www.bjorl.org
Brazilian
Journal
of
OTORHINOLARYNGOLOGY
ORIGINAL
ARTICLE
Specific
immediate
early
gene
expression
induced
by
high
doses
of
salicylate
in
the
cochlear
nucleus
and
inferior
colliculus
of
the
rat
夽
Paula
Santos
a,b,
Lilian
Eslaine
Costa
Mendes
da
Silva
a,
Ricardo
Maurício
Leão
b,∗aUniversidadedeSãoPaulo(USP),FaculdadedeMedicinaRibeirãoPreto(FMRP),DepartamentodeOftalmologiae
OtorrinolaringologiaeCirurgiadeCabec¸aePescoc¸o,RibeirãoPreto,SP,Brazil
bUniversidadedeSãoPaulo(USP),FaculdadedeMedicinaRibeirãoPreto(FMRP),DepartamentodeFisiologia,RibeirãoPreto,SP,
Brazil
Received29April2015;accepted16February2016 Availableonline25April2016
KEYWORDS
Salicylate; Tinnitus;
Cochlearnucleus; Inferiorcolliculus
Abstract
Introduction:Salicylateathighdosesinducestinnitus inhumansandexperimentalanimals. However,themechanismsandlociofactionofsalicylateininducingtinnitusarestillnotwell known. TheexpressionofImmediateEarly Genes(IEG)istraditionallyassociated with long-termneuronalmodificationsbutitisstillnotclearhowandwhereIEGsareactivatedinanimal modelsoftinnitus.
Objectives: Herewe investigatedtheexpressionofc-fosandEgr-1,two IEGs,intheDorsal CochlearNucleus(DCN),theInferiorColliculus(IC),andthePosteriorVentralCochlearNucleus (pVCN)ofrats.
Methods:Ratsweretreatedwithdosesknowntoinducetinnitusinrats(300mg/kgi.p.daily, for 3 days), andc-fos andEgr-1 protein expressions were analyzed usingwestern blot and immunocytochemistry.
Results:Afteradministrationofsalicylate,c-fosproteinexpressionincreasedsignificantlyin theDCN,pVCNandICwhenassayedbywesternblot.Immunohistochemistrystainingshoweda moreintenselabelingofc-fosintheDCN,pVCNandICandasignificantincreaseinc-fospositive nucleiinthepVCNandIC.WedidnotdetectincreasedEgr-1expressioninanyoftheseareas.
Conclusion: OurdatashowthatahighdoseofsalicylateactivatesneuronsintheDCN,pVCN andIC.Theexpressionofthesegenesbyhighdosesofsalicylatestronglysuggeststhatplastic changesintheseareasareinvolvedinthegenesisoftinnitus.
© 2017 Associac¸˜ao Brasileira de Otorrinolaringologia e Cirurgia C´ervico-Facial. Published by Elsevier Editora Ltda. This is an open access article under the CC BY license (http:// creativecommons.org/licenses/by/4.0/).
夽
Pleasecitethisarticleas:SantosP,daSilvaLE,LeãoRM.Specificimmediateearlygeneexpressioninducedbyhighdosesofsalicylate inthecochlearnucleusandinferiorcolliculusoftherat.BrazJOtorhinolaryngol.2017;83:155---61.
∗Correspondingauthor.
E-mail:[email protected](R.M.Leão).
PeerReviewundertheresponsibilityofAssociac¸ãoBrasileiradeOtorrinolaringologiaeCirurgiaCérvico-Facial.
http://dx.doi.org/10.1016/j.bjorl.2016.02.011
PALAVRAS-CHAVE
Salicilato; Zumbido; Núcleococlear; Colículoinferior
Expressãoespecíficadegeneprecocesimediatosinduzidapordoseselevadas desalicilatononúcleococlearecolículoinferiorderato
Resumo
Introduc¸ão:Salicilatoemdoseselevadasinduzzumbidonossereshumanoseemanimais exper-imentais.Noentanto,osmecanismoselocideac¸ãodosalicilatonainduc¸ãodezumbidoainda não sãobemconhecidos.A expressãodosgenes precocesimediatos (GPIs)está tradicional-menteassociadaaalterac¸õesneuronaisemlongoprazo,masaindanãoestáclarocomoeonde osGPIssãoativadosemmodelosanimaisdezumbido.
Objetivos: Nopresenteestudoinvestigamosaexpressãodec-foseEgr-1,doisGPIs,nonúcleo cocleardorsal(NCD),colículoinferior(CI)enúcleococlearventralposterior(NCVp)deratos.
Métodos: Osratosforamtratadoscomdosesque,conhecidamente,induzemzumbidoemratos (300mg/kgIP/dia,portrêsdias)easexpressõesdasproteínasc-foseEgr-1foramanalisadas pormeiodeWesternbloteimunoistoquímica.
Resultados: Após a administrac¸ão de salicilato, a expressão da proteína c-fos aumentou significativamentenoNCD,NCVpeCI,quandoanalisadosporWesternblot.Acolorac¸ão imunois-toquímicamostrou uma marcac¸ão maisintensa de c-fosnoNCD, NCVp eCI eum aumento significativodenúcleospositivosdec-fosnoNCVpeCI.Nãodetectamosaumentodaexpressão deEgr-1emnenhumadessasáreas.
Conclusão:NossosdadosmostramqueumadosealtadesalicilatoativaneurôniosnoNCD,NCVp eCI.Aexpressãodessesgenespordosesaltasdesalicilatosugerequeasalterac¸õesplásticas nessasáreasestãoenvolvidasnagênesedozumbido.
© 2017 Associac¸˜ao Brasileira de Otorrinolaringologia e Cirurgia C´ervico-Facial. Publicado por Elsevier Editora Ltda. Este ´e um artigo Open Access sob uma licenc¸a CC BY (http:// creativecommons.org/licenses/by/4.0/).
Introduction
Tinnitusisaphantomsoundsensationthatcanbethe conse-quenceofseveralfactorsincludingacoustictrauma,drugs, temporal mandibular disorders or deafness.1 The
mech-anisms of tinnitus induction and maintenance are still a matterofdebate,especiallysincetinnituscanresultfrom severaldifferentconditions.Highdosesofsalicylateinduce tinnitusinhumans,andatdosesof150---400mg/kgitinduces behavioralsymptomsoftinnitusinexperimentalanimalsin lessthan24h; thiseffectsubsideswithin72hafter treat-mentinterruption.2
Despite the common use of salicylate as a tinnitus-inducingagent,itsmechanismsandlociofaction arestill obscure. In vivo imaging experimentsin rats have shown thathigh doses ofsalicylate induces hyperactivity in spe-cific auditory areas, including the Inferior Colliculus (IC), the Dorsal Cochlear Nucleus (DCN) and the Auditory Cor-tex (AC), but not the Ventral Cochlear Nucleus (VCN).3,4
On the other hand, studies of expression of the immedi-ateearlygenec-fosshowedlessconsistentresults.Asingle dose of salicylate (350mg/kg) increased c-fos expression only in theAC of gerbils.5 Aprevious study didnot show
c-fos expression in the auditory brainstem after a single dose of salicylate, but only in non-auditoryareas such as thelocuscoeruleusandperiaqueductalgrayarea.6Another
studyobservedadecreaseof c-fosexpression intheICof gerbils.7Anotherstudyusingchronictreatmentwith
salicy-late(250mg/mL)showedincreasedc-fosexpressiononlyin theICandnotintheDCN.8Mostofthesedataare
inconsis-tentwiththeobservationsthattinnitusinducedbysalicylate
activatesextralemniscalauditorypathways,especiallythe DCN.2,3
Theexpression ofImmediateEarlyGenes(IEGs)is con-sideredamarkerofincreasedbrainactivityinresponseto diverse stimuli.Thesegenesaretranscription factorsthat trigger theexpressionofother genesresponsiblefor long-term changesin neurons. The expression of the IEG c-fos is a commonly usedmarker of neuronal activity and it is quicklyupregulatedafterneuronalstimulation.9---12TheIEG
Egr-1 is activated in response to neuronal calcium influx andpromotesfunctionalandstructuralchangesinneurons, includingintheauditorysystem.13,14
Inthisstudy,weaimedtoinvestigatetheactivation of c-fos andEgr-1intheDCNand ICin auditorypathways of rats subjected to a protocol of salicylate administration, whichiseffectiveininducingtinnitusinrats(3dailydosesof 300mg/kg).3DuetoitsproximitytotheDCNwealsostudied
theexpressionofthesegenesintheposteriordivisionofthe VentralCochlearNucleus(pVCN).
Methods
Animalsanddrugtreatment
All experimental procedures performed on animals were approvedbytheinstitution’sAnimalCareandUse Commit-tee(protocoln◦011/2013)andfollowedtheguidelinesand
cageandkept undera12 hourlight/darkcycle withfood andwateradlibitum.
Animals wereinjected i.p. with300mg/kg sodium sal-icylate (Sigma) (10L/g), dissolved in saline, for three consecutivedayswitha24hourintervalbetweeneachdose. The animals were then anesthetized with isoflurane and killedbydecapitationonthethirdday,3hafterdrug admin-istration.Controlgroupswereadministeredsalinefollowing thesameprotocol.Thisprotocolofsalicylateadministration hasbeendemonstratedtobeeffectiveininducingtinnitus inrats.3
DetectionofEgr-1andc-fosbywesternblotting
Forty animals were used for these experiments:20 were naïve animals and 20 were submitted to salicylate treat-ment. Slideswithfreshsections(90m)of thebrainstem containingthepVCN,DCNandICwereobtainedina cryo-stat.Toobtainatissuepunchofthedesiredareaweused a metalcylinder witha pistilof 0.5mm diameter. Dueto theirsmallsizeandcontiguouslocation,theDCNandpVCN wereextractedinthesamepunch.Onepunchwasobtained fromeachoffouranimalsandtheywerepooledtoincrease proteinyield.Thetissuewashomogenizedinalysisbuffer containing137mMNaCl,20mMTris,1%IgepalCA-630,10% glycerol, 2mM sodiumorthovanadate, 1% sodium dodecyl sulphate,50mMsodiumfluoride,2mMEDTA,and10% pro-teaseinhibitorcocktailatpH7.4.Tissuehomogenateswere centrifugedat15,000rpmfor10minat4◦C.Protein
concen-tration in tissues homogenates was determined using a modifiedLowryassay(DC ProteinAssay,Bio-Rad).Aliquots containing 90g protein were dissolved in loading buffer andheatedat95◦Cfor5min,andtheproteinsseparatedby
7.5%Tris---glycineSDS-PAGE(GEHealthcare-Bioscience)and transferredtoPVDFmembranes(AmershamBiosciences).
Immunoblots were blocked withalbumin 5% and incu-batedwith primaryantibodies at 4◦C.Primary antibodies
included antibodiesagainstc-fos andEgr-1(1:1000;Santa Cruz Biotechnology). After incubation, membrane was washedandincubatedwithsecondary antibody (1:10,000; ECLanti-RabbitIgG;GEHealthcare)for1hatroom temper-ature.Afterfinalwashes,labeledproteinsweredetectedby chemiluminescence(RPN2132;GEHealthcare).Forstripping andreprobing, themembranewassubmergedin stripping buffer(100mM20-mercaptoethanol,2%sodiumdodecyl sul-phate,62.5mMTris---HCl,pH6.8)at50◦C,for8min,washed
for90minundertapwater,rehydratedwithmethanoland washedwithTBS-Tbeforeblockingandreprobedwith pri-mary antibody against GAPDH (1:5000; Abcam). Western blotswerephotographedandquantifiedwithimageanalysis (MolecularImagingSystems).
c-fosImmunohistochemistry
Ten rats were used, 5 naïve and 5 submitted to salicy-latetreatment.Ratswereperfusedtranscardiallywith0.9% NaClfollowedby4%paraformaldehydein 0.1Mphosphate buffer (pH 7.4). The brain wasremoved, postfixed in the samesolutionfor1h,andcryoprotectedwith30%sucrose in0.1Mphosphatebufferfor2daysat4◦C.Thebrainswere
150
100
50
c-f
os/GAPDH
Naïve Salicylate
**
N
62 kDa c-fos
GAPDH 37 kDa
S N S N S N S N S
0
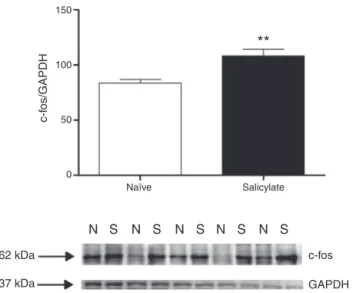
Figure1 c-fosexpressioninthe CN.Top. quantification of c-fosexpression.The c-fossignal intensitywasnormalizedto theGAPDH signal intensity(**p=0.0058;n=5for eachgroup containingmaterialfrom4animals).Westernblotsshowingthe immunoreactivityofc-fosandGAPDHinpunchescontainingthe CN(N,naïve;S,salicylate).
individuallysectioned(50m)inthetransverseplaneusing acryostat.
The sections werewashed in PBS(0.01M, pH 7.4)and incubated for 30min in PBS containing 1% hydrogen per-oxidetoinactivate endogenousperoxidases.Afterseveral rinsesinPBSfor30min,thesectionswereplacedin5% nor-malgoatserum(Vector)for1handincubatedfor48hat4◦C
withprimaryantic-fosantibodygeneratedinrabbits(1:100; Santa Cruz). After rinsedin PBS,the sections were incu-batedfor1.5hatroomtemperaturewithbiotinylatedgoat anti-rabbitIgG(1:150;Vector);then,subsequently,washed inPBSandplacedfor1.5hinavidin---biotinperoxidase com-plex(Vectastain,Vector).Theimmunolabelingwasrevealed by5---10minofincubationwith0.05%3,3′-diaminobenzidine
tetrachloride and 0.1% hydrogen peroxide. The sections weremountedongelatin-coatedslides,dehydrated,cleared withxyleneandcoverslippedwithEntellan®.
Dataanalysis
Westernblotdata arepresentedasthe ratioof c-fosand GAPDHsignalintensity.StainednucleiinICCsectionswere countedmanually.ElevensectionscontainingtheIC,9 con-tainingtheDCNand6containingthepVCNwerecountedfor eachanimal.Alldataareexpressedasmean±SD. Statisti-calanalysiswascarriedoutusingStudent’st-test.p<0.05 wasconsideredstatisticallysignificant.
Results
100
80
60
40
20
Naïve
c-f
os/GAPDH
N
62 kDa c-fos
GAPDH 37 kDa
S N S N S N S N S
Salicylate
**
0
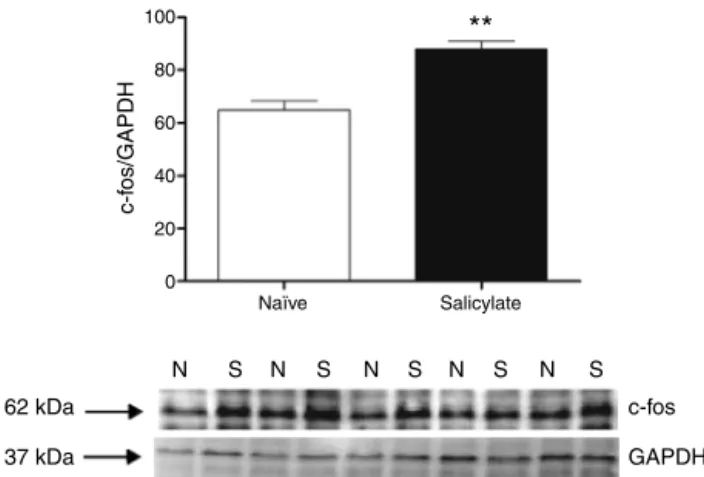
Figure2 c-fosexpressionintheIC.Topquantificationof c-fosexpression.Thec-fossignalintensitywasnormalizedtothe GAPDHsignalintensity(**p=0.0012;n=5foreachgroup con-taining material from 4animals). Western blotsshowing the immunoreactivityofc-fosandGAPDHinpunchescontainingthe IC(N,naïve;S,salicylate).
CN
IC
N S N S S
S S S N N
82 kDa EGR 1
EGR 1 82 kDa
Figure3 Westernblotsshowingtheimmunoreactivityof Egr-1inpunchescontainingtheDCN/pVCNandICfromnaïverats andaftersodiumsalicylatetreatment(N,naïve;S,salicylate).
proteinexpressionwasnotdetectedaftersalicylate treat-mentintheseregions(Fig.3).
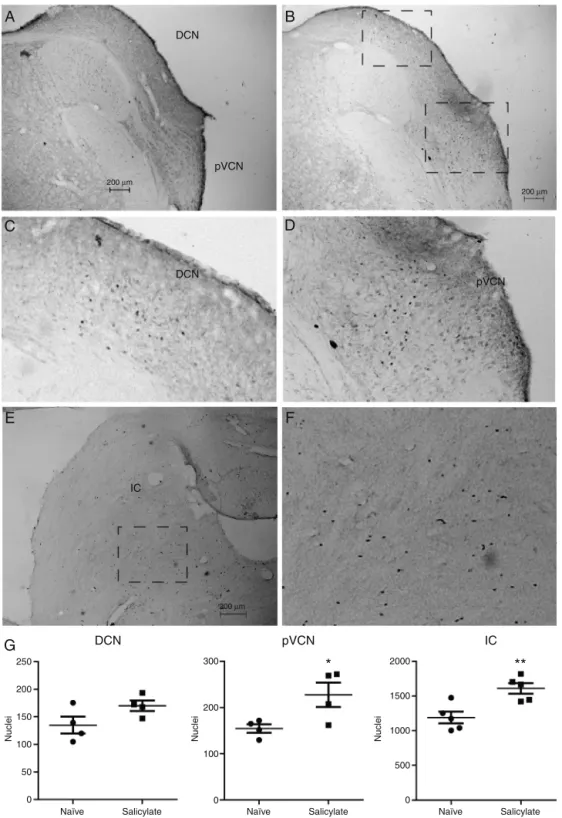
Because the location of the c-fos protein cannot be identifiedbyWesternBlot,weperformedc-fos immunocy-tochemistryin sectionscontainingtheDCN,pVCNandthe IC.Wedetectedc-fosinsectionsoftheDCN,pVCNandIC,as showninFig.4.Insectionsfromnaïveanimalsfaintnuclei stainingwasobservedinall3regions(Fig.4A).Insections fromsalicylate treatedanimalsweobservedmoreintense labelednucleiinDCN,pVCNandIC(Fig.4B).IntheDCNthis intenselyc-fos labelingwasobservedin themoreinternal layers,thedeeplayerandpossiblythefusiformlayer(Fig.4B andC),anditwasobservedmostlyinthemedial/dorsal sec-tionoftheDCN.Wedidnotobservelabelinginthemolecular layer.Wealsoobservedincreasedc-foslabelinginthepVCN adjacenttotheDCN(Fig.4BandD).IntheICweobservedan increaseintheimmunoreactivitymostlyinthecentralpart, ascanbeseeninFig.4EandF.Nolabelingwasobservedin thesuperiorolive(notshown).
Quantitative analysisof nucleiexpressing c-fosshowed a significant increase of c-fos positive nuclei in the pVCN(naïve:155±9.2nuclei;salicylate:228±26.4nuclei; p=0.04,n=4)andintheIC(naïve:1190±86nuclei; salicy-late:1611±79nuclei;p=0.0061;n=5),butnotintheDCN
(naïve:135±15nuclei;salicylate:170±9.5nuclei;p=0.1; n=4),ascanbeseeninFig.4G.
Discussion
Ourdatashowthat3daysoftreatmentwithhighdosesof salicylate increase the expression of the products of the IEG c-fos, but notof Egr-1, in pVCN, DCN andIC of rats. Although we observed increased c-fos protein levels and a more intense nuclei labeling in the IC, DCN and pVCN after salicylate, thenumber of stainednuclei was signifi-cantlyincreasedonlyinthepVCNandICandnotintheDCN. Thisdataisinaccordancewiththeobservedhyperactivity of the DCNand ICin rats submittedto asimilarprotocol of salicylate treatment3,15,16 and in other animal models
of tinnitus.17,18 On theother hand, we found asignificant
increaseinc-fosexpressionintheadjacentpVCN,aregion not usually related to tinnitus induction.3 This increased
expression ofc-fos inDCN,pVCNandICsuggests thatthe expression of theseIEGs promotes the expression of pro-teins thatcanchange thephysiological andmorphological propertiesoftheneuronsoftheseregions,whichcouldbe asubstratefortinnitusinduction.
Others4---7studiedc-fosexpressionafteracutesalicylate
treatmentusingimmunocytochemistry.Thosestudiesfound thatc-fosexpressionwasnotconsistentlyincreasedinthe DCNand IC.Ourresultsonthe otherhand show aclearly increasedexpression ofc-fos positivenuclei in theICand pVCN.Webelievethesediscrepanciescanbeattributedto thefactthatweusedyoungeranimalsandthatthosestudies usedasingledoseofsodiumsalicylate,insteadofthe3daily dosesusedbythisstudy.Consistentwiththishypothesis,a studyusing6daysoftreatmentwithsalicylatealsoobserved asignificantincreaseofc-fospositivenucleiintheIC,but notintheDCN.8
Inexperimental animals salicylate treatment increases the firing of neurons in both DCN and IC.15,16 The
mech-anisms of this effect are still unknown and could reflect a direct effect of salicylate on the neuronal excitabil-ity of these neurons or their incoming synapses, or a downstream effectof salicylate-induced geneexpression. Contrary toadirect excitatory effectof salicylate,it has beenshownthatdirectapplicationofsalicylateonDCN neu-ronsin brainslicesdecreases thespontaneousandevoked firing of DCN principal neurons.19,20 On the other hand,
directapplicationofsalicylateincreasesthefiringofmost IC neurons in vitro.21 Salicylate could also enhance the
excitability of DCN and IC neurons by decreasing their glycinergic inhibitory transmission.22 However, glycinergic
currentsinDCNfusiformneuronswerenotaffectedbyacute applications of millimolar concentrations of salicylate.20
Salicylate can also inhibit GABAergic receptors23 and
enhanceNMDAcurrents24whichcouldpotentiallydisturbthe
excitation-inhibition balance. Because most DCN neurons firespontaneously,25afiringincreaseproducedbysalicylate
A
C
E
G
B
D
F
DCNpVCN
DCN
IC
DCN pVCN IC
2000 300
250
200
150
100
50
Naïve Salicylate
Nuclei Nuclei Nuclei
Naïve Salicylate
*
Naïve Salicylate
**
0
200
100
0
1500
1000
500
0
pVCN
200 µm
200 µm
200 µm
Figure4 Detectionofc-fosbyimmunocytochemistryintheDCNandIC.(A)SectioncontainingtheDCNandposteriorVentral CochlearNucleus(pVCN)fromanaïveanimal.(B)SectioncontainingtheDCNandposteriorVentralCochlearNucleus(pVCN)from ananimaltreatedwithsalicylate.(C)Detailofthec-fosexpressingnucleiintheDCNfromthesquareareashownin(B).(D)Detailof thec-fosexpressingnucleiinthepVCNfromthesquareareashownin(B).(E)SectioncontainingtheICfromananimaltreatedwith salicylate.(F)Detailofthec-fosexpressingnucleiintheICfromthesquareareashownin(E).(G)Quantificationoftheexpression ofc-fosintheDCN,pVCNandIC[*p=0.04;**p=0.006;n=4(DCN,pVCN)and5(IC)].
thatsalicylateisactivatingitsneurons,andthatthisregion couldberelevantfortheperceptionoftinnitus.
Both c-fos and Egr-1 are expressed in the audi-tory pathways in response to sound and intracochlear stimulation.10,12,26,27 Interestingly, in the DCN c-fos
expression elicited by intracochlear stimulation was mostobservedinglycinergicinterneuronsinthemolecular layer.27Thisfindingisintriguingbecausetheseinterneurons
information. We only observed c-fos labeling near or in the deeplayer of the DCN in both control and salicylate conditions, suggesting that c-fos is expressed mainly by neuronscomputingtheauditoryinformation. Ontheother hand,wedidnotfindexpressionofEgr-1,aIEGthatislinked tothe formation of long-term plasticity13 after salicylate
treatment, suggesting thatsalicylate does nottrigger the expressionofgenesrelatedtolong-termsynapticplasticity. Interestingly, a previous study found a decrease in Egr-1 geneexpressionaftersalicylatetreatmentintheIC.28
There are similarities of tinnitus with neuropathic pain,29,30 suggesting thattheycouldshare similar
molecu-larmechanisms.Forinstance,patientswithtinnitusoften present withhyperacusis,and patients withchronic pain, hyperalgesia, both of which are enhanced reactions to normal stimuli intensities. Like tinnitus,31 chronic pain is
believed to be a result of long-term plastic changes in sensoryneurons andsynapses.32 TheIEGc-fosis also
acti-vated in the spinal cord in models of chronic pain and inflammation.33,34 Theirexpressionis postulatedtobethe
firstsignaloflong-termchangesintheseneuronswhichwill lead to chronic pain. Thus, it is likely that c-fos is acti-vatingsimilarcascades of geneexpression in tinnitusand chronic pain, leading tochanges that would underlie the symptomatologyoftheseconditions.
Conclusion
Ourdata shows that tinnitus-inducing doses of salicylate3
increasec-fos,but notEgr-1,expression inauditoryareas oftheauditorybrainstem,thedorsalcochlearnucleus,the posteroventralcochlearnucleusandinferiorcolliculusofthe rat.Thus,wepostulatethatc-fosisactivatingcascadesof geneexpressionintheseareasleadingtochangesthatcould underliethesymptomatologyoftinnitus.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
References
1.Holmes S, Padgham ND. More than ringing in the ears: a review of tinnitus and its psychosocialimpact. JClin Nurs. 2009;18:2927---37.
2.StolzbergD,SalviRJ,AllmanBL.Salicylatetoxicitymodelof tinnitus.FrontSystNeurosci.2012;6:28.
3.HoltAG,BissigD,MirzaN,RajahG,BerkowitzB.Evidenceof keytinnitus-relatedbrainregionsdocumentedbyaunique com-binationofmanganese-enhancedMRIandacousticstartlereflex testing.PLoSOne.2010;5:e14260.
4.PaulAK,LobarinasE,SimmonsR,WackD,LuisinJ,SpernyakJ, etal.Metabolicimagingofratbrainduring pharmacologically-inducedtinnitus.Neuroimage.2009;44:312---8.
5.Wallhäusser-Franke E, Mahlke C,Oliva R, BraunS, Wenz G, Langner G. Expression of c-fosin auditory and non-auditory brainregionsofthegerbilaftermanipulationsthatinduce tinn-itus.ExpBrainRes.2003;3153:649---54.
6.Wallhausser-Franke E. Salicylate evokes c-fos expression in the brain stem: implications for tinnitus. Neuroreport. 1997;8:725---8.
7.MahlkeC,Wallhäusser-FrankeE.Evidencefortinnitus-related plasticity in the auditory and limbic system, demon-strated byarg3.1and c-fosimmunocytochemistry. Hear Res. 2004;195:17---34.
8.Wu JL, Chiu TW, Poon PW. Differential changes in fos-immunoreactivity at the auditory brainstem after chronic injectionsofsalicylateinrats.HearRes.2003;176:80---93. 9.BarthAL,GerkinRC,DeanKL.Alterationofneuronalfiring
prop-ertiesafterinvivoexperienceinaFosGFPtransgenicmouse.J Neurosci.2004;24:6466---75.
10.FriaufE.Tonotopicorderintheadultanddevelopingauditory systemoftheratasshownbyc-fosimmunocytochemistry.Eur JNeurosci.1992;4:798---812.
11.SmeyneRJ,SchillingK,RobertsonL,LukD,OberdickJ,CurranT, etal.fos-lacZtransgenicmice:mappingsitesofgeneinduction inthecentralnervoussystem.Neuron.1992;8:13---23. 12.Nakamura M, Rosahl SK, Alkahlout E, Walter GF,Samii MM.
Electricalstimulation of thecochlearnerve in rats:analysis of c-fos expression in auditory brainstem nuclei. Brain Res. 2005;1031:39---55.
13.MaddoxSA,MonseyMS,SchafeGE.Earlygrowthresponsegene 1(Egr-1)isrequiredfornewandreactivatedfearmemoriesin thelateralamygdala.Learn.Mem.2011;18:24---38.
14.MelloCV,VelhoTA,PinaudR.Song-inducedgeneexpression:a windowonsongauditoryprocessingand perception.AnnNY AcadSci.2004;1016:263---81.
15.ChenGD,JastreboffPJ.Salicylate-inducedabnormalactivityin theinferiorcolliculusofrats.HearRes.1995;82:158---78. 16.JastreboffPJ,SasakiCT.Salicylate-inducedchangesin
sponta-neousactivityofsingleunitsin theinferior colliculusofthe guineapig.JAcoustSocAm.1986;80:1384---91.
17.Kaltenbach JA, ZacharekMA, Zhang J, Frederick S. Activity inthedorsal cochlearnucleus ofhamsters previously tested for tinnitus following intense tone exposure. Neurosci Lett. 2004;355:121---5.
18.Brozoski TJ, Bauer CA, Caspary DM. Elevated fusiform cell activityinthedorsalcochlearnucleusofchinchillaswith psy-chophysicalevidenceoftinnitus.JNeurosci.2002;22:2383---90. 19.BastaD,GoetzeR,ErnstA.Effectsofsalicylateapplicationon thespontaneousactivityinbrainslicesofthemousecochlear nucleus,medialgeniculatebodyandprimaryauditorycortex. HearRes.2008;240:42---51.
20.WeiL,DingD,SunW,Xu-FriedmanMA,SalviR.Effectsofsodium salicylateonspontaneousandevokedspikerateinthedorsal cochlearnucleus.HearRes.2010;267:54---60.
21.BastaD,ErnstA.Effectsofsalicylateonspontaneousactivityin inferiorcolliculusbrainslices.NeurosciRes.2004;50:237---43. 22.LuYG,TangZQ,YeZY,WangHT,HuangYN,ZhouKQ,etal.
Sal-icylate,anaspirinmetabolite,specificallyinhibitsthecurrent mediatedbyglycinereceptorscontainingalpha1-subunits.BrJ Pharmacol.2009;157:1514---22.
23.XuH,GongN,ChenL,XuTL.Sodiumsalicylatereducesgamma aminobutyricacid-inducedcurrentinratspinaldorsalhorn neu-rons.Neuroreport.2005;16:813---6.
24.RuelJ,CharbbertC,NouvianR,BendrisR,EybalinM,LegarCM, et al. Salicylate enables cochlear arachidonic-acid-sensitive NMDAreceptorresponses.JNeurosci.2008;28:7313---23. 25.LeaoRM,LiS,DoironB,TzounopoulosT.Diverselevelsofan
inwardly rectifying potassium conductance generate hetero-geneousneuronalbehaviorinapopulationofdorsalcochlear nucleuspyramidalneurons.JNeurophysiol.2012;107:3008---19. 26.Illing RB, Michler SA, Kraus KS, Laszig R. Transcription fac-tormodulation and expression intheratauditorybrainstem following electrical intracochlear stimulation. Exp Neurol. 2002;175:226---44.
alltypesofneuronsintheratauditorybrainstem.ExpNeurol. 2007;208:193---206.
28.Hu SS, Mei L, Chen JY, Huang ZW, Wu H. Expression of immediate-earlygenes intheinferiorcolliculusand auditory cortexinsalicylate-inducedtinnitusinrat. EurJHistochem. 2014;58:2294.
29.MollerAR.Tinnitusandpain.ProgBrainRes.2007;166:47---53. 30.TondorffJ.Theanalogybetweentinnitusandpain:a
sugges-tionfor a physiologicalbasis for chronic tinnitus. Hear Res. 1987;28:271---5.
31.KaltenbachJA,ZhangJ,FinlaysonP.Tinnitusasaplastic phe-nomenonand itspossible neuralunderpinnings inthedorsal cochlearnucleus.HearRes.2005;206:200---26.
32.Latremoliere A, Woolf CJ. Central sensitization:a generator of pain hypersensitivity bycentral neural plasticity. JPain. 2009;10:895---926.
33.Lantéri-Minet M, de Pommery J, Herdegen T, Weil-Fugazza J,Bravo R,Menétrey D.Differentialtimecourse andspatial expressionoffos.Jun,andKrox-24proteinsinspinalcordof ratsundergoingsubacute orchronicsomaticinflammation.J CompNeurol.1993;333:223---35.