Rev. Soc. Bras. Med. Trop. vol.46 número4
Texto
Imagem
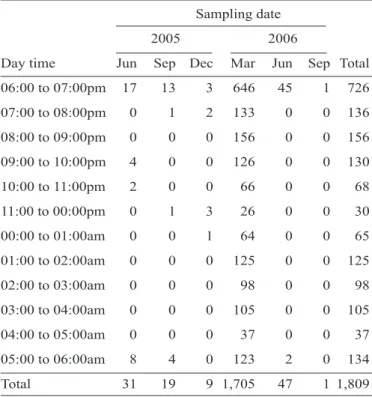
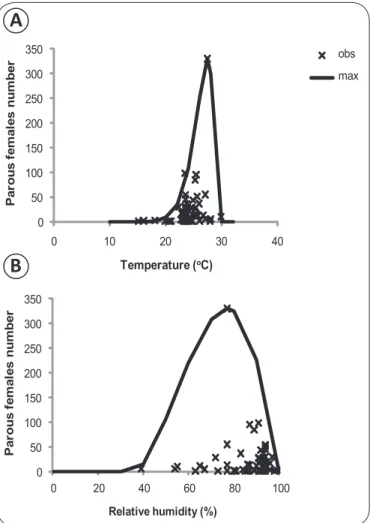
Documentos relacionados
É importante destacar que as práticas de Gestão do Conhecimento (GC) precisam ser vistas pelos gestores como mecanismos para auxiliá-los a alcançar suas metas
Ao Dr Oliver Duenisch pelos contatos feitos e orientação de língua estrangeira Ao Dr Agenor Maccari pela ajuda na viabilização da área do experimento de campo Ao Dr Rudi Arno
Neste trabalho o objetivo central foi a ampliação e adequação do procedimento e programa computacional baseado no programa comercial MSC.PATRAN, para a geração automática de modelos
Ousasse apontar algumas hipóteses para a solução desse problema público a partir do exposto dos autores usados como base para fundamentação teórica, da análise dos dados
In an earlier work 关16兴, restricted to the zero surface tension limit, and which completely ignored the effects of viscous and magnetic stresses, we have found theoretical
The decrease of HRV values during music listening doesn’t clearly mean that music decreases HRV, because of the different time period used in both moments
The probability of attending school four our group of interest in this region increased by 6.5 percentage points after the expansion of the Bolsa Família program in 2007 and
Como consequência da delimitação mais clara do episódio narrado, uma das características fundamentais dos lais é a de apresentar uma concentração dramática maior. Além dos