Despite the success of osseointegrated implants, failures have increased significantly, associated with development of implantitis. Multiple factors influence the peri-implant bone loss, including environmental and genetic causes. BMPs (Bone morphogenetic proteins) are growth factors that induce bone formation. FGF (fibroblast growth factors) and their receptors (FGFRs) play important roles by controlling the levels of cell proliferation, differentiation and migration. BMP/FGF relationship is responsible for promoting bone regeneration and bone loss.The aim of this study was to analyze the correlation between BMP4, FGF3, FGF10 and FGFR1 genes and peri-implant bone loss. Two hundred and fifteen volunteers, with 754 dental implants, were submitted to oral examination and divided in healthy group (n=129) and peri-implantitis group (n=86). Thirteen polymorphisms in BMP4, FGF3, FGF10 and FGFR1 genes were analyzed individually and in haplotype. The chi-square test correlated genotypes, allelic and haplotype frequencies. Values of p<0.05 were considered significant. Volunteers with peri-implantitis demonstrated high incidence of total edentulism (p<0.0001) and thin peri-implant phenotype (p<0.04). Higher incidence of spontaneous bleeding, plaque and implant mobility was observed in peri-implantitis group (p<0.0001 for all). The TT polymorphic genotype for BMP4 rs2761884 was associated with healthy peri-implant (p=0.01). FGF3 rs4631909 (TT+CT genotype) also showed association with the control group (p=0.04). The frequency of C allele for FGF3 rs4631909 showed a tendency for association with peri-implantitis (p=0.08). FGF10 CCTG (p=0.03), BMP4 GAAA (p=0.05) and GGGA (p=0.02) haplotypes were associated with peri-implantitis (p=0.03). Therefore, it may be concluded that BMP4 and FGF10 haplotypes are associated with peri-implantitis.
Haplotypes in BMP4 and FGF Genes
Increase the Risk of Peri-Implantitis
Renata Barboza Coelho1, Roberto Gonçalves Junior1, Ricardo de Mello Villas-Boas1, Leticia Ladeira Bonato1, Valquiria Quinelato1, Aristides da Rosa Pinheiro2, Aldir Machado2, Carlos Henrique Ramirez Nunes1, Rackel Gonçalves1, Alexandre Resende Vieira3, José Mauro Granjeiro4, Priscila Ladeira Casado5
1Post-graduate Program in Dentistry, UFF - Universidade Federal Fluminense, Niterói, RJ, Brazil 2Post-graduate Program in Dentistry and Implantology, UFF - Universidade Federal Fluminense, Niterói, RJ, Brazil 3Department of Oral Biology, School of Dental Medicine, University of Pittsburgh, Pittsburgh, PA, USA 4Clinical Research Unit and Biology Institute, UFF - Universidade Federal Fluminense, Niterói, RJ, Brazil and Instituto Nacional de Metrologia, Rio de Janeiro, RJ, Brazil 5Department of Clinical Dentistry, School of Dentistry, UFF - Universidade Federal Fluminense, Niterói, RJ, Brazil Correspondence: Priscila Ladeira Casado, Rua Mario Santos Braga, 28, Centro, Campus do Valonguinho, 24020-140 Niterói, RJ, Brasil. Tel: +55-21-2629-9803. e-mail: [email protected]
Key Words:
peri-implantitis, polymorphism, haplotype, FGF, BMP
Introduction
Osseointegrated oral implants have been widely used as first-line therapy in cases of total or partial dental arch rehabilitation (1). However, despite the reported high success rate, failures in implant dentistry have a significant increase because of peri-implantitis development, characterized by the pathological resorption of adjacent bone to the endosseous implant and may result in implant loss (2).This process of bone loss is influenced by a multitude of factors, some of which are understood while others are still unknown and share several environmental and genetic causes (3). Recent studies have associated peri-implantitis with the host’s destructive inflammatory response characterized by the high expression of proinflammatory cytokines and osteoclast activation (4).
In bone repair, the biological response to bacterial or traumatic tissue damage can result in wound healing or culminate in advanced bone loss. Hematoma formation during bone regeneration is the basis for bone repair. The release of growth factors, including platelet-derived growth factors (PDGF), tumor growth factor type-β (TGF-β), interleukins (IL) 1 and 6, during inflammatory response increase osteoprogenitor cells and fibroblasts proliferation
and are associated with bone morphogenetic proteins (BMP) and fibroblast growth factors (FGF) expressions. As the hematoma matures, a collagen matrix develops with neovascularization, providing a scaffold for these cells (5).When this regenerative ability fails to maintain osseointegration by a cascade of bone remodeling processes, the stimulation of bacterial presence associated with intrinsic factors triggers pathological bone loss (6).
BMPs (bone morphogenetic proteins) are growth factors and belong to the TGF-β superfamily located in chromosome 14q22-23, originally discovered due to their ability of inducing bone formation and cartilage. BMPs are pivotal in building a group of morphogenetic signals orchestrating tissue architecture throughout the body (7). Recent research shows that the highest concentration of BMPs is in articular cartilage, osteoblasts, osteoclasts and osteoprogenitor cells (8,9). BMP-4 is essential for the skeletal development during embryogenesis and its expression increases in adulthood during fracture healing. At the molecular level, specific genetic variants of BMP4 are associated with the risk of decreased bone density of the hip in postmenopausal women (10).
R. B
. Coelho et al.
morphogenesis and organogenesis by controlling the levels of cell proliferation, differentiation, migration, adhesion and death (11).
The combination of angiogenic and osteogenic growth factors such as BMP/TGF- β and BMP/FGF is able to promote bone regeneration by enhancing osseointegration in vivo. The antagonism between BMP and FGF contributes to healing in bone lesions. FGFR-1 is a positive regulator of bone formation and interacts with FGF3 and FGF10 promoting angiogenesis and fracture healing (12,13).
The variation in severity during peri-implantitis development, observed among volunteers that underwent similar environmental factors, suggests that the genetic aspects of peri-implantitis need to be investigated to understand the regulation in the pathogenesis associated with this disease (3). Therefore, considering the interaction and modulation of the BMP/FGF axis during bone remodeling, the study hypothesis was that the process of peri-implantitis may be related to genetic alterations in the BMP and FGF families. Thus, the aim of this study was to analyze the correlation between genetic polymorphisms in BMP4, FGF3, FGF10 and FGFR1 and the bone loss process around dental implant.
Material and Methods
Volunteers
This is an observational, cross-sectional and double-blinded study. Two hundred and fifteen volunteers, with 754 dental implants, were enrolled for the study from the pool of the patients attended at the Dental Implantology Clinic of the School of Dentistry at UFF, Brazil, over a course of one year. The clinical procedures were conducted in accordance with the recommendations of the local Research Ethics Committee (Registration number 238/10). The term of informed consent was obtained from all participants. The initial clinical parameters of the volunteers are in Table 1. All participants provided their medical and dental history (Table 2) and reported smoking habits. The exclusion criterion was implant failure (pathologic bone loss, implant mobility or implant loss) before the osseointegration period (3 months for mandible and six months for maxillary), bisphosphonate use, pregnancy and/or lactation in female volunteers, no preoperative radiography, one stage or immediate implant placement, concurrent bone grafting required, early implant exposure during osseointegration period,non-treated periodontitis and non-compliance with study protocol. The inclusion criteria were as follows: at least one osseointegrated endosseous implant, immediate postoperative radiography showing the vertical bone level around implant to compare bone level after the osseointegration period, periapical radiography showing periodontal status before implant placement, and annual
follow-up clinical and radiographic examinations. All implants were placed in a submerged healing modality (two-stage concept) in sites that had previously showed favorable bone quality and quantity (14).
Diagnosis of Peri-implantitis
Peri-implant sites were submitted to clinical examination based on Junior et al. (14), which consisted of visual inspection and palpation, analysis of mucosa inflammation, probing pocket depth, bleeding on probing and spontaneous bleeding in four aspects (mesial, buccal, distal and lingual/ palatine), presence of plaque, peri-implant phenotype, implant mobility (any mobility during percussion test), osseointegration period and implant platform type. Conventional periapical radiography, using the paralleling technique, was used to assess the presence of vertical bone loss around the implants by measuring the height of peri-implant bone and comparing it with the initial radiography taken immediately after implant placement. According to the clinical and radiographic characteristics of the peri-implant sites, volunteers were characterized as having healthy sites - no clinical signs of inflammation in the peri-implant mucosa and no signs of bone loss – or peri-implantitis with radiographic signs of pathologic bone loss in at least one region. Physiological bone loss was characterized considering a normal bone loss of 1 mm during the first year following implant placement and 0.2 mm for subsequent years. The diagnosis of peri-implantitis was based on radiographic bone level. If the total bone loss was higher than 1 mm and 0.2 mm per year, the volunteer was diagnosed with peri-implantitis.
Selection of Single Nucleotide Polymorphism (SNP) and Genotyping
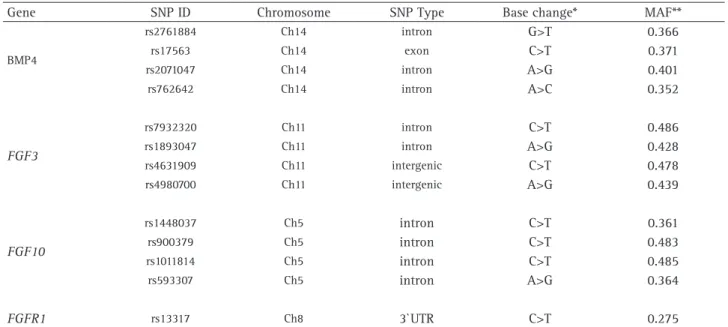
DNA from all participants was extracted from buccal cells after vigorously rinsing with 5 mL of saline solution for 60 s, as described previously (15). A NanoDrop spectrophotometer (Thermo Scientific, Wilmington, DE, USA) was used to determine the amount and purity of the DNA. Only DNA samples showing A260 nm/A280 nm ratios greater than 1.9 were used. All procedures included in SNP selection and analysis followed the STREGA reporting recommendations (16). Linkage disequilibrium relationships and gene structure were considered to select the thirteen SNPs in the candidate’s genes included in this study. The least allele frequencies reported in the database of the National Center for Biotechnology Information (http:// www.ncbi.nlm.nih.gov/ SNP/) was >0.2 (Table 4).
PTC-BMP4 and FGF haplotypes in peri-implantitis
225 (Peltier Thermal Cycler, Bio Rad Life Science, Corston, UK). As BMP4, FGF3 and FGF10 genes are found in the same chromosome, it is believed that they may act as a unit. Therefore, polymorphisms in these genes were not analyzed individually but in combination, termed as haplotype.
Statistical Analysis
The nominal variables were expressed as frequencies and percentages. The chi-square was used to access the significance of the nominal variables among the groups. Continuous variables were expressed as mean and standard deviation. After the Shapiro-Wilk test showed normal distribution, analysis of variance and the Student’s t-test were applied. Differences in the frequency of genotypes and alleles between peri-implantitis and the control groups were analyzed using Fisher’s exact test and chi-square test, after fitting in Hardy-Weinberg equilibrium. The p values of <0.05 were considered statistically significant, and the risk of individual alleles and genotypes associated with the development of the disease was calculated by odds ratio (OR) in a 95% confidence interval. Multivariate regression analysis was used to explore many covariates simultaneously. Statistical analysis was applied using STATA 11.1 (Stata Corp, College Station, TX, USA). To calculate the haplotypes was used the computer program ARLEQUIN (v.20-ttp://anthro. unige.ch/arlequin).
Results
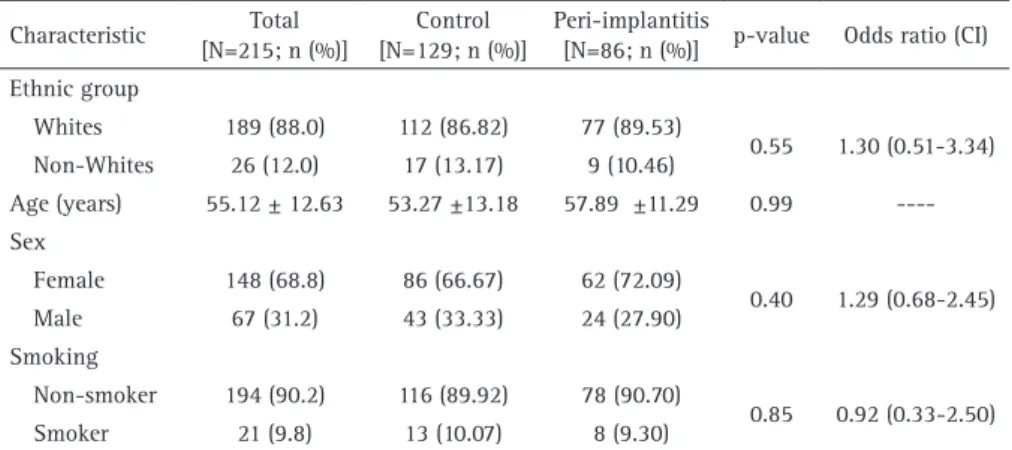
Among all 215 volunteers evaluated over the course of one year, 148 (69%) were women and 67 (31%) men, with mean age 55.12±12.63 years. No difference was found between the test group (peri-implantitis) when compared with the control group (healthy) regarding the ethnicity, gender, age and smoking habit (Table 1).
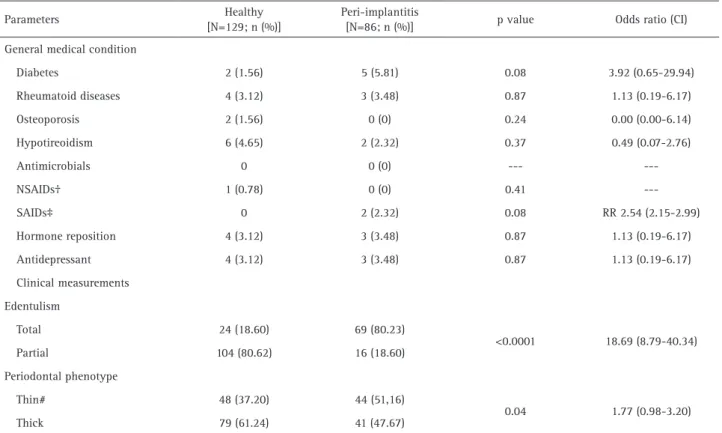
Total edentulism (p<0.0001) and thin peri-implant phenotype (p=0.04) were highly incident in volunteers with peri-implantitis (Table 2). The peri-implant status
(Table 3) showed a high prevalence for bleeding on probing, spontaneous bleeding, high probing pocket depth, presence of plaque and implant mobility in peri-implantitis volunteers (p<0.001 for all).
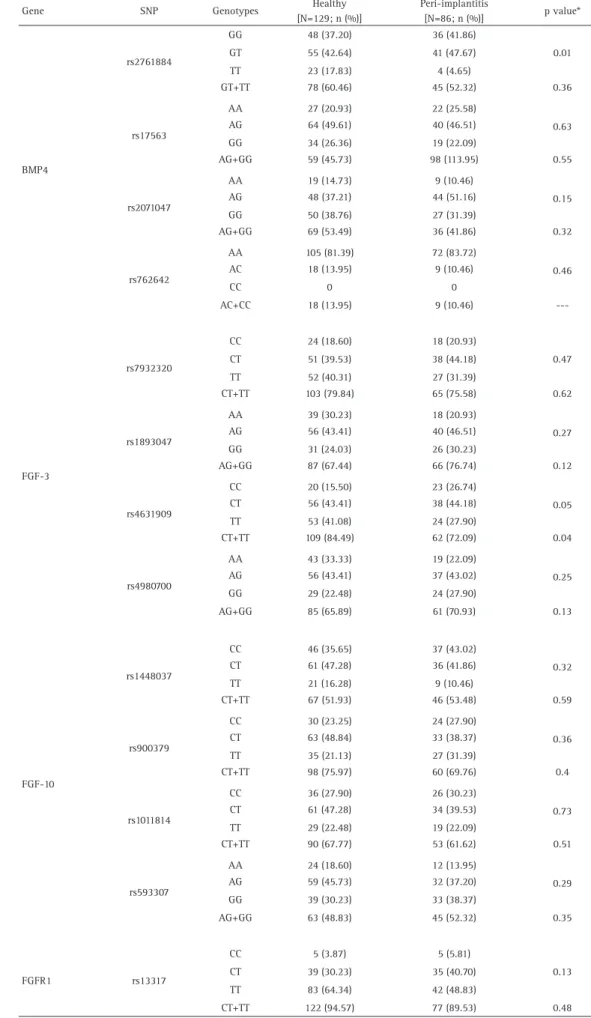
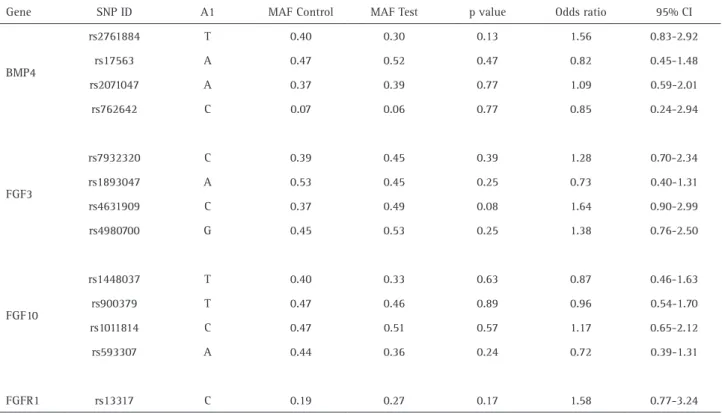
The polymorphic genotype TT of BMP4 rs2761884 was associated with the healthy group (p=0.01). FGF3 rs4631909 TT+CT genotypes also showed association with the control group (p=0.04) (Table 5). In addition, the frequency of C allele for FGF3 rs4631909 showed a tendency for association with the peri-implantitis group (p=0.08) (Table 6). Genotype distributions were within Hardy-Weinberg equilibrium.
Haplotypes distribution in both groups (Table 7). The distribution of haplotypes arranged as alleles was constructed for BMP4, FGF3 and FGF10 genes and assessed for association. A significant association between FGF10 CCTG haplotype and peri-implantitis was evident (p=0.03). There also was a significant association between the GAAA (p=0.05) and GGGA (p=0.02) haplotypes of the BMP4 gene and occurrence of peri-implantitis. FGF3 TGCG haplotype showed a tendency for association with peri-implantitis.
In order to assess risk factors concurrently, a multivariate logistic regression of individual parameters in the test groups was performed (Table 8). This yielded adjusted odds ratios (OR) for individual parameters, including age, gender, BMP4 rs2761884 and FGF3 rs4631909, previously associated with protection against peri-implantitis. This analysis confirmed previous univariate results.
Discussion
Evidence shows that peri-implant diseases are related to genetic factors (17). Considering the interaction and modulation of the BMP/FGF axis during bone remodeling, the study aimed to analyze the correlation between BMP4, FGF3, FGF10 and FGFR1 genes in the pathological process of peri-implant bone loss. To elucidate such association, this study was conducted to evaluate a total of 215 volunteers divided into two groups, according to the presence or
absence of peri-implantitis. This study confirmed the association of BMP4, FGF3 and FGF10 haplotypes with peri-implantitis, irrespective of the presence of previously d e s c r i b e d r i s k f a c t o rs , suggesting that different pathways can trigger this disease.
Peri-implantitis affects a group of subjects and it is correlated to risk factors that have been pre-established in the literature, such as smoking, Table 1. Baseline characteristics of the complete sample
Characteristic Total
[N=215; n (%)]
Control [N=129; n (%)]
Peri-implantitis
[N=86; n (%)] p-value Odds ratio (CI)
Ethnic group
Whites 189 (88.0) 112 (86.82) 77 (89.53)
0.55 1.30 (0.51-3.34)
Non-Whites 26 (12.0) 17 (13.17) 9 (10.46)
Age (years) 55.12 ± 12.63 53.27 ±13.18 57.89 ±11.29 0.99
----Sex
Female 148 (68.8) 86 (66.67) 62 (72.09)
0.40 1.29 (0.68-2.45)
Male 67 (31.2) 43 (33.33) 24 (27.90)
Smoking
Non-smoker 194 (90.2) 116 (89.92) 78 (90.70)
0.85 0.92 (0.33-2.50)
R. B
. Coelho et al.
Table 2. Clinical findings and anamnesis data of the discovery sample
Parameters Healthy
[N=129; n (%)]
Peri-implantitis
[N=86; n (%)] p value Odds ratio (CI)
General medical condition
Diabetes 2 (1.56) 5 (5.81) 0.08 3.92 (0.65-29.94)
Rheumatoid diseases 4 (3.12) 3 (3.48) 0.87 1.13 (0.19-6.17)
Osteoporosis 2 (1.56) 0 (0) 0.24 0.00 (0.00-6.14)
Hypotireoidism 6 (4.65) 2 (2.32) 0.37 0.49 (0.07-2.76)
Antimicrobials 0 0 (0) ---
NSAIDs† 1 (0.78) 0 (0) 0.41
SAIDs‡ 0 2 (2.32) 0.08 RR 2.54 (2.15-2.99)
Hormone reposition 4 (3.12) 3 (3.48) 0.87 1.13 (0.19-6.17)
Antidepressant 4 (3.12) 3 (3.48) 0.87 1.13 (0.19-6.17)
Clinical measurements
Edentulism
Total 24 (18.60) 69 (80.23)
<0.0001 18.69 (8.79-40.34)
Partial 104 (80.62) 16 (18.60)
Periodontal phenotype
Thin# 48 (37.20) 44 (51,16)
0.04 1.77 (0.98-3.20)
Thick 79 (61.24) 41 (47.67)
NSAIDs: Nonsteroidal anti-inflammatory drugs. SAIDs: Steroidal anti-inflammatory drugs.
Table 3. Peri-implant status of the volunteers
Peri-implant status Healthy
[N=129; n (%)]
Peri-implantitis
[N=86; n (%)] p value Odds ratio (CI)
Bleeding on probing 19 (14.73) 52 (60.46) <0.001 8.55 (4.41-17.96)
Spontaneous bleeding 0 (0.0) 11 (12.79) <0.001 RR 2.72 (2.27-3.26)
Probing pocket depth (mm) 1.86±0.99 2.46±1.26 <0.001
Plaque index 11 (8.52) 22 (25.58) <0.001 3.69 (1.58-8.72)
Peri-implant phenotype
Thin 50 (38.75) 43 (50,00)
0.07
1.65 (0.91-2.99)
Thick 79 (61.25) 43 (50.00)
Mobility (absence/ presence) 0 8 (9.30) <0.001 RR 2.65 (2.23-3.16)
Implant-platform type
External Hexagon 66 (51.16) 50 (58.13)
0.43
Internal Hexagon 10 (7.75) 8 (9.30)
Morse Cone 50 (38.76) 27 (31.39)
Others 5 (3.87) 1 (1.16)
Implant region
Superior 63 (48.83) 43 (50.00)
0.78 0.93 (0.52-1.66)
Inferior 68 (52.71) 43 (50.00)
Osseointegration
period (months) 33.95±31.07 35.85±37.42 0.70
---BMP4 and FGF haplotypes in peri-implantitis
poor oral hygiene, diabetes and history of periodontal diseases (18). Therefore, with the purpose of better understanding the correlation between risk factors and the development of peri-implantitis, this study included smokers and subjects with systemic disease, showing that there is a correlation between diabetes with the development of peri-implantitis. However, it was not evident that smoking is a possible risk indicator for peri-implantitis, which may indicate that other non-described factors may trigger peri-implant bone loss.
Although no studies in the literature discuss the importance of the correlation between BMP and FGF in implant dentistry, the whole process of bone formation, bone repair and homeostasis is widely mediated by these soluble factors associated with their receptors (19).
In an endeavor to elucidate the relationship between the genetic factor and peri-implantitis, genotypes and allele frequencies in specific regions in BMP4, FGF3, FGF10 and FGFR1 genes were evaluated using the data from the International HapMap Project (20). The results of the single marker showed that BMP4 rs2761884 (G>T) and FGF3 rs4631909 (C>T) substitutions were associated with absence of peri-implantitis and there seems to be no major influence of the other gene encoding mediator in bone remodeling. However, when different regions in the same gene were analyzed together, in an endeavor to understand the effect of the different regions responsible for the expression of the gene, an association among GGGA and GAAA BMP4, TGCG FGF3 and CCTG FGF10 haplotypes with pathologic peri-implant bone loss was clearly observed.
The study of Ren et al. (21) found that BMP4 TGGGCTT
Table 4. Characteristics of genetic markers
Gene SNP ID Chromosome SNP Type Base change* MAF**
BMP4
rs2761884 Ch14 intron G>T 0.366
rs17563 Ch14 exon C>T 0.371
rs2071047 Ch14 intron A>G 0.401
rs762642 Ch14 intron A>C 0.352
FGF3
rs7932320 Ch11 intron C>T 0.486
rs1893047 Ch11 intron A>G 0.428
rs4631909 Ch11 intergenic C>T 0.478
rs4980700 Ch11 intergenic A>G 0.439
FGF10
rs1448037 Ch5 intron C>T 0.361
rs900379 Ch5 intron C>T 0.483
rs1011814 Ch5 intron C>T 0.485
rs593307 Ch5 intron A>G 0.364
FGFR1 rs13317 Ch8 3`UTR C>T 0.275
*Base change according to Applied Biosystem; **MAF: minor allele frequency according to GenBank; Base change in BMP4 rs 17563 had as reference the Esembl.
haplotype was associated with the overexpression of BMP4 mRNA in subjects with ossification of the posterior longitudinal ligament. Guimaraes et al. (8) analyzed multiple variants in the BMP4 gene and believe that the BMP4 haplotype may trigger the exacerbated expression of BMP4, interfering in bone homeostasis and causing the non-union of fractures.
The antagonism between BMP and FGF contributes to the repair of bone lesions. FGF/FGFR signaling is present in endochondral and intramembranous bones and plays important roles in regulating their development and growth (22). In the present study, it is clear that individuals carrying CC genotype for FGF3 rs4631909 have increased risk of peri-implantitis, in contrast to the TT genotype, which acts as a protective factor even when analyzed together with other areas of the same chromosome. Furthermore, it is important to consider that FGFR1 is a positive regulator for bone formation: it has a synergistic effect with FGFR2 and stimulates osteoblastic differentiation (23). FGFR1 and FGFR2 are important in regulating the morphology and patent of craniofacial sutures (24). However, in this study, FGFR1 rs13317 was not associated with peri-implantitis. It is possible that the variant of FGFR1 favors adequate bone repair.
R. B
. Coelho et al.
Table 5. Genotype frequencies of the SNPs in all volunteers
Gene SNP Genotypes Healthy
[N=129; n (%)]
Peri-implantitis
[N=86; n (%)] p value*
BMP4
rs2761884
GG 48 (37.20) 36 (41.86)
0.01
GT 55 (42.64) 41 (47.67)
TT 23 (17.83) 4 (4.65)
GT+TT 78 (60.46) 45 (52.32) 0.36
rs17563
AA 27 (20.93) 22 (25.58)
0.63
AG 64 (49.61) 40 (46.51)
GG 34 (26.36) 19 (22.09)
AG+GG 59 (45.73) 98 (113.95) 0.55
rs2071047
AA 19 (14.73) 9 (10.46)
0.15
AG 48 (37.21) 44 (51.16)
GG 50 (38.76) 27 (31.39)
AG+GG 69 (53.49) 36 (41.86) 0.32
rs762642
AA 105 (81.39) 72 (83.72)
0.46
AC 18 (13.95) 9 (10.46)
CC 0 0
AC+CC 18 (13.95) 9 (10.46)
---FGF-3
rs7932320
CC 24 (18.60) 18 (20.93)
0.47
CT 51 (39.53) 38 (44.18)
TT 52 (40.31) 27 (31.39)
CT+TT 103 (79.84) 65 (75.58) 0.62
rs1893047
AA 39 (30.23) 18 (20.93)
0.27
AG 56 (43.41) 40 (46.51)
GG 31 (24.03) 26 (30.23)
AG+GG 87 (67.44) 66 (76.74) 0.12
rs4631909
CC 20 (15.50) 23 (26.74)
0.05
CT 56 (43.41) 38 (44.18)
TT 53 (41.08) 24 (27.90)
CT+TT 109 (84.49) 62 (72.09) 0.04
rs4980700
AA 43 (33.33) 19 (22.09)
0.25
AG 56 (43.41) 37 (43.02)
GG 29 (22.48) 24 (27.90)
AG+GG 85 (65.89) 61 (70.93) 0.13
FGF-10
rs1448037
CC 46 (35.65) 37 (43.02)
0.32
CT 61 (47.28) 36 (41.86)
TT 21 (16.28) 9 (10.46)
CT+TT 67 (51.93) 46 (53.48) 0.59
rs900379
CC 30 (23.25) 24 (27.90)
0.36
CT 63 (48.84) 33 (38.37)
TT 35 (21.13) 27 (31.39)
CT+TT 98 (75.97) 60 (69.76) 0.4
rs1011814
CC 36 (27.90) 26 (30.23)
0.73
CT 61 (47.28) 34 (39.53)
TT 29 (22.48) 19 (22.09)
CT+TT 90 (67.77) 53 (61.62) 0.51
rs593307
AA 24 (18.60) 12 (13.95)
0.29
AG 59 (45.73) 32 (37.20)
GG 39 (30.23) 33 (38.37)
AG+GG 63 (48.83) 45 (52.32) 0.35
FGFR1 rs13317
CC 5 (3.87) 5 (5.81)
0.13
CT 39 (30.23) 35 (40.70)
TT 83 (64.34) 42 (48.83)
BMP4 and FGF haplotypes in peri-implantitis
Table 6. Allele distribution between groups
Gene SNP ID A1 MAF Control MAF Test p value Odds ratio 95% CI
BMP4
rs2761884 T 0.40 0.30 0.13 1.56 0.83-2.92
rs17563 A 0.47 0.52 0.47 0.82 0.45-1.48
rs2071047 A 0.37 0.39 0.77 1.09 0.59-2.01
rs762642 C 0.07 0.06 0.77 0.85 0.24-2.94
FGF3
rs7932320 C 0.39 0.45 0.39 1.28 0.70-2.34
rs1893047 A 0.53 0.45 0.25 0.73 0.40-1.31
rs4631909 C 0.37 0.49 0.08 1.64 0.90-2.99
rs4980700 G 0.45 0.53 0.25 1.38 0.76-2.50
FGF10
rs1448037 T 0.40 0.33 0.63 0.87 0.46-1.63
rs900379 T 0.47 0.46 0.89 0.96 0.54-1.70
rs1011814 C 0.47 0.51 0.57 1.17 0.65-2.12
rs593307 A 0.44 0.36 0.24 0.72 0.39-1.31
FGFR1 rs13317 C 0.19 0.27 0.17 1.58 0.77-3.24
A1: Minor allele (based on whole sample)/ MAF_C: minor allele frequency in control/ MAF_T: minor allele frequency in test. *p calculated by chi-square test. p-values <0.05 are considered significant
Table 7. Haplotypes distributions in all groups
Gene Haplotype
Frequency estimation
p value Odds Ratio 95% CI
Healthy [N=129; n (%)]
Peri-implantitis [N=86; n (%)]
BMP4
(rs2761884; rs17563; rs2071047; rs762642)
TGGA 0.36 0.25 --- 1.00
---GAAA 0.29 0.32 0.05 0.57 0.32-1.01
GGGA 0.15 0.22 0.02 0.48 0.25-0.91
GAGA 0.07 0.09 0.11 0.52 0.23-1.15
GAAC 0.05 0.04 0.81 1.14 0.41-3.11
TAGA 0.03 0.03 0.58 0.72 0.22-2.36
FGF3
(rs7932320; rs1893047; rs4631909; rs4980700)
TATA 0.52 0.43 --- 1.00
---CGCG 0.30 0.36 0.11 0.71 0.46-1.08
TGCG 0.05 0.10 0.05 0.48 0.23-1.01
CGTG 0.07 0.04 0.58 1.28 0.54-3.04
TGTA 0.01 0.01 0.93 1.09 0.15-8.02
FGF10
(rs1448037; rs900379; rs1011814; rs593307)
CTCG
0.50 0.49 --- 1.00
---TCTA 0.39 0.31 0.33 1.24 0.81-1.89
CCTG 0.05 0.13 0.03 0.46 0.23-0.94
CTCA 0.01 0.01 0.73 1.43 0.19-10.91
CCTA 0.01 0.01 0.98 0.96 0.08-11.09
R. B
. Coelho et al.
of peri-implantitis, and thus provide new approaches to molecular-level investigations when treating these volunteers, increasing the rate of success in implant rehabilitation.
Resumo
Apesar do alto índice de sucesso em implantodontia, falhas tem aumentado drasticamente, estando associadas ao desenvolvimento de peri-implantite. A perda óssea peri-implantar é influenciada por múltiplos fatores, incluindo causas genéticas e ambientais. As BMPs (proteínas ósseas morfogenéticas) são fatores de crescimento indutores da formação óssea. Os FGFs (fatores de crescimento dos fibroblastos) e seus receptores (FGFRs) desenvolvem importante função na proliferação, diferenciação e migração celular. A relação BMP/FGF é responsável pela regeneração e perda óssea. O objetivo deste estudo foi estudar a possível correlação entre os genes BMP4, FGF3, FGF10 e FGFR1 e a perda óssea peri-implantar. Duzentos e quinze voluntários, com 754 implantes, foram submetidos ao exame oral e divididos em grupo saúde (n=129) e peri-implantite (n=86). Treze polimorfismos nos genes BMP4, FGF3, FGF10 e FGFR1 foram analisados individualmente e como haplótipos. O teste do qui-quadrado correlacionou as frequências dos genótipos, alelos e haplótipos. Valores de p<0,05 foram considerados estatisticamente significantes. Voluntários com peri-implantite mostraram alta incidência de edentulismo total (p<0,0001) e biotipo periodontal fino (p<0,04). Sangramento espontâneo, placa e mobilidade do implante foram altamente incidentes no grupo peri-implantite (p<0,0001). O genótipo polimórfico TT para BMP4 rs2761884 foi associado com saúde peri-implantar (p=0,01). FGF3 rs4631909 (genótipos TT+CT) mostraram associação com o grupo controle (p=0,04). A frequência do alelo C para FGF3 rs4631909 mostrou uma tendência de associação com peri-implantite (p=0,08). Os haplótipos FGF10 CCTG (p=0,03), BMP4 GAAA (p=0,05) e GGGA (p=0,02) foram associados com peri-implantite (p=0,03). Sendo assim, conclui-se que os haplótipos BMP4 e FGF10 estão associados com peri-implantite.
Acknowledgements
The authors gratefully thank the assistance of all Department of Oral Biology at the University of Pittsburgh and Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro.
References
1. Ferreira SD, Silva GLM, Cortelli JR. Costa FO. Prevalence and risk variables
for peri-implant disease in Brazilian volunteers. J Clin Periodontol 2006;33:929-935.
2. Lindhe J, Meyle J. Peri-implant diseases: Consensus Report of the Sixth
European Workshop on Periodontology. J Clin Periodontol 2008;35.
3. Lee S, Kim JY, Hwang J, Kim S, Lee JH, Han HD. Investigation of
pathogenic genes in peri-implantitis from implant cluster failure volunteers: a whole-exome sequence pilot study. PLOS ONE
2014;9:e99360.
4. Casado PL, Otazu IB, Balduino A, Mello W, Barboza EP, Duarte MEL.
Identification of periodontal pathogens in healthy periimplant sites. Implant Dent 2011;20:226-235.
5. Borreli J Jr, Pape C, Hak D, Hsu J, Lin S, Giannoudis P, et al.. Physiological
challenges of bone repair. J Orthop Trauma 2012;26:708-711.
6. Parithimarkalaignan S, Padmanabhan TV. Osseointegration: An upadate.
J Indian Protsthodont Soc 2013;13:2-6.
7. Reddi AH, Reddi A. Bone morphogenetic proteins (BMPs): from
morphogens to metabologens. Cytokine Growth Factor Rev 2009;20:341-342.
8. Guimarães JM, Guimarães IC, Duarte ME, Vieira ME, Vieira T, Vianna
VF, et al.. Polymorphisms in BMP4 and FGFR1 gene are associated with fracture non-union. J Orthop Res 2013;31:1971-1979.
9. Anderson HC, Hodges PT. Aguilera XM. Bone morphogenetic protein
(BMP) localization in developing human and rat growth plate. Metaphysis, epiphysis and articular cartilage. J Histochem Cytochem 2000;48:1493-1502.
10. Ramesh BL, Wilson SG, Dick IM, Islam FM, Devine A, Prince RL. Bone mass effects of a BMP4 gene polymorphism in postmenopausal women. Bone 2005;36:555-561.
11. Johnson DE, Williams LT. Structural and functional diversity in the FGF receptor multigene family. Adv Cancer Res 1993;60:1-41.
12. Marie PJ. Fibroblast growth factor signaling controlling osteoblast differentiation. Gene; 2003;316:23-32.
13. Patel ZS, Young S, Tabata Y, Jansen JA, Wong ME, Mikos AG. Dual delivery of an angiogenic and an osteogenic growth factor for bone regeneration in a critical size defect model. Bone 2008;43:931-940 14. Junior RG, Pinheiro AR, Schoichet JJ, Nunes CHR, Gonçalves R,
Bonato LL, et al.. MMP13, TIMP2 and TGFB3 gene polymorphisms in Brazilian chronic periodontitis and periimplantitis subjects. Braz Dent J 2016;27:128-134.
15. Küchler EC, Tannure PN, Falagan-Lotsch P, Lopes TS, Granjeiro JM, Amorim LMF. Buccal cells DNA extraction to obtain high quality human genomic DNA suitable for polymorphism genotyping by PCR-RFLP and Real-Time PCR. J Appl Oral Sci 2012;20:467-471.
16. Little J, Higgins JP, Ioannidis JP, Moher D, Gagnon F, von Elm E, et al.. STrengthening the REporting of Genetic Association studies (STREGA) -an extension of the STROBE statement. Eur J Clin Invest 2009;39:247-266.
17. Casado PL, Villas-Boas R, de Mello W, Duarte ME, Granjeiro JM. Peri-implant disease and chronic periodontitis: is interleukin-6 gene promoter polymorphism the common risk factor in a Brazilian population? Int J Oral Maxillofac Implants 2013;28;35-43.
18. Casado PL, Pereira MC, Duarte MEL, Granjeiro JM. History of chronic periodontitis is a high risk factor for peri-implant disease. Braz Dent J 2013;24:134-141.
19. Coutu DL, François M, Galipeau J. Inhibition of cellular senescence by developmentally regulated FGF receptors in mesenchymal stem cells. Blood 2011;117:6801-6812.
20. The international HapMap Consortium. The international HapMap project. Nature 2003;426:789-796.
21. Ren Y, Feng J, Liu ZZ, Wan H, Li JH, Lin X. A new haplotype in BMP4 implicated in ossification of the posterior longitudinal ligament (OPLL) in a Chinese population. J Orthop Res 2012;30:748-756.
22. Nie X, Luukko K, Kettunen P. FGF signalling in craniofacial development and developmental disorders. Oral Dis 2006;12:102-111.
23. Hajihosseini MK, Lalioti MD, Arthaud S, Burgar HR, Brown JM, Twigg SR , et al.. Skeletal development is regulated by fibroblast growth factor receptor 1 signalling dynamics. Development 2004;131:325-335. 24. Johnson D, Iseki S, Wilkie AO, Morriss-Kay GM. Expression patterns
of Twist and Fgfr1, 2 and 3 in the developing mouse coronal suture suggest a key role for twist in suture initiation and biogenesis. Mech Dev 2000;91:341-345.
Received March 16, 2016 Accepted May 30, 2016
Table 8. Multinominal logistic regression results for the diseased groups (reference = peri-implantitis group)
Parameters Z p-value Odds Ratio 95% CI
Age -2.58 0.01 0.96 0.94-0.99
BMP4 rs2761884
GG --- 1.0 ---
GT 0.36 0.71 1.11 0.60-2.06
TT 2.61 0.009 4.40 1.44-13.43
FGF3
CG --- 1.0 ---
CT 1.45 0.14 1.75 0.82-3.75