4393C>T p.Arg1465X
(Perrault et al., 2007), (Brancati et al., 2007) (CORS)
LCA-JS-1 Belgium - X I40 c.5587-1G>C Splice defect E31 c.3793C>T p.Gln1265X (Perrault 2007), (Baala et al., 2007) (ML) LCA-JS-2
II-1
ND + X
E54 c.7366_7369del p.Thr2457AlafsX27 E54 c.7366_7369del p.Thr2457AlafsX27 LCA-JS-2
II-2 E54 c.7366_7369del p.Thr2457AlafsX27 E54 c.7366_7369del p.Thr2457AlafsX27
LCA-JS-3 Belgium - NA I28-E29
c.3310-1_3310delinsAA Splice defect E54 c.7341dup p.Leu2448ThrfsX8 (Sayer et al., 2006)
CRB1
LCA-28 Belgium - NA E7 c.2401A>T* p.Lys801X* E7 c.2401A>T* p.Lys801X* (den Hollander et al.,
2001) LCA-29
(Yzer et al., 2006)
Belgium - NA E7 c.2401A>T* p.Lys801X* E5 c.1084C>T p.Gln362X
(den Hollander et al., 2001), (Yzer et al., 2006)
LCA-30
Belgium - X E7 c.2401A>T* p.Lys801X* E7 c.2290C>T* p.Arg764Cys*
(den Hollander et al., 2001), (Lotery et al., 2001)
LCA-31 (Yzer et al., 2006)
Belgium - X E7 c.2401A>T* p.Lys801X* E8 c.2688T>A* p.Cys896X*
(den Hollander et al., 2001), (Hanein et al., 2004)
LCA-32 (Yzer et al., 2006)
Belgium - NA E7 c.2401A>T* p.Lys801X* E8 c.2688T>A* p.Cys896X*
(den Hollander et al., 2001), (Hanein et al., 2004)
LCA-33 Belgium - NA E7 c.2401A>T* p.Lys801X* E9 c.2843G>A* p.Cys948Tyr*
(den Hollander et al., 2001), (Lotery et al., 2001)
LCA-34 Belgium - X E7 c.2401A>T* p.Lys801X* I11 c.4006-1G>T Splice defect (den Hollander et al., 2001)
LCA-35 Belgium + NA E9 c.2843G>A* p.Cys948Tyr* E9 c.2843G>A* p.Cys948Tyr* (Lotery et al., 2001)
LCA-36 ND - NA E9 c.2843G>A* p.Cys948Tyr* E11 c.3988G>T* p.Glu1330X* (Lotery et al., 2001),
(LCA chip)
LCA-37 Belgium - X E9 c.2843G>A* p.Cys948Tyr* I8 c.2842+5G>A* Splice defect*
(Lotery et al., 2001), (den Hollander et al., 1999)
LCA-38 Belgium - X E9 c.2843G>A* p.Cys948Tyr* I8 c.2842+5G>A* Splice defect*
(Lotery et al., 2001), (den Hollander et al., 1999)
LCA-39a
Belgium - X
E9 c.2843G>A* p.Cys948Tyr* I8 c.2842+5G>A* Splice defect*
(Lotery et al., 2001), (den Hollander et al., 1999)
LCA-39b I11 c.4005+1G>A Splice defect I8 c.2842+5G>A Splice defect
(Hanein et al., 2004), (den Hollander et al., 1999)
LCA-40 Belgium - NA I11 c.4005+1G>A* Splice defect* I8 c.2842+5G>A* Splice defect*
(Hanein et al., 2004), (den Hollander et al., 1999)
Patient Origin Par cons Segr
Allele 1 Allele 2 Reference
Intron/ exon Nucleotide change Amino acid change Intron/ exon Nucleotide change Amino acid change LCA-41 II-1
Belgium - X E7 c.2441_2442del p.Leu814ArgfsX23 E9 c.3713_3716dup p.Cys1240ProfsX24
LCA-41 II-2 E7 c.2441_2442del p.Leu814ArgfsX23 E9 c.3713_3716dup p.Cys1240ProfsX24
LCA-42 ND + NA E11 c.3879G>A* p.Trp1293X* E11 c.3879G>A* p.Trp1293X* (Hanein et al., 2004)
EORD-1 II-1
Belgium - X
E9 c.2843G>A* p.Cys948Tyr* E7 c.2401A>T* p.Lys801X*
(Lotery et al., 2001), (den Hollander et al., 2001)
EORD-1 II-2 E9 c.2843G>A p.Cys948Tyr E7 c.2401A>T p.Lys801X
(Lotery et al., 2001), (den Hollander et al., 2001)
EORD-2 Belgium NA E9 c.2843G>A p.Cys948Tyr E4 c.929G>A p.Cys310Tyr (Lotery et al., 2001)
EORD-3 Belgium - X E9 c.2843G>A* p.Cys948Tyr* E6 c.1472A>T p.Asp491Val UV (Lotery et al., 2001)
EORD-4 Belgium NA E5 c.1084C>T* p.Gln362X* E5 c.1084C>T* p.Gln362X* (Yzer et al., 2006)
EORD-5 Belgium - NA E7 c.2290C>T* p.Arg764Cys* E7 c.2290C>T* p.Arg764Cys* (Lotery et al., 2001)
RPE65
LCA-43 Turkey FC X E3 c.131G>A* p.Arg44Gln* E3 c.131G>A* p.Arg44Gln* (Simovich et al., 2001)
LCA-44 Turkey FC X E6 c.542C>T p.Pro181Leu E6 c.542C>T p.Pro181Leu
LCA-45a
Belgium - X E7 c.700C>T* p.Arg234X* E9 c.991_993dup p.Trp331dup (Marlhens et al., 1997)
LCA-45b E9 c.991_993dup p.Trp331dup E9 c.991_993dup p.Trp331dup
LCA-46 Portugal
X (c.10 22T> C)
E10 c.1022T>C* p.Leu341Ser* E5 c.361delT p.Ser121LeufsX6
de novo
(Morimura et al., 1998) (ARRP)
LCA-47 Belgium - X E14 c.1590del* p.Phe530LeufsX40* E14 c.1590del* p.Phe530LeufsX40* (Yzer et al., 2006)
LCA-48 Belgium - X E14 c.1590del p.Phe530LeufsX40 E5 c.370C>T* p.Arg124X*
(Yzer et al., 2006), (Morimura et al., 1998)
LCA-49 Belgium - X E14 c.1590del* p.Phe530LeufsX40* I1 c.11+5G>A* Splice defect* (Yzer et al., 2006),
(Gu et al., 1997) LCA-50
Belgium /Russia (mother)
- X E9 c.886dupA p.Arg296LysfsX7 I1 c.11+5G>A* Splice defect* (Gu et al., 1997)
GUCY2D
LCA-51 Morocco/
Belgium - NA E2 c.389del p.Pro130LeufsX36 I13 c.2577-2A>C Splice defect (Perrault et al., 1996)
LCA-52 Turkey TC NA E8 c.1694T>C* p.Phe565Ser* E8 c.1694T>C* p.Phe565Ser* (Perrault et al., 1996)
LCA-53 Belgium NA E12 c.2302C>T* p.Arg768Trp* E12 c.2302C>T* p.Arg768Trp* (Lotery et al., 2000)
LCA-54 Morocco/
Belgium - X E12 c.2302C>T* p.Arg768Trp* E8 c.1694T>C* p.Phe565Ser*
(Lotery et al., 2000), (Perrault et al., 1996)
LCA-55 Belgium - X E12 c.2302C>T* p.Arg768Trp* E14 c.2598G>C p.Lys866Asn (Lotery et al., 2000)
LCA-56 Belgium/
France - X E2 c.587A>T p.Glu196Val UV E11 c.2132C>T p.Pro711Leu UV
LCA-57 Africa - NA E8 c.1724C>T* p.Pro575Leu* UV ? ? ? (Koenekoop et al.,
2002)
AIPL1
LCA-58 (Yzer et al., 2006)
Belgium - X E6 c.834G>A* p.Trp278X* E6 c.834G>A p.Trp278X (Sohocki et al., 2000a)
LCA-59 Belgium - NA E6 c.834G>A* p.Trp278X* E6 c.834G>A* p.Trp278X* (Sohocki et al., 2000a)
LCA-60 Belgium NA E6 c.834G>A* p.Trp278X* E6 c.834G>A* p.Trp278X* (Sohocki et al., 2000a)
LCA-61 Belgium - X E6 c.834G>A* p.Trp278X* E6 c.834G>A* p.Trp278X* (Sohocki et al., 2000a)
LCA-62 Africa - in cis E3
E6
c.341C>T* c.1126C>T
p.Thr114Ile* UV
p.Pro376Ser UV ? ? ? (Sohocki et al., 2000b)
CRX
LCA-63 Belgium SC NA E4 c.425A>G* p.Tyr142Cys* UV ? ? ? (Vallespin et al.,
Patient Origin Par cons Segr
Allele 1 Allele 2 Reference
Intron/ exon Nucleotide change Amino acid change Intron/ exon Nucleotide change Amino acid change
LCA-64 Ruanda NA E3 c.724G>A* p.Val242Met* UV ? ? ? (Swain et al., 1997)
RDH12
EORD-6 Belgium - X E6 c.806_810del* p.Ala269GlyfsX2* E8 c.698T>A p.Val233Asp
(Janecke et al., 2004), https://www.carverlab. org/carver-mutation-database
EORD-7 Belgium + X E6 c.806_810del* p.Ala269GlyfsX2* E7 c.524C>T p.Ser175Leu (Janecke et al., 2004)
EORD-8 Belgium E6 c.806_810del p.Ala269GlyfsX2 E6 c.806_810del p.Ala269GlyfsX2 (Janecke et al., 2004)
RPGRIP1
LCA-65 Belgium E16 c.2668C>T* p.Arg890X* ? ? ? (Gerber et al., 2001)
Novel mutations are indicated in bold. +: patients carrying a heterozygous mutation in an additional gene: LCA-3 (AHI1, c.2273A>C, p.His758Pro), LCA-16 (RPE65, c.253C>T, p.Arg85Cys) (Stone, 2007), LCA-20 (CRB1, c.2401A>T, p.Lys801X) (den Hollander et al., 2001) and CORS-1 (AHI1, c.2433T>G, p.Asn811Lys). *: identified through LCA chip analysis. F: LCA-7 and LCA-25 are distantly related. X: segregation analysis performed and segregation confirmed. NA: no material available of family members. Reference: first publication describing the mutation in patients with LCA or EORD. In case of CEP290, these references may also refer to papers dealing with other phenotypes (phenotype mentioned between brackets). Seven patients were already described (corresponding reference is indicated in the first column). Abbreviations used: par cons: parental consanguinity; segr: segregation; FC: first cousins; SC: second cousins; TC: third cousins; SD: second degree; ND: no data; UV, unclassified variant; LCA, Leber Congenital Amaurosis; SLS, Senior-Loken syndrome; JS, joubert syndrome; ARRP, autosomal recessive retinitis pigmentosa; CORS, cerebello-oculo-renal syndrome; ML, Meckel-like syndrome.
Clinical findings
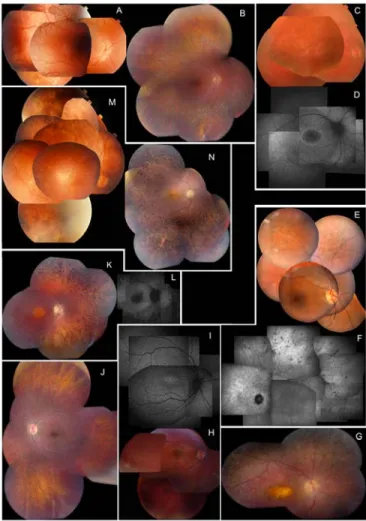
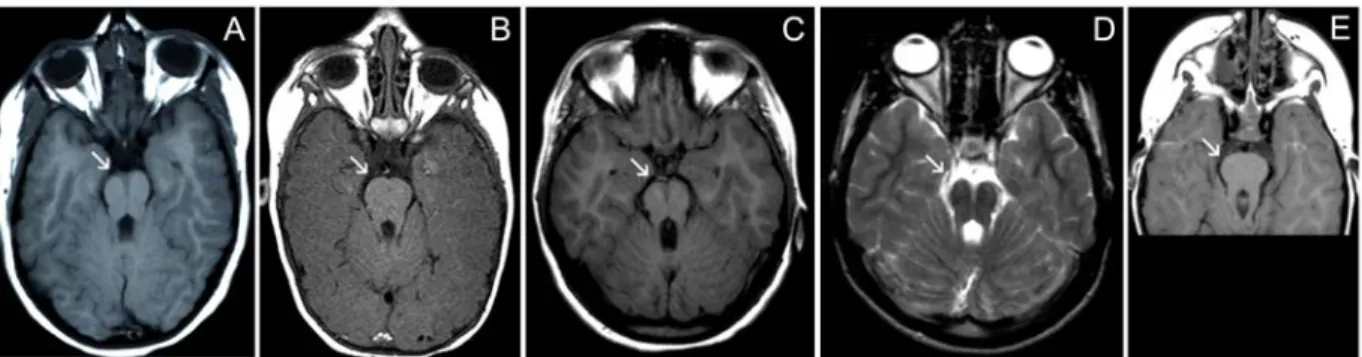
Extensive ophthalmological data (best corrected visual acuity [BCVA], refraction, ERG, visual fields, color vision testing, fundus aspect both with white light and autofluorescence imaging and the presence of nystagmus, night blindness, photophobia and additional features) as well as associated manifestations in patients with CRB1, RPE65, GUCY2D, AIPL1, CRX, RPGRIP1 and RDH12 mutations are summarized in detail in Supp. Table S3. In addition, Figure 1 depicts several representative fundi from patients with an established molecular diagnosis. An MTS due to midbrain abnormalities with cerebellar vermis aplasia was demonstrated in five patients with JS-LCA/CORS and CEP290 mutations (Figure 2).
Special attention was paid to the CEP290-related ocular phenotype, since this has been described in only a few LCA studies so far. It appeared that this phenotype displays only limited fundus alterations in the first few years of life. In a small subset of patients, no fundus abnormalities were obvious early on, while in the majority a marbleized fundus and/or salt and pepper aspect was seen during the first decade. This aspect further evolved from young adulthood into progressive outer retinal atrophy in the midperiphery with relative sparing of the central macula. Abnormalities of the central macula were absent in our patient cohort, despite the impression that CEP290-related disease is probably of a cone-rod type (based on ERG findings and the occurrence of photophobia). Of note is the presence of a hyperautofluorescent ring around the central macula on AF imaging, observed in four patients starting from the age of six (LCA-2, LCA-3, LCA-7 and LCA-25). Mild intraretinal spicular pigment migration occurred in three patients at an age between 7 and 33 years old (LCA-JS-1, LCA-6 and LCA-7). This aspect became even more pronounced at the age of 49 in patient LCA-7, where a predominant spicular pigmentation was mixed with less frequent intraretinal pigment migration with a nummular aspect. Visual acuity of this group was mostly limited to light perception. In the few patients with better preserved central vision, basic color vision was present and visual fields varied from severely concentrically constricted (LCA-8) to sparing of the central 30° at an age of 49 (LCA-7) (Supp. Table S3).
Figure 1. Clinical characteristics of eight LCA patients with an established molecular diagnosis, illustrating characteristic phenotypic features associated with different genotypes. CEP290. A & B: Early and later stage phenotype in right eye (RE) in LCA-3 at age 3 and 18 years respectively; note marbleized aspect of midperiphery at age 3, evolving towards atrophy later; macula stays well-preserved throughout evolution. C & D: Fundus and autofluorescence (AF) image of RE of LCA-25 at age eight years; note concentric hyperautofluorescent ring around macula suggesting a watershed zone between better and more affected retina with probably central area the better; midperipheral retina shows diffuse mottled hyperautofluorescence suggesting widespread outer retinal disease. E & F: Fundus image of RE and infrared image of left eye (LE) of LCA-7 at age 33 and 49 years respectively; note pigment epithelium alterations in the mid- and far periphery of retina but no intraretinal pigmentation, and with fair preservation of macular area at age 33; at age 49 macula is still fairly well-preserved, but outer retinal atrophy and spicular intraretinal pigmentation is now prominent. CRB1.G: Fundus of RE of LCA-39a at age 16, showing typical yellowish discoloration of atrophic macula, surrounded by nummular type of intraretinal pigmentation; mild pseudopapilledema and prepapillary paravascular fibrosis also visible, as is peripheral greyish hue of outer retinal atrophy with fine white flecks and nummular pigmentation. GUCY2D. H & I: Fundus and AF image of RE of LCA-55 at age 9 years; fundus is essentially quite normal with only mild pigment epithelium alterations in the retinal periphery; however, AF image shows hyperautofluorescence in central macular area. RPE65. J: Fundus of LE of LCA-49 at age 10 who subsequently underwent gene therapy with AAV2-hRPE65v2 in RE (Maguire et al. 2009); apart from some discrete pigment epithelium alterations fundus is essentially normal; autofluorescence imaging could not be obtained due to lack of lipofuscin accumulation in retinal pigment epithelium (RPE) typical of this type of LCA. AIPL1. K & L: Fundus and AF image of RE of LCA-61 at age 19 years; central macular atrophy with yellowish hue is surrounded by area of better preserved peripheral macula; outer retinal atrophy with spicular intraretinal pigmentation visible in periphery; AF shows black area of atrophic central macula, but is typically not surrounded by hyperautofluorescent ring. RDH12. M & N: Fundus of EORD-7 at age 5 and 19 years respectively; note mild macular RPE changes which become more prominent with age; mild predominantly spicular intraretinal pigmentation also increases with age; however, preservation of patches of normal peripheral retina are most striking feature; these patches remain over time.
Figure 2. Magnetic resonance imaging (MRI) showing characteristic molar tooth sign (MTS) in five patients with Joubert syndrome/cerebello-oculo-renal syndrome due to mutations in CEP290 (all are axial sections through midbrain). Images organized from left to right; arrows indicate MTS of midbrain present in all due to midbrain malformation with hypoplastic cerebellar vermis and midline cleft (all images are T1 weighted except for panel D which is T2 weighted). A) CORS-1 at age 5 years; B) LCA-JS-1 at age 7 years; C) LCA-JS-2 II-1 at age 14 years; D) LCA-JS-2 II-2 at age 17 years and E) LCA-JS-3 at age 2 years.
DISCUSSION
Genotypes of the Belgian LCA population
In a cohort of 91 unrelated LCA patients, mainly originating from Belgium, a total of 61 different mutations (including 9 UVs) were found in 7 genes. Homozygous or compound heterozygous mutations were detected in 65% (59/91) of probands, whereas only one heterozygous mutation could be identified in 4% (4/91) of probands. In addition to isolated LCA, this study also identified mutations in eight probands with EORD and seven probands with syndromic LCA (SLS, LCA-JS and CORS) (Table 1).
LCA chip analysis proved to be a powerful initial tool as mutations were found in 41% (37/91) of patients with LCA. Subsequent sequencing of 6 genes (CEP290, CRB1, RPE65, GUCY2D, AIPL1 and CRX) enabled us to identify mutations in an additional 28% of cases. Of note, the majority of mutations found in the latter probands are now included in the LCA chip, increasing its detection rate to 65% if our cohort would have been analyzed using the latest version (v8, 641 variants).
Segregation of disease alleles was demonstrated in 41 out of 43 families available, with two exceptions. The first one is LCA-62, in which the AIPL1 variants p.Thr114Ile and p.Pro376Ser were located in cis on the maternal chromosome. This finding challenges a previous study proposing this genotype as causal in an LCA patient (Sohocki et al., 2000b). In the second case, only one of two mutations identified in LCA-46 was found in the mother but not in father, suggesting that the other mutation arose de novo (RPE65, p.Ser121LeufsX6, non-paternity excluded) (Table 1). Of note, two different mutations in CEP290 were identified in CORS-1, originating from a consanguineous marriage, which illustrates that assuming homozygosity in offspring from a consanguinous mating can be a potential pitfall for the identification of the causal defect (Table 1).
Our data demonstrate a key role for CEP290 in the Belgian LCA population, as CEP290 mutations were identified in 27 probands (30%). Although the prevalence of CEP290 mutations is not this high worldwide (Simonelli et al., 2007; Vallespin et al., 2007b; Seong et al., 2008; Li et al., 2009; Sundaresan et al., 2009), this study corroborates the importance of CEP290 in the Northwestern European population (den Hollander et al., 2006; Perrault et al., 2007). The second most frequently mutated gene in our LCA population was CRB1 (16%), followed by RPE65 (9%), GUCY2D (8%) and AIPL1 (5%), which is in agreement with previous data (den Hollander et al., 2008). The RPGRIP1 gene – in this study only investigated with the LCA chip – accounted for less than 1% of the LCA population.
In total, 30 novel mutations/variants were identified in this study (Table 1, Supp. Table S2). Interestingly, 5 out of 16 novel mutations in CEP290 are located nearby known changes: c.384_385del (c.384_387del, c.381_382delinsT) (Baala et al., 2007; Perrault et al., 2007), 2A>C (4_2222del and c.2218-15_2220del) (Sayer et al., 2006; Stone, 2007), c.3310-1_3310delinsAA (c.3310-1G>C) (Tory et al., 2007),
c.5519_5537del (c.5515_5518del) (Sayer et al., 2006) and c.5865_5867delinsGG (c.5866G>T) (den Hollander et al., 2006). A similar observation was made for RDH12, in which the novel mutation p.Ser175Leu affects the same codon as the known mutation p.Ser175Pro (Perrault et al., 2004). Overall, these regions/codons might be more prone to mutational events.
For several genes, a limited number of recurrent mutations made up the majority of mutated alleles. This was certainly the case for CEP290, in which c.2991+1655A>G was found in 89% of all LCA patients with CEP290-related pathology. Together with p.Lys1575X and c.3310-1_3310delinsAA, a significant fraction of mutated alleles was identified in the LCA population (35/53 alleles). So far, p.Lys1575X has only been found in patients originating from northern France (Perrault et al., 2007) or Belgium (Brancati et al., 2007). This potential founder effect is supported by our study, since all patients who carry p.Lys1575X live in Flanders (northern part of Belgium). A similar regional prevalence was seen for p.Ala1832ProfsX19, which was inherited from the Greek father of LCA-16. The same mutation occurred in an Italian patient with CORS (Brancati et al., 2007) and in two consanguineous families of Kosovar-Albanian and Kosovar origin with MKS, sharing a common haplotype (Frank et al., 2008). For the other genes, the following mutations presented with a gene-specific allele-frequency of at least 20% in the LCA population: p.Lys801X (CRB1, 27%), p.Cys948Tyr (CRB1, 23%), p.Phe530LeufsX40 (RPE65, 25%), p.Arg768Trp (GUCY2D, 31%), p.Phe565Ser (GUCY2D, 23%) and p.Trp278X (AIPL1, 89%). The presence of all but one of these mutations on the LCA chip significantly contributed to its high detection rate.
In six LCA patients, only one mutation was found after sequencing of the gene apparently involved following chip testing. Notably, we might have failed to detect deep intronic and regulatory mutations or multi-exon deletions, as were recently demonstrated in CEP290 (Travaglini et al., 2009). In addition, it cannot be excluded that the phenotype is caused by mutations in a different gene, as was the case for LCA-20. Furthermore, new mutations in the other known genes cannot be ruled out here. Their contribution is expected to be limited however, taking into account the high detection rate obtained using the current strategy. Finally, these patients may carry mutations in as yet unknown genes.
Phenotypes of the Belgian LCA population
In addition, the phenotypes of patients with a molecular diagnosis were extensively studied. For all genes, nystagmus and hypermetropia were recurrent features. The oculodigital sign (plus enophthalmos) was often seen in all but RPE65-related LCA patients. A relatively higher incidence of both keratoconus and cataract was observed in the CRB1-related group, which may reflect secondary effects of a more severe retinal dystrophy compared to other genes. Indeed, several retinal abnormalities such as macular atrophy and intraretinal pigment migration already became apparent in the first decade of life in patients with CRB1-related disease, being earlier than generally seen for the other genes. In addition, a yellowish discoloration of the central macula was often observed. However, this feature is not entirely gene-specific, since it also occurred in a patient carrying RDH12 mutations (EORD-7). This feature may be due to more severe outer retinal atrophy in the macula in CRB1- and RDH12-related disease, which may cause more intense scleral light reflection due to less absorption by the atrophic retinal pigment epithelium, with consequent highlighting of the macular luteal pigment.
The RPE65-related phenotype proved to be typically associated with a fundus appearance which is essentially normal during first years and displays only later on fundus alterations which are initially mild. Visual acuity is generally somewhat better than that seen in CRB1-related LCA. However, it seems to be the relatively slow evolution of the phenotype which makes it particularly suitable for therapeutic intervention (Bainbridge et al., 2008; Hauswirth et al., 2008; Maguire et al., 2008). For two patients from our cohort (LCA-47 and LCA-49), RPE65 gene-replacement therapy resulted in better visual function (Maguire et al., 2009). In addition, two patients with RPE65 mutations reported a period of increased visual function, possibly reflecting postnatal physiological cone maturation (LCA-44 and LCA-47) (Koenekoop et al., 2007). Hanein et al. classified both CRB1- and RPE65-related LCA as rod-cone dystrophies because of a predominant occurrence of night blindness (Hanein et al., 2004). All RPE65-related phenotypes in this study correspond to this classification. In the CRB1-group, however, six patients with LCA and two with EORD also suffered from photophobia, even before the onset of night blindness in EORD-3.
Similarly, the GUCY2D-related phenotype was found to have only limited fundus abnormalities. Although this phenotype was previously categorized as cone-rod dystrophy, one patient in our cohort had severe night blindness before photophobia became apparent (LCA-54).
In the patients with AIPL1 mutations, an RP-like phenotype emerged by their teenage years at the latest, and a maculopathy with (partial) outer retinal atrophy was typically present in the majority of cases (Dharmaraj et al., 2004).
A unique feature of RDH12-related early-onset dystrophy was the occurrence of areas with complete preservation of the chorioretina in the retinal periphery, alternating with regions of total atrophy (EORD-7).
Notably, our study is one of the first reporting on the ocular phenotype of a larger group of LCA patients with CEP290 mutations (Supp. Table S3). In keeping with previously reported findings (Perrault et al., 2007), it appeared to be that of a severe cone-rod type retinal dystrophy. Visual acuity was mostly limited to light perception, as recently described (Walia et al., 2010). Interestingly, a limited subset of patients displayed no obvious retinal abnormalities in the first years of life. In general, the fundus contained either small white dots or, more frequently, a marbleized or salt and pepper aspect in the first to second decade. In two patients aged 18 and 49, predominant spicular pigment migration was observed (LCA-6 and LCA-7), in contrast to the reported nummular pigmentation in one patient in the fourth decade (den Hollander et al., 2006). In one patient, nummular pigmentation was described in the first decade (LCA-10). Interestingly, a more severe phenotype was seen in LCA-20, who carried a heterozygous CRB1 null allele on top of two mutations in CEP290.
Extra-ocular features of CEP290-related LCA and potential modifier alleles in AHI1
Several patients with CEP290-related retinal dystrophy showed additional systemic features. Two patients with isolated LCA had several symptoms suggestive of renal dysfunction (3 and 23, Supp. Table S3). LCA-3 suffered from growth retardation, polydipsia, enuresis nocturna and diurnal incontinence. Kidney US at the age of seven, however, was normal. In LCA-23, kidney US at the age of three revealed increased echogenicity and kidneys without clear cortico-medullar differentiation. Despite this observation, no clinical nephrological manifestations were present at the age of 17. Since the age of onset of end-stage renal disease caused by CEP290 mutations may exceed the age of 20 (Helou et al., 2007; Tory et al., 2007), a close nephrological follow-up of these patients is required. Interestingly, both of these patients carry the recurrent c.2991+1655A>G mutation, which so far has only been reported in LCA patients without any other associated pathology. Of note, kidney US was available for only a subset of patients, and in general performed very early in life, when developing kidney disease might be difficult to detect.
In addition, four patients suffered from recurrent otitis media (OM) (LCA-5, SLS-1, SLS-3 and CORS-1, Supp. Table S3). Although this is common in childhood, it is worth mentioning that it is also a clinical manifestation often seen in primary ciliary dyskinesia (PCD), a genetically heterogeneous disorder of motile cilia (Leigh et al., 2009). In a few cases, PCD with OM was associated with X-linked RP, caused by mutations in RPGR (Shu et al., 2007). Notably, RPGR is a centrosomal protein that interacts with CEP290 (Chang et al., 2006). Moreover, loss-of-function experiments of CEP290 in zebrafish caused developmental abnormalities of the otic cavity (Sayer et al., 2006).
Strikingly, 33% of patients with CEP290-related isolated LCA presented with mental retardation and/or autism, in contrast to only 8% of patients with mutations in the other genes. Subtle brain abnormalities such as broadened lateral ventricles were seen on MRI in some patients. Of note, brain imaging was not available for a subset of patients. Additional neurological manifestations included movement abnormalities (LCA-21), and dyspraxia and balance/coordination problems (LCA-18), the latter of which was also evident in two patients with syndromic CEP290-related LCA (SLS-2 and CORS-1) (Supp. Table S3).
Taken together, these extra-ocular manifestations fit well into the broad clinical spectrum of CEP290 mutations, varying from isolated LCA to the lethal MKS. In addition, the CEP290 allelic spectrum is highly complex. Mutations associated with isolated LCA in this study were previously reported in other ciliopathies (Table 1), albeit always in compound heterozygosity with a different mutation in the distinct phenotypes. Moreover, identical CEP290 genotypes can display interfamilial variable expressivity and intrafamilial variation of the neurological phenotype was observed in several families with CEP290-related pathology (Coppieters et al., 2010). The complexity of CEP290-related disease is further illustrated by two cases from this study.
The first one is SLS-1, homozygous for p.Trp7Cys. Valente and coworkers identified the same mutation in patient with CORS, also of Pakistani origin (COR22, II:1). Despite a similar ocular and renal phenotype, both patients significantly differ in their neurological phenotype. A second and even more pronounced example is the variability in both nephrological and neurological involvement in three unrelated patients with the same
p.Lys1575X/p.Arg1465X genotype (SLS-2, SLS-3 and CORS-1). Patients SLS-3 and CORS-1 displayed a similar clinical course of renal disease, with renal failure at the age of 16 and 14 years, respectively. In contrast, renal insufficiency in patient SLS-2 was not substantiated until the age of 30 (Supp. Table S3). Neurological signs of these three patients ranged from a mild mental handicap (SLS-2) over severe autism in combination with moderate mental retardation (SLS-3) to severe mental retardation associated with ataxia and a MTS (CORS-1). An MRI was not available for the other two patients, however.
The AHI1 gene was screened as a candidate modifier gene in these three patients. Strikingly, CORS-1, with the most severe nephrologic and neurologic phenotype, carries a heterozygous novel p.Asn811Lys mutation in AHI1, which was absent in the two other patients. Upon screening of AHI1 in five additional patients with CEP290-related disease and neurological involvement, a novel missense variant, p.His758Pro, was identified in LCA-3. Mutations in AHI1 encoding Jouberin are responsible for JS, with retinal involvement in 75% and renal involvement in less than 10% of all AHI1-associated patients (Kroes et al., 2008). Interestingly, both p.Asn811Lys and p.His758Pro affect conserved residues and are located in the predicted WD40-repeat, a domain conserved across all eukaryotes, mediating functions such as vesicular trafficking (Li and Roberts, 2001). Since AHI1 and CEP290 appear to be in the same pathway through their interaction with rab8a, mutations in one of both genes may modify a phenotype caused by the other (Kim et al., 2008; Tsang et al., 2008; Hsiao et al., 2009). Tory and colleagues already suggested a similar potential epistatic effect of CEP290 and AHI1 mutations on phenotypes related to NPHP1, encoding another interactor of AHI1 (Tory et al., 2007; Eley et al., 2008). Strikingly, one of the AHI1 variants they described as a potential modifier for neurological involvement in patients with NPHP1 mutations, p.Arg830Trp, was recently identified as a modifier allele for retinal degeneration in patients with NPHP, independent of a primary NPHP1 mutation (Louie et al., 2010). Of note, four out of seven patients in the study from Tory and coworkers carrying p.Arg830Trp displayed visual impairment, with one blind individual (Tory et al., 2007). The p.Arg830Trp variant might affect AHI1 complex stability/formation (Louie et al., 2010). The variants identified here, assumed to represent a neurological modifier in patients with LCA, might disrupt interactions with other proteins, thereby influencing AHI1 function in other organ systems.
Overall, a molecular diagnosis of CEP290 mutations might have considerable consequences towards the clinical prognosis of an individual. Given the potential involvement of ciliary modifiers and the presence of cilia throughout the whole body, the development of various additional clinical manifestations should be taken into account. As for LCA, both children and (young) adults should have a long-term close clinical neurological and nephrological follow-up, since some features have a later onset.
In conclusion, molecular testing identified mutations in 69% of our LCA cohort, with a major involvement of CEP290. Detailed phenotyping of all patients with a molecular diagnosis revealed novel insights, mainly into the CEP290-related retinal phenotype, which is well documented for the first time in a larger patient group. The variable age-of-onset of the extra-ocular features emphasizes the importance of long-term clinical follow-up of LCA patients with CEP290 mutations. Moreover, the identification of potential modifiers of CEP290-related disease might contribute to a refined prognosis based on a molecular diagnosis. Finally, our findings fit with previous observations suggesting that the phenotype of ciliopathies is most likely determined by the synergistic effect of all variants occurring in the ciliary proteome.
ACKNOWLEDGMENTS
Contract grant sponsor: Research Foundation – Flanders (KAN 1.5.174.09, 01F01206, 3F001206, OZP 3G004306); Bijzonder Onderzoeksfonds (BOF06_Asp_FC UGent); Fund for Research in Ophthalmology.
REFERENCES
Baala L, Audollent S, Martinovic J, Ozilou C, Babron MC, Sivanandamoorthy S, Saunier S, Salomon R, Gonzales M, Rattenberry E, Esculpavit C, Toutain A, Moraine C, Parent P, Marcorelles P, Dauge MC, Roume J, Le Merrer M, Meiner V, Meir K, Menez F, Beaufrere AM, Francannet C, Tantau J, Sinico M, Dumez Y, MacDonald F, Munnich A, Lyonnet S, Gubler MC, Genin E, Johnson CA, Vekemans M, Encha-Razavi F, Attie-Bitach T. 2007. Pleiotropic effects of CEP290 (NPHP6) mutations extend to Meckel syndrome. Am J Hum Genet 81(1):170-9.
Bainbridge JW, Smith AJ, Barker SS, Robbie S, Henderson R, Balaggan K, Viswanathan A, Holder GE, Stockman A, Tyler N, Petersen-Jones S, Bhattacharya SS, Thrasher AJ, Fitzke FW, Carter BJ, Rubin GS, Moore AT, Ali RR. 2008. Effect of gene therapy on visual function in Leber's congenital amaurosis. N Engl J Med 358(21):2231-9.
Bowne SJ, Sullivan LS, Mortimer SE, Hedstrom L, Zhu J, Spellicy CJ, Gire AI, Hughbanks-Wheaton D, Birch DG, Lewis RA, Heckenlively JR, Daiger SP. 2006. Spectrum and frequency of mutations in IMPDH1 associated with autosomal dominant retinitis pigmentosa and leber congenital amaurosis. Invest Ophthalmol Vis Sci 47(1):34-42.
Brancati F, Barrano G, Silhavy JL, Marsh SE, Travaglini L, Bielas SL, Amorini M, Zablocka D, Kayserili H, Al-Gazali L, Bertini E, Boltshauser E, D'Hooghe M, Fazzi E, Fenerci EY, Hennekam RC, Kiss A, Lees MM, Marco E, Phadke SR, Rigoli L, Romano S, Salpietro CD, Sherr EH, Signorini S, Stromme P, Stuart B, Sztriha L, Viskochil DH, Yuksel A, Dallapiccola B, Valente EM, Gleeson JG. 2007. CEP290 mutations are frequently identified in the oculo-renal form of Joubert syndrome-related disorders. Am J Hum Genet 81(1):104-13.
Chang B, Khanna H, Hawes N, Jimeno D, He S, Lillo C, Parapuram SK, Cheng H, Scott A, Hurd RE, Sayer JA, Otto EA, Attanasio M, O'Toole JF, Jin G, Shou C, Hildebrandt F, Williams DS, Heckenlively JR, Swaroop A. 2006. In-frame deletion in a novel centrosomal/ciliary protein CEP290/NPHP6 perturbs its interaction with RPGR and results in early-onset retinal degeneration in the rd16 mouse. Hum Mol Genet 15(11):1847-57.
Chen S, Wang QL, Xu S, Liu I, Li LY, Wang Y, Zack DJ. 2002. Functional analysis of cone-rod homeobox (CRX) mutations associated with retinal dystrophy. Hum Mol Genet 11(8):873-84.
Cideciyan AV, Hauswirth WW, Aleman TS, Kaushal S, Schwartz SB, Boye SL, Windsor EA, Conlon TJ, Sumaroka A, Pang JJ, Roman AJ, Byrne BJ, Jacobson SG. 2009. Human RPE65 gene therapy for Leber congenital amaurosis: persistence of early visual improvements and safety at 1 year. Hum Gene Ther 20(9):999-1004.
Coppieters F, Lefever S, Vandesompele J, Leroy BP, De Baere E. 2010. CEP290, a gene with many faces: mutation overview and presentation of CEP290base. Hum Mutat 31, e-pub before print.
den Hollander AI, Heckenlively JR, van den Born LI, de Kok YJ, van der Velde-Visser SD, Kellner U, Jurklies B, van Schooneveld MJ, Blankenagel A, Rohrschneider K, Wissinger B, Cruysberg JR, Deutman AF, Brunner HG, Apfelstedt-Sylla E, Hoyng CB, Cremers FP. 2001. Leber congenital amaurosis and retinitis pigmentosa with Coats-like exudative vasculopathy are associated with mutations in the crumbs homologue 1 (CRB1) gene. Am J Hum Genet 69(1):198-203. den Hollander AI, Koenekoop RK, Mohamed MD, Arts HH, Boldt K, Towns KV, Sedmak T, Beer M, Nagel-Wolfrum K,
McKibbin M, Dharmaraj S, Lopez I, Ivings L, Williams GA, Springell K, Woods CG, Jafri H, Rashid Y, Strom TM, van der Zwaag B, Gosens I, Kersten FF, van Wijk E, Veltman JA, Zonneveld MN, van Beersum SE, Maumenee IH, Wolfrum U, Cheetham ME, Ueffing M, Cremers FP, Inglehearn CF, Roepman R. 2007. Mutations in LCA5, encoding the ciliary protein lebercilin, cause Leber congenital amaurosis. Nat Genet 39(7):889-95.
den Hollander AI, Koenekoop RK, Yzer S, Lopez I, Arends ML, Voesenek KE, Zonneveld MN, Strom TM, Meitinger T, Brunner HG, Hoyng CB, van den Born LI, Rohrschneider K, Cremers FP. 2006. Mutations in the CEP290 (NPHP6) gene are a frequent cause of Leber congenital amaurosis. Am J Hum Genet 79(3):556-61.
den Hollander AI, Roepman R, Koenekoop RK, Cremers FP. 2008. Leber congenital amaurosis: genes, proteins and disease mechanisms. Prog Retin Eye Res 27(4):391-419.
den Hollander AI, ten Brink JB, de Kok YJ, van Soest S, van den Born LI, van Driel MA, van de Pol DJ, Payne AM, Bhattacharya SS, Kellner U, Hoyng CB, Westerveld A, Brunner HG, Bleeker-Wagemakers EM, Deutman AF, Heckenlively JR, Cremers FP, Bergen AA. 1999. Mutations in a human homologue of Drosophila crumbs cause retinitis pigmentosa (RP12). Nat Genet 23(2):217-21.
Dephoure N, Zhou C, Villen J, Beausoleil SA, Bakalarski CE, Elledge SJ, Gygi SP. 2008. A quantitative atlas of mitotic phosphorylation. Proc Natl Acad Sci U S A 105(31):10762-7.
Dharmaraj S, Leroy BP, Sohocki MM, Koenekoop RK, Perrault I, Anwar K, Khaliq S, Devi RS, Birch DG, De Pool E, Izquierdo N, Van Maldergem L, Ismail M, Payne AM, Holder GE, Bhattacharya SS, Bird AC, Kaplan J, Maumenee IH. 2004. The phenotype of Leber congenital amaurosis in patients with AIPL1 mutations. Arch Ophthalmol 122(7):1029-37. Dryja TP, Adams SM, Grimsby JL, McGee TL, Hong DH, Li T, Andreasson S, Berson EL. 2001. Null RPGRIP1 alleles in
patients with Leber congenital amaurosis. Am J Hum Genet 68(5):1295-8.
Eley L, Gabrielides C, Adams M, Johnson CA, Hildebrandt F, Sayer JA. 2008. Jouberin localizes to collecting ducts and interacts with nephrocystin-1. Kidney Int 74(9):1139-49.
Frank V, den Hollander AI, Bruchle NO, Zonneveld MN, Nurnberg G, Becker C, Du Bois G, Kendziorra H, Roosing S, Senderek J, Nurnberg P, Cremers FP, Zerres K, Bergmann C. 2008. Mutations of the CEP290 gene encoding a centrosomal protein cause Meckel-Gruber syndrome. Hum Mutat 29(1):45-52.
Freund CL, Wang QL, Chen S, Muskat BL, Wiles CD, Sheffield VC, Jacobson SG, McInnes RR, Zack DJ, Stone EM. 1998. De novo mutations in the CRX homeobox gene associated with Leber congenital amaurosis. Nat Genet 18(4):311-2. Friedman JS, Chang B, Kannabiran C, Chakarova C, Singh HP, Jalali S, Hawes NL, Branham K, Othman M, Filippova E,
Thompson DA, Webster AR, Andreasson S, Jacobson SG, Bhattacharya SS, Heckenlively JR, Swaroop A. 2006. Premature truncation of a novel protein, RD3, exhibiting subnuclear localization is associated with retinal degeneration. Am J Hum Genet 79(6):1059-70.
Gerber S, Perrault I, Hanein S, Barbet F, Ducroq D, Ghazi I, Martin-Coignard D, Leowski C, Homfray T, Dufier JL, Munnich A, Kaplan J, Rozet JM. 2001. Complete exon-intron structure of the RPGR-interacting protein (RPGRIP1) gene allows the identification of mutations underlying Leber congenital amaurosis. Eur J Hum Genet 9(8):561-71.
Gu SM, Thompson DA, Srikumari CR, Lorenz B, Finckh U, Nicoletti A, Murthy KR, Rathmann M, Kumaramanickavel G, Denton MJ, Gal A. 1997. Mutations in RPE65 cause autosomal recessive childhood-onset severe retinal dystrophy. Nat Genet 17(2):194-7.
Hagstrom SA, North MA, Nishina PL, Berson EL, Dryja TP. 1998. Recessive mutations in the gene encoding the tubby-like protein TULP1 in patients with retinitis pigmentosa. Nat Genet 18(2):174-6.
Hanein S, Perrault I, Gerber S, Tanguy G, Barbet F, Ducroq D, Calvas P, Dollfus H, Hamel C, Lopponen T, Munier F, Santos L, Shalev S, Zafeiriou D, Dufier JL, Munnich A, Rozet JM, Kaplan J. 2004. Leber congenital amaurosis: comprehensive survey of the genetic heterogeneity, refinement of the clinical definition, and genotype-phenotype correlations as a strategy for molecular diagnosis. Hum Mutat 23(4):306-17.
Hanein S, Perrault I, Gerber S, Tanguy G, Rozet JM, Kaplan J. 2006. Leber congenital amaurosis: survey of the genetic heterogeneity, refinement of the clinical definition and phenotype-genotype correlations as a strategy for molecular diagnosis. Clinical and molecular survey in LCA. Adv Exp Med Biol 572:15-20.
Hauswirth WW, Aleman TS, Kaushal S, Cideciyan AV, Schwartz SB, Wang L, Conlon TJ, Boye SL, Flotte TR, Byrne BJ, Jacobson SG. 2008. Treatment of leber congenital amaurosis due to RPE65 mutations by ocular subretinal injection of adeno-associated virus gene vector: short-term results of a phase I trial. Hum Gene Ther 19(10):979-90.
Helou J, Otto EA, Attanasio M, Allen SJ, Parisi MA, Glass I, Utsch B, Hashmi S, Fazzi E, Omran H, O'Toole JF, Sayer JA, Hildebrandt F. 2007. Mutation analysis of NPHP6/CEP290 in patients with Joubert syndrome and Senior-Loken syndrome. J Med Genet 44(10):657-63.
Henderson RH, Waseem N, Searle R, van der Spuy J, Russell-Eggitt I, Bhattacharya SS, Thompson DA, Holder GE, Cheetham ME, Webster AR, Moore AT. 2007. An assessment of the apex microarray technology in genotyping patients with Leber congenital amaurosis and early-onset severe retinal dystrophy. Invest Ophthalmol Vis Sci 48(12):5684-9.
Hsiao YC, Tong ZJ, Westfall JE, Ault JG, Page-McCaw PS, Ferland RJ. 2009. Ahi1, whose human ortholog is mutated in Joubert syndrome, is required for Rab8a localization, ciliogenesis and vesicle trafficking. Hum Mol Genet 18(20):3926-41. Janecke AR, Thompson DA, Utermann G, Becker C, Hubner CA, Schmid E, McHenry CL, Nair AR, Ruschendorf F,
Heckenlively J, Wissinger B, Nurnberg P, Gal A. 2004. Mutations in RDH12 encoding a photoreceptor cell retinol dehydrogenase cause childhood-onset severe retinal dystrophy. Nat Genet 36(8):850-4.
Keen TJ, Mohamed MD, McKibbin M, Rashid Y, Jafri H, Maumenee IH, Inglehearn CF. 2003. Identification of a locus (LCA9) for Leber's congenital amaurosis on chromosome 1p36. Eur J Hum Genet 11(5):420-3.
Khanna H, Davis EE, Murga-Zamalloa CA, Estrada-Cuzcano A, Lopez I, den Hollander AI, Zonneveld MN, Othman MI, Waseem N, Chakarova CF, Maubaret C, Diaz-Font A, Macdonald I, Muzny DM, Wheeler DA, Morgan M, Lewis LR, Logan CV, Tan PL, Beer MA, Inglehearn CF, Lewis RA, Jacobson SG, Bergmann C, Beales PL, Attie-Bitach T, Johnson CA, Otto EA, Bhattacharya SS, Hildebrandt F, Gibbs RA, Koenekoop RK, Swaroop A, Katsanis N. 2009. A common allele in RPGRIP1L is a modifier of retinal degeneration in ciliopathies. Nat Genet.
Kim J, Krishnaswami SR, Gleeson JG. 2008. CEP290 interacts with the centriolar satellite component PCM-1 and is required for Rab8 localization to the primary cilium. Hum Mol Genet 17(23):3796-805.