ISSN 0104-6632 Printed in Brazil
www.abeq.org.br/bjche
Vol. 30, No. 04, pp. 729 - 735, October - December, 2013
Brazilian Journal
of Chemical
Engineering
PRODUCTION OF CELLULOSE-DEGRADING
ENZYME ON SISAL AND OTHER
AGRO-INDUSTRIAL RESIDUES USING A NEW
BRAZILIAN ACTINOBACTERIA STRAIN
Streptomyces
sp. SLBA-08
E. P. Macedo
1, C. L. O. Cerqueira
1, D. A. J. Souza
1, A. S. R. Bispo
1,
R. R. R. Coelho
2and R. P. Nascimento
1,3*1
Universidade Federal do Recôncavo da Bahia (UFRB), Centro de Ciências Agrárias, Ambientais e Biológicas (CCAAB), 44380-000, Cruz das Almas - BA, Brazil.
2Universidade Federal do Rio de Janeiro (UFRJ), Instituto de Microbiologia Prof. Paulo de Góes
(IMPPG), 21942-590, Ilha do Fundão, Rio de Janeiro - RJ, Brazil.
3
Universidade Federal do Rio de Janeiro (UFRJ), Centro de Tecnologia (CT), Escola de Química (EQ), Phone/Fax: + (55) (21) 25627639, Departamento de Engenharia Bioquímica (DEB),
Bloco E, Sala 203, CEP: 21941-909, Ilha do Fundão, Rio de Janeiro - RJ, Brazil. E-mail: [email protected]
(Submitted: March 1, 2012 ; Revised: December 17, 2012 ; Accepted: December 18, 2012)
Abstract - Several types of lignocellulosic raw materials including wheat straw, sisal bagasse and sugarcane bagasse have different contents of cellulosic components. In our studies, aimed at isolating cellulose-degrading actinobacteria from Brazilian soils, a promising strain was selected and identified as Streptomyces sp. SLBA-08. This strain was tested for cellulase production under submerged fermentation in a mineral medium using different carbon sources (sisal bagasse, sugarcane bagasse and straw), as well as ammonium sulphate in different concentrations as nitrogen source. The results showed that medium containing 2.4% (w/v) sisal bagasse and 0.3% (w/v) ammonium sulphate resulted in the highest production of carboxymethylcellulase (1.11 U mL-1), after 48 hours. The pH and temperature profile showed optimal activity at pH 6.0 and 50 °C. As for thermostability, carboxymethylcellulases were tolerant at 50 °C, retaining 70% of the maximal activity even after 2h of incubation. The results obtained indicate that Streptomyces sp. SLBA-08 was capable of producing CMCase using lignocellulosic residues, especially sisal bagasse.
Keywords: Streptomyces sp. SLBA-08; Cellulose-degrading enzyme; Sisal bagasse; Sugarcane straw; Sugarcane bagasse.
INTRODUCTION
Actinobacteria are Gram positive filamentous bacteria abundantly found in soil, considered the most economically important and biotechnologically valu-able prokaryotes. They account for the production of about half of the discovered bioactive secondary me-tabolites (Bérdy, 2005), notably enzymes (Goodfellow
et al., 2005; Nascimento et al., 2009; Da Vinha et al., 2011). These cellulose degrading enzymes are related to industrial processes, like food, brewery and wine, agriculture, textile, detergent, animal feed, pulp and paper industries, as well as research and development technologies (Bhat, 2000; Jang and Chen, 2003; Coelho and Nascimento, 2011).
The utilization of agricultural by-products and other natural compounds for growing microor-ganisms may constitute an interesting alternative for enzyme production with lower costs. In Brazil, the bagasse portion of sugarcane comprises one of the largest cellulosic agro-industrial residues, and contains approximately 50% cellulose and 25% hemicellulose and lignin, while the straw is composed of 37.4% cellulose, 30% hemicellulose and 18.5% lignin (Pandey et al., 2000). Both substrates have already been tested as the main carbon source for the production of several enzymes including cellulases (Pandey et al., 2000; Adsul et al., 2004; Sukumaran et al., 2009; Maeda et al., 2010; Barros et al., 2010). As to sisal (Agaveae sisalana), this is the main worldwide plant used for fiber extraction, Brazil being the world’s largest producer (Andrade, 2011). Sisal fiber contains about 65% cellulose, 12% hemicellulose, 10% lignin and 10% starch, and may represent an important alternative source for enzymatic processes and biofuel production. However, it has not been reported yet as a substrate for enzyme production or other biotech-nological applications.
Studies dealing with cellulase (CMCase) produc-tion by actinobacteria using low-cost agro-industrial residues are scarce in literature, however our group has been isolating cellulolytic strains from Brazilian tropical soils for some time, obtaining promising results (Grigorevski-Lima et al., 2005; Nascimento et al., 2009; Da Vinha et al., 2011; Nascimento and Coelho, 2011).
In the present work we have isolated an actino-bacteria strain from a Brazilian soil under sisal cultivation, and tested it qualitatively for cellulase production. The CMCase production in submerged fermentation was then evaluated using three different agro-industrial residues as the main carbon source, sugarcane bagasse, sugarcane straw and sisal bagasse.
MATERIALS AND METHODS
Microorganism Isolation, Maintenance and Pre-liminary Test for Cellulolytic Activity
Actinobacteria SLBA-08 strain (CBMAI-1473) was isolated from a soil sample under Agave sisalana (sisal) cultivation in the semiarid region of Brazil,
using the dilution plate technique and starch-casein medium (Kuster and Williams, 1964). After incubation at 28 °C for 7-14 days, colonies with actinobacterial characteristics were isolated and tested qualitatively for cellulolytic activity. The first test was performed after growth on solid medium containing CMC and using Congo red to reveal CMC-degrading zones (Sazci, 1986); in a second one, after growth on cellu-lose-azure medium, liberation of azure dye was observed followed by the degradation of cellulose-azure (Plant et al., 1988). Stock cultures were main-tained on yeast extract-malt extract-agar plates (Shirling and Gottlieb, 1966) and the spore suspen-sion was prepared according to Hopwood et al. (1985) after cultivation (28 °C/15 days) in this same me-dium. Spores were maintained in 30% glycerol (v/v) at -18 °C.
Submerged Fermentation
Cells were cultivated in a mineral medium (Da Vinha et al., 2011) containing (g L-1): KH2PO4, 6.0; K2HPO4, 3.0; MgSO4.7H2O, 0.2; CaCl2, 0.05; MnSO4.7H2O, 0.01; ZnSO4.7H2O, 0.001. The medium was supplemented with three different carbon sources [sugarcane bagasse (SCB), sugarcane straw (SCS) and sisal bagasse (SB)] in two concentrations [0.8 and 2.4 % (w/v)], and a nitrogen source [ammonium sul-phate (AS)] also in two concentrations [0.3% (w/v) and 1.3 % (w/v], generating 4 runs for each carbon source (Table 1).
Table 1: Medium composition used in the different submerged fermentation conditions.
Culture Medium Carbon Sources %(w/v)
Nitrogen Source % (w/v)
1 0.8 0.3 2 2.4 0.3 3 0.8 1.3 4 2.4 1.3 All media were supplemented with a mineral solution (see Materials and Methods)
out in duplicate, and results were expressed as average values. Statistical tools were employed to evaluate the regression analysis and the significance of the experiments (standard deviation).
Enzymatic Assay
Carboxymethylcellulase (CMCase) activity was assayed by measuring the release of reducing sugars in a reaction mixture of 1.0 mL of the crude extract and 1.0 mL of 2.0% (w/v) carboxymethylcellulose (CMC) sodium salt (SIGMA®, St Louis, MO, USA) solution in 50 mM sodium citrate buffer (pH 4.8) incubated at 50 °C for 20 min. Reducing sugars were assayed by the dinitrosalicylic acid (DNS) method (Miller 1959). One unit (U) of CMCase activity corre-sponded to 1 µmol of glucose equivalent released per minute under the assay conditions (Ghose, 1987).
Partial Crude Enzyme Characterization: Influence of Temperature, pH Effect and Thermostability
Supernatants from strain SLBA-08 cultivated in the best conditions (SB 2.4% and AS 0.3%) were used as crude enzyme. The temperature profile for CMCase activity, assayed as described above, was determined by varying the incubation temperature between 20 °C and 90 °C at pH 4.8. In the same way, CMCase activity was determined in the pH range of 2.0–10.0, with the following buffers (50 mM) incu-bated at 50 °C: glycine-HCl for pH 2.0–3.0, sodium citrate for pH 3.0–6.0, sodium phosphate for pH 6.0– 8.0, Tris–HCl for pH 8.0–9.0 and glycine–NaOH for pH 9.0–10.0. To study the CMCase thermal stability, the supernatant was pre-incubated at 50 °C for 0.5, 1, 2, 4, 6, 8 and 16 hours (pH 4.8). The residual activity was measured by usual enzyme activity determina-tion procedures as described above. For comparison, we used the commercial cellulase Indiage Super L (Genencor). All the experiments were performed in duplicate, and results expressed as average values.
RESULTS AND DISCUSSION
Several actinobacterial strains were obtained from soil under sisal plantations in a semi-arid region of Brazil. However, preliminary tests (Congo-red and cellulose-azure) showed that strain SLBA-08 was the most promising and worthy of a more detailed study. This strain was further identified within the genus Streptomyces sp., based on a 16S RNA sequence analysis and spore chain morphology (data not show). The strain Streptomyces sp. SLBA-08 (number code
1473) was deposited in the “Coleção Brasileira de Microrganismos de Ambiente e Indústria (CBMAI)”.
The studies performed under submerged fermen-tation showed that the strain was capable of producing cellulases using sisal bagasse as the sole organic substrate. However, two other substrates (sugarcane bagasse and sugarcane straw) were also good sources for cellulase production. The kinetic profiles of en-zyme production in the four media tested (Table 1) for each substrate, obtained after five days of cultiva-tion under agitacultiva-tion at 30 °C, are described in Fig. 1. The best CMCase activity (1.11 U mL-1) occurred after two days of fermentation, when sisal bagasse was used in medium 02 [2.4% (w/v) of SB and 0.3% (w/v) AS] (Fig. 1(a)). However, very similar CMCase titers (1.01 U mL-1) were observed when sugarcane straw was the carbon source, after one day fermentation (Fig. 1(b)), at these same C and N source concentra-tions (medium 02). Streptomyces sp SLBA-08 was also able to produce cellulase using sugarcane bagasse as the sole organic source, with maximal levels of CMCase of 0.66 U mL-1, obtained after three days fermentation, also on the same medium (Fig 1(c)).
(a)
(c)
Figure 1: Fermentations time-course for CMCase production by Streptomyces sp. SLBA-08 at 30 oC in culture medium containing (a) sisal bagasse, (b) sugarcane straw and (c) sugarcane bagasse as carbon source at different concentrations, and ammonium sulphateas nitrogen source, as described in Table 1. Medium 1 (- -), Medium 2 (--), Medium 3 (-U-), Medium 4 (-S-).
The use of low-cost residues from agro-industries as the main substrates for research aimed at enzyme production by microorganisms, including cellulase production (Adsul et al., 2004), has increased in the last few years, since it significantly reduces costs. Our group has been studying this subject for some time, and interesting results have already been described for xylanase and protease production by actinobacteria using different residues (Nascimento et al., 2002; Nascimento et al., 2005; De Azeredo et al., 2006). We also studied cellulase production by several Streptomyces strains isolated from Brazilian soils growing on distilled dried grains (DDG), wheat bran, brewer’s spent grains and sugarcane bagasse in different concentrations and using corn steep liquor, which is also a low-cost residue from agriculture, as the main nitrogen source. Streptomyces drozdowiczii grown in 1.0% (w/v) DDG and 0.3% (w/v) corn steep liquor was able to produce around 0.15 U mL-1 CMCase in the crude supernatant, whereas when using wheat bran in the same conditions an activity of 0.22 U mL-1 was obtained (Grigorevski-Lima et al., 2005). Nascimento et al. (2009) obtained values of 0.72 U mL-1 as maximum activity for CMCase when growing Streptomyces malaysiensis AMT-3 on 0.5% (w/v) brewer's spent grains and 1.2% (w/v) corn steep liquor, after four days of fermentation. More recently, even better results were obtained by Da Vinha et al. (2011), who detected a high CMCase produc-tion (2.00 U mL-1) by Streptomyces viridobrunneus
SCPE-09 cultivated on 2.0% (w/v) wheat bran and 0.19% (w/v) corn steep liquor after five fermentation days. Very few other reports are described besides those reported by our group. Tuncer et al. (2004), for instance, studied the production of endoglucanase, among other enzymes, with Streptomyces sp. F262 grown with 1.2% (w/v) ball-milled wheat straw supplemented with yeast extract and observed a very low production. However, it must be emphasized that a comparison between results described in the literature is difficult, since the conditions for cellulase activity and enzyme production are not always the same. Nonetheless, the results presented here suggest that Streptomyces sp. SLBA-08 is a good producer of CMCase using lignocellulosic wastes as carbon source, especially sisal bagasse or sugarcane straw. At the present time, as far as we are concerned, there is no citation in the literature describing the use of sisal bagasse for the production of hydrolytic enzymes like cellulases, xylanases or amylases.
According to the National Supply Company (CONAB, Brazil), the Brazilian production of dry sisal fiber was estimated to be 139,700 tons in 2004, at the same time that 489,000 tons of sisal bagasse were discarded. These values represent a significant amount (almost 3 times the fiber production weight) and should somehow be recovered. Indeed, in Brazil this waste is habitually abandoned in the field, and only a small portion is used as fertilizer or animal feed (Andrade, 2011).
Thus, the use of sisal bagasse may represent a new alternative for the production of bioactive sub-stances such as lignocellulolytic enzymes from residual plant biomass, particularly cellulases. Also consid-ering bioethanol in the present international scenario, it is evident that second generation ethanol produc-tion will soon become a reality. In this sense, sisal becomes as an excellent option for the cheap production of cellulase.
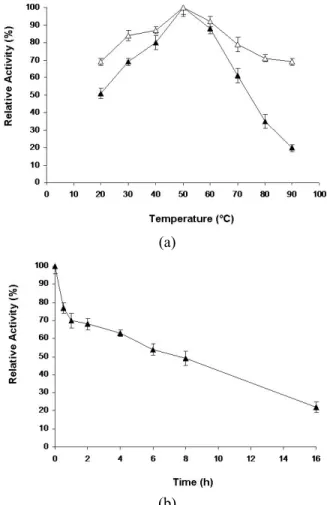
at temperatures between 40° and 60 °C (Nascimento et al., 2009). Thermal stability experiments are shown in Fig. 2(b). Crude enzyme was able to retain 70% residual activity at 50 °C for 2 h, the half-life being 6 h at this temperature. Half-lives of 24 h at 40 °C or 8 h at 50 °C have been cited in the literature for some Streptomyces strains (Grigorevski-Lima et al., 2005; Nascimento et al., 2009; Da Vinha et al., 2011).
(a)
(b)
Figure 2: Effect of temperature (a) and thermal stability (b) at 50 ºC on the activity (pH 4.8) of CMCase produced by Streptomyces sp. SLBA-08 (S) grown on 2.7% (w/v) sisal bagasse and 0.8% (w/v) ammonium sulphate and the commercial enzyme Indi-age Super L (U). Residual activity is expressed as a percentage of the original activity. Error bars represent the standard deviation of each experimental point (n=2).
The pH activity profile of the crude extract obtained under the best growth conditions (Fig. 3) shows that 80% CMCase activity was maintained over the pH range 4.0–7.0, with optimal activity oc-curring at pH 6.0. Although commercial Indi-Age cellulase is superior at pH values below 4 or above 8,
our strain showed promising results in the pH range from 4 to 8.
Figure 3: Effect of pH on activity (50 ºC) of CMCase produced by Streptomyces sp. SLBA-08 (S) grown in 2.7% (w/v) sisal bagasse and 0.8% (w/v) ammonium sulphate and 0.25% (w/v) peptone and the commercial enzyme Indi-age Super L (U). The ionic strength for all buffers was 50mM. Residual activity is expressed as a percentage of the original activity. Error bars represent the standard deviation of each experimental point (n=2).
Da Vinha et al. (2011) described a similar profile (above 80% of relative activity) for S. viridobrunneus SCPE-09 in the pH range 4.0–7.0, with optimal ac-tivity around 5.0. CMCase acac-tivity in this pH range (4.0–8.0) was also detected by Grigorevski-Lima et al. (2005) for CMCase of S. drozwdowiczii. Nascimento et al. (2009) also observed a similar profile for CMCase of Streptomyces malaysiensis AMT-3.
CONCLUSIONS
In the present research we were able to isolate from a Brazilian soil under sisal cultivation an acti-nobacterial strain, Streptomyces sp SLBA-08, that was able to grow and produce cellulases (CMCases) in a cultivation medium containing solely a mineral salt solution and very cheap agriculture residues, either sisal bagasse, sugarcane straw or sugarcane bagasse as carbon source. Values of up to 1.11 U mL-1 of enzyme activity were obtained after only two days fermentation when sisal bagasse was used. The crude extract thus obtained gave maximal activity in an acidic pH range (4.0-7.0) and temperatures around 50-60 °C, with a half life of 6h at 50 °C.
characteris-tics, these results indicate the possibility of using these enzymes in biotechnological processes. As already stressed, sisal bagasse is a very abundant agricultural residue in Brazil, being almost entirely left in the field, and so could be easily recovered for enzyme production. In fact, the present work reports, for the first time, the use of sisal bagasse as a carbon source for cellulase production by an actinobacteria strain. However, it can represent an important alter-native lignocellulosic source for the production of many other enzymes of biotechnological importance. Our results also confirm the importance of search-ing for new biotechnological microbial sources in different ecosystems. Brazilian soils have shown a great microbial biodiversity with different enzyme profiles, especially cellulases, xylanases, and pro-teinases, and may have a great potential for the discovery of new strains with novel characteristics.
ACKNOWLEDGEMENTS
The authors would like to thank the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), and Fundação de Amparo a Pesquisa do Estado da Bahia (FAPESB) for financial support.
REFERENCES
Andrade, W., Sisal fiber in Brazil. Brazilian Fiber Publishing Web. http://www.braziliansisal.com (2011). (Accessed: December 04, 2011).
Adsul, M. G., Ghule. J. E., Singh, R., Shaikh, H., Bastawde, K. B., Gokhale, D. V., Varma, A. J., Polysaccharides from bagasse: Applications in cellulase and xylanase production. Carbohydrate Polymer, 57, 67-72 (2004).
Barros, R. R. O., Oliveira, R. A., Gottschalk, L. M. F., Bon, E. P. S., Production of cellulolytic enzymes by fungi Acrophialophora nainiana and Ceratocystis paradoxa using different carbon sources. Applied Biochemistry Biotechnology, 161, 448-454 (2010). Bérdy, J., Bioactive microbial metabolites. Journal of
Antibiotic, 58, 1-26 (2005).
Bhat, M. K., Cellulases and related enzymes in biotechnology. Biotechnology Advances, 18, 355-383 (2000).
Carriales, V., Jaffe, W., Solid state fermentation, an appropriate biotechnology for developing coun-tries. Interciencia, 11, 9-15 (1986).
Da Vinha, F. N. M., Gravina-Oliveira, M. P., Franco, M. N., Macrae, A., Bon, E. P. S., Nascimento, R. P., Coelho, R. R. R., Cellulase production by Strep-tomyces viridobrunneus SCPE-09 using ligno-cellulosic biomass as inducer substrate. Applied Biochemistry Biotechnology, 164, 256-267 (2011). Dawson, L., Boopathy, R., Use of post-harvest sugar
cane residue for ethanol production. Bioresource Technology, 98, 1695-1699 (2007).
De Azeredo, L. A. I., De Lima, M. B., Coelho, R. R. R., Freire, D. M. G., A low-cost fermentation me-dium for thermophilic protease production by Streptomyces sp. 594 using feather meal and corn steep liquor. Current Microbiology, 53, 335-339 (2006).
Deepchand, K., Economics of electricity production from sugar cane tops and leaves, a preliminary study. International Sugar Journal, 88, 210-216 (1986).
George, S. P., Ahmad, A., Rao, M. B., Studies on carboxymethyl cellulose produced by an alka-lothermophilic actinomycete. Bioresource Tech-nology, 77, 171-175 (2001).
Ghose, T. K., Measurement of cellulase activities. Pure Applied Chemistry, 59, 257-268 (1987).
Goodfellow, M., Williams, S. T., Mordarski, M., Actinomycetes in Biotechnology. Academic Press, London (1988).
Grigorevski-Lima, A. L., Nascimento, R. P., Bon, E. P. S., Coelho, R. R. R., Streptomyces drozdowiczii cellulase production using agro-industrial by-products and its potential use in the detergent and textile industries. Enzyme and Microbial Tech-nology, 37, 272-277 (2005).
Hopwood, D. A., Bibb, M. J., Chater, K. F., Kieser, T., Bruton, C. J., Kieser, H. M., Lydiate, D. J., Smith, C. P., Ward, J. M., Schrempf, H., Genetic Ma-nipulation of Streptomyces. A Laboratory Manual. The John Innes Institute, Norwich, United Kingdom (1985).
Hoshiro, E., Wada, Y., Nishizawa, K., Improvements in the hygroscopic properties of cotton cellulose by treatment with an endo-type cellulase from Streptomyces sp. KSM-26. Journal of Bioscience and Bioengineering, 88, 519-525 (1999).
Jang, H. D., Chen, K. S., Production and charac-terization of thermostable cellulases from Streptomyces transformant T3-1. World Journal of Microbiology and Biotechnology, 19, 263-268 (2003).
Maeda, R. N., Da Silva, M. M. P., Santa Anna, L. M. M., Pereira-Jr. N., Nitrogen source optimization for cellulase production by Penicillium funiculosum, using a sequential experimental design methodology and the desirability function. Appl. Biochem. Biotechnol., 161, 411-422 (2010).
Miller, G. L., Use of dinitrosalicylic acid reagent for determination of reducing sugar. Analytical Chemistry, 31, 426-428 (1959).
Nascimento, R. P., Coelho, R. R. R., Cellulases: from production to biotechnological applications. In: Cellulase: Types and Action, Mechanism, and Uses. (1st Ed.) Nova Science Publishers, Inc., New York, p. 109-134 (2011).
Nascimento, R. P., Coelho, R. R. R., Marques, S., Alves, L., Gírio, F. M., Bon, E. P. S., Collaço, M. T. A., Production and partial characterisation of xylanases from Streptomyces sp. strain AMT-3 isolated from Brazilian cerrado soil using agro-industrial by-products. Enzyme and Microbial Technology, 31, 549 - 555 (2002).
Nascimento, R. P., Alves Jr. N., Pereira-Jr. N., Bon, E. P. S., Coelho, R. R. R., Brewer’s spent grain and corn steep liquor as substrates for cellulolytic enzymes production by Streptomyces malaysiensis. Letters in Applied Microbiology, 48, 529-535 (2009).
Palmarola-Adrados, B., Choteborská, A., Galbe, M., Zacchi, G., Ethanol production from non-starch carbohydrates of wheat bran. Bioresource Tech-nology, 96, 843-850 (2005).
Pandey, A., Soccol, C. R., Nigam, P., Soccol, V. T., Biotechnological potential of agroindustrial resi-dues. I: Sugar cane bagasse. A review. Bioresource Technology, 74, 69-80 (2000).
Papinutti, V. L., Forchiassin, F., Lignocellulolytic enzymes from Fomes sclerodermeus growing in solid-state fermentation. Journal of Food Engi-neering, 81, 54-59 (2007).
Plant, J. E., Attwell, R. W., Smith, C. A., A
semi-micro quantitative assay for cellulolytic activity in microorganisms. Journal of Microbiology Methods, 7, 259-263 (1988).
Sazci, A., Radford, A., Erenler, K., Detection of cellulolytic fungi by using Congo red as an indi-cator: A comparative study with the dinitrosali-cyclic acid reagent method. Journal of Applied Bacteriology, 61, 559-562 (1986).
Semêdo, L. T. A. S., Gomes, R. C., Linhares, A. A., Duarte, G. F., Nascimento, R. P., Rosado, A. S., Alviano, C. S., Manfio, G. P., Soares, R. M. A., Coelho, R. R. R., Streptomyces drozdowiczii sp. nov., a novel cellulolytic streptomycete from soil in Brazil. International Journal of Systematic and Evolutionary Microbiology, 54, 1323-1328 (2004). Shirling, E. B., Gottlieb, D. Methods for charac-terization of Streptomyces species. International Journal of Systematic Bacteriology, 16, 313-340 (1966).
Souza, R. F., Coelho, R. R. R., Macrae, A., Soares, R. M. A., Nery, D. C. M., Semêdo, L. T. A. S., Alviano, C. S., Gomes, R. C., Streptomyces lunalinharesii sp. nov., a chitinolytic streptomycete isolated from cerrado soil in Brazil. International Journal of Systematic and Evolutionary Microbiology, 58, 2774-2778 (2008).
Sukumaran, R. K., Singhania, R. R., Mathew, G. M., Pandey, A., Cellulase production using biomass feed stock and its application in lignocellulose saccharification for bioethanol production. Re-newable Energy, 34, 421-424 (2009).
Schrempf, H., Walter, S., The cellulolytic system of Streptomyces reticuli. International Journal of Biological Macromolecules, 17, 353-355 (1995). Tuncer, M., Kuru, A., Isikli, M., Sahin, N., Çelenk,