Braz. J. Chem. Eng. vol.23 número2
Texto
Imagem
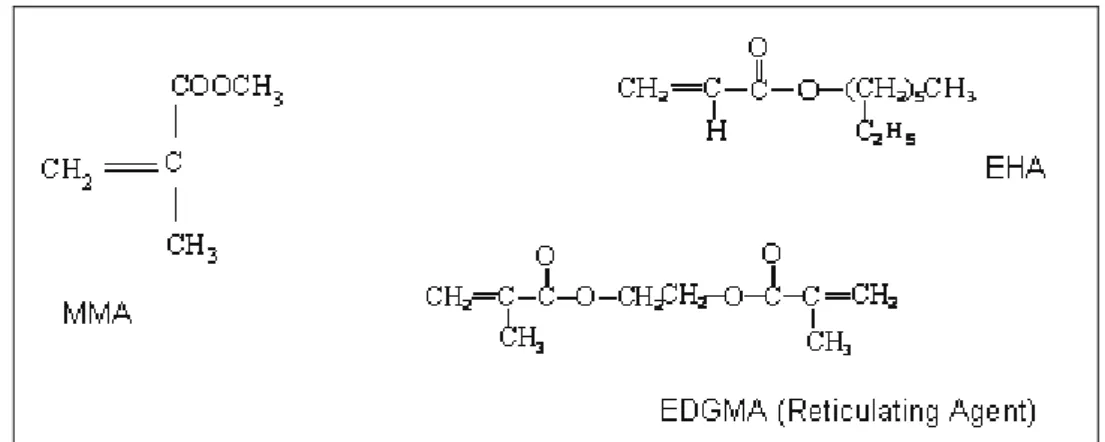
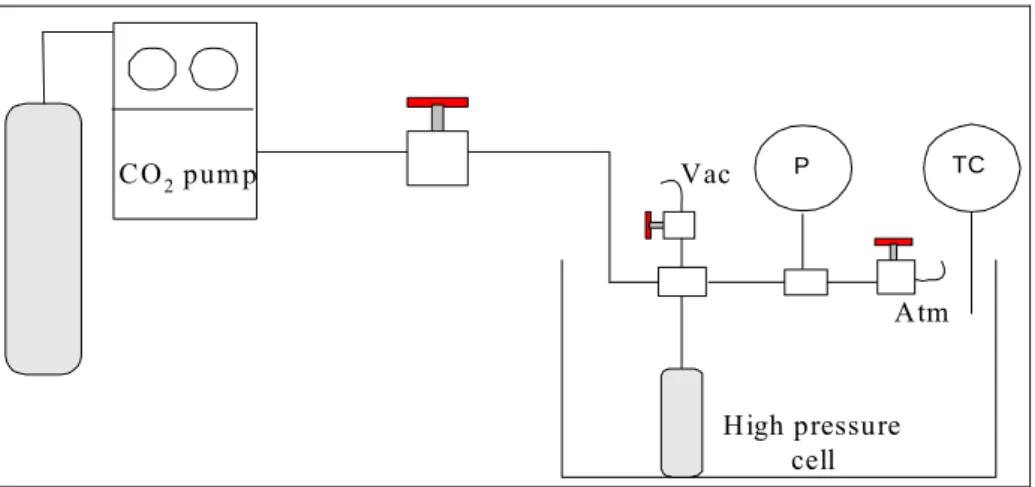

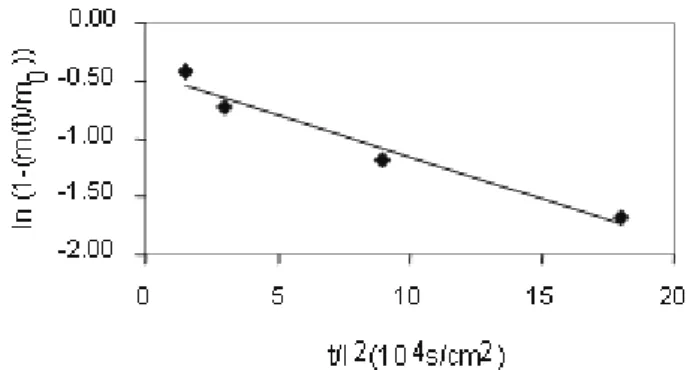
Documentos relacionados
Procurando responder às três questões levantadas, foram apontados alguns objetivos específicos, considerados essenciais na análise da importância geoestratégica que o Mar tem para
Prevalência de violência sexual entre escolares do 9º ano do ensino fundamental e OR bruto, segundo aspectos sociodemográficos e variáveis do contexto familiar, saúde
Prevalence of sexual violence among students of the 9th year of primary school and raw OR, according to sociodemographic aspects and variables of family context, mental health,
O Diabetes Mellitus referido entre os idosos se apresentou como um indicador não aconselhável para verificar a prevalência da doença na popula- ção estudada, em virtude da
From the 12 studies included (13 entries) in this meta-analysis, they all started from the same research assumption, in which the elderly with classifications according to the
During the development of the second edition of the Dietary Guidelines, this classification was improved; then were presented four groups: in natura or minimally processed
No capítulo 2 descrevem-se as formas adoptadas para estimar os mecanismos de deposição e de erosão de sedimentos coesivos, e o transporte total sólido de sedimentos não-coesivos,
Finalmente, a presença da natureza e de diferentes espécies do mundo vivo nos anúncios publicitários pode ser uma via diferente de aprender características acerca do