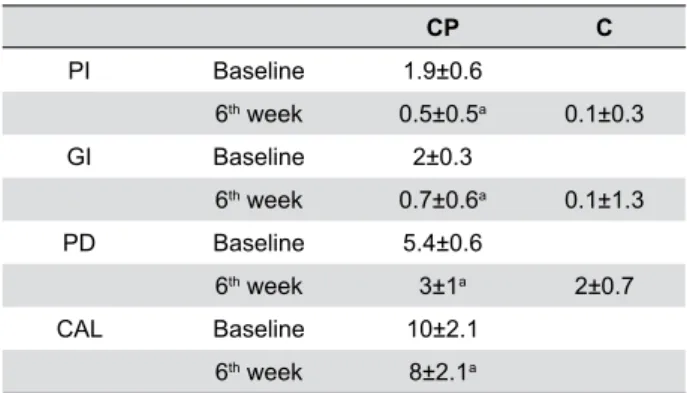
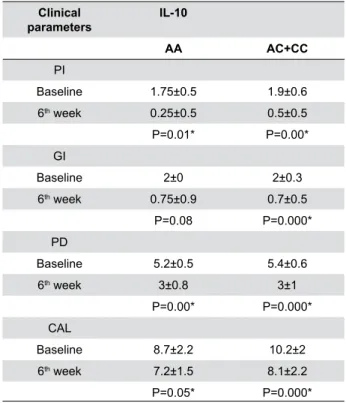
The effects of IL-10 gene polymorphism on serum, and gingival crevicular fluid levels of IL-6 and IL-10 in chronic periodontitis
Texto
Imagem
Documentos relacionados
É nesta mudança, abruptamente solicitada e muitas das vezes legislada, que nos vão impondo, neste contexto de sociedades sem emprego; a ordem para a flexibilização como
the equilibrium, increasing the methane and carbon monoxide conversion to more hydrogen. This is known as sorption-enhanced reaction process. The effluent stream of the SERP is
Este capítulo aborda a revisão de literatura em sete seções: primeira, os principais conceitos de logística e logística hospitalar para adequar à utilização do termo;
Professora Simey de Souza Leão Pereira Magnata cujo objetivo é apresentar para os alunos estratégias de ensino da Biologia evolutiva à luz do desenho universal da aprendizagem,
Estabelecer hum Juizo de Provedoria Mór da Saúde, para regular *s quarentenas , que devem fazer os Navios, que vem dos diverssos Por- tos , e os que trazem carregação de
A modalidade de alimentos abordada no presente artigo, disciplinados no atual Código Civil, refere-se possibilidade de requerer de alguém a prestação de alimentos nos
Para a análise das diferentes estruturas de articulação da cidade de Viana do Castelo com a sua frente de água temos como base duas reflexões centrais: o valor urbanístico (eixos
Como desempenho para representar os resultados de toda base, esta mesma arquitetura apresentou R² de 0,997 e RMSE de 2,165 kWh/m².ano, demonstrando como uma Rede Neural