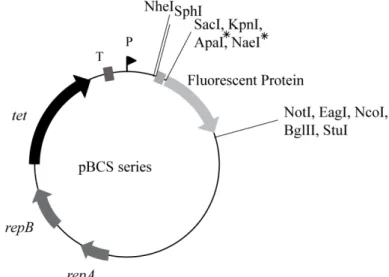
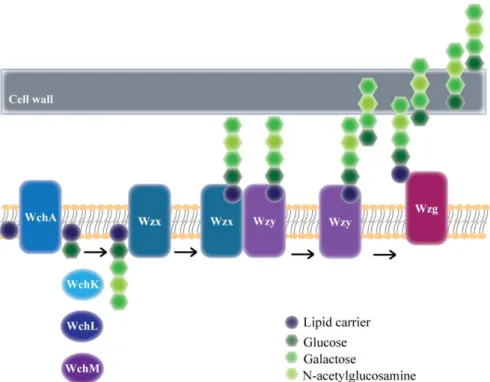
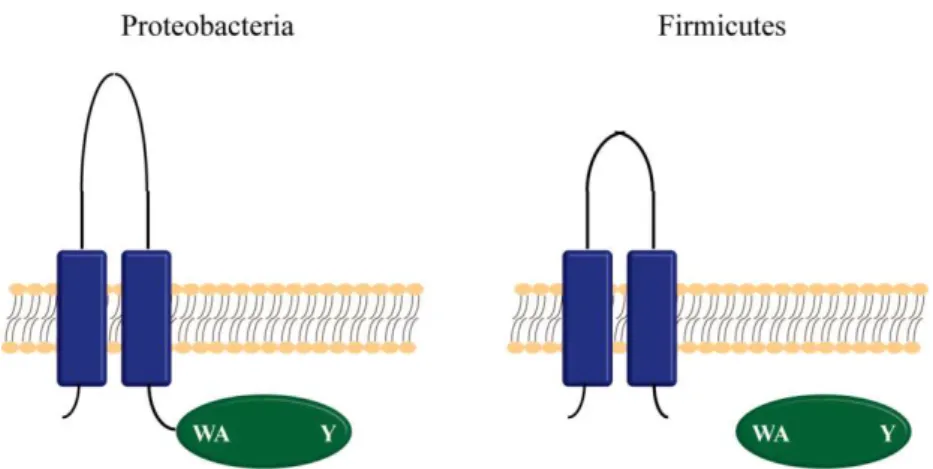
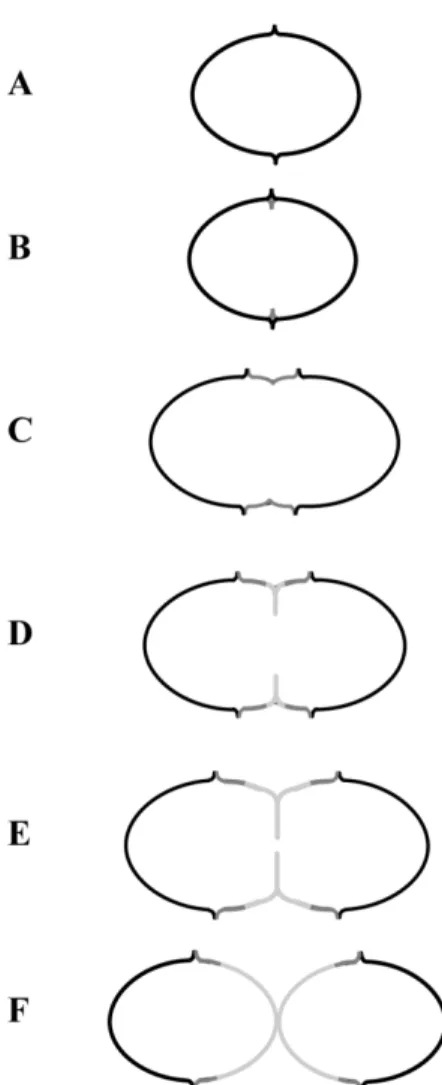
PhD Thesis Mafalda S. Henriques
Texto
Imagem
Documentos relacionados
Desenvolvemos um Sistema de Apoio à Decisão para o Conselho Executivo da Escola que integra: (1) um sistema de classificação automática de alunos com base em parâmetros
This log must identify the roles of any sub-investigator and the person(s) who will be delegated other study- related tasks; such as CRF/EDC entry. Any changes to
Nota-se que há poucos artigos encontrados na biblioteca virtual em saúde, e de modo geral não há pesquisas consideráveis que buscam quantificar os resultados obtidos com
Fisi amente, dois triângulos são semelhantes se pudermos dilatar e/ou girar.. Por tal razão, as mesmas são
Esta intervenção foi comparada com a intervenção usual praticada na instituição implicada (grupo de controlo - GC). Foram encontrados resultados estatisticamente
Material e Método Foram entrevistadas 413 pessoas do Município de Santa Maria, Estado do Rio Grande do Sul, Brasil, sobre o consumo de medicamentos no último mês.. Resultados
Foi elaborado e validado um questionário denominado QURMA, específico para esta pesquisa, em que constam: a) dados de identificação (sexo, idade, profissão, renda familiar,
São objetivos: analisar a atenção à saúde da pessoa com amputação na perspectiva dessas e dos enfermeiros; compreender como as políticas públicas dispõem e ordenam