Mat. Res. vol.6 número1
Texto
Imagem
Documentos relacionados
There was an interaction between sexes and sedentary behaviors (p < 0.001); therefore, re- gression analyses were performed separately for sex (Table 5). The crude analysis
Ao preço de NCz$ 0,18, que representava o valor de comercialização na época da pesquisa, o "coeficiente de eficiência" nos grupos analisados está de acordo com a Tabela 6.
Nos anos 2000 ocorreram mudanças na sociedade brasileira a partir da ação do sujeito político feminista no campo e sua interação com o Estado em transformação. O feminismo no
This study has shown that the resin-based materials (polyacid modified and conventional composite resins) provided better marginal adaptation to both deciduous and immature
Abstract: The purpose of this study was to evaluate the compressive strength of two glass ionomer cements, a conventional one (Vitro Fil - DFL) and a resin-modiied material (Vi- tro
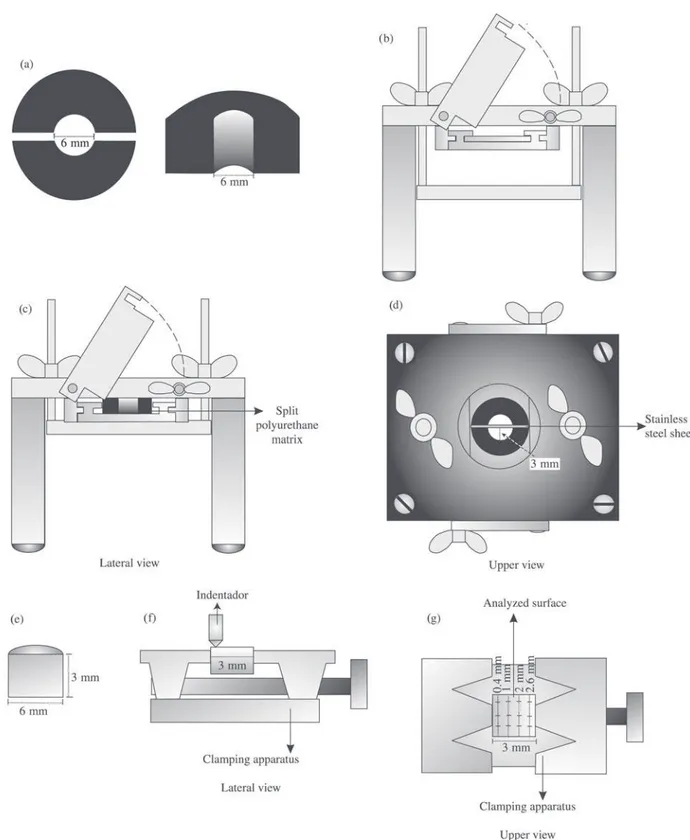
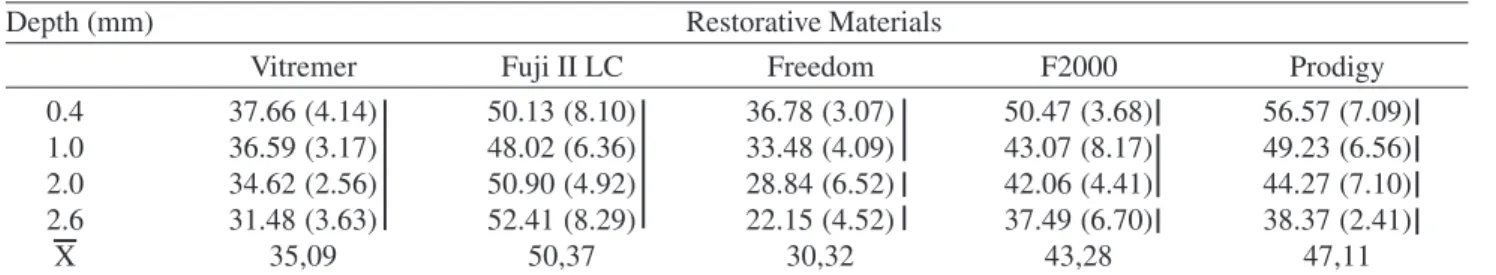
This study evaluated the surface microhardness and fluoride release of 5 restorative materials - Ketac-Fil Plus, Vitremer, Fuji II LC, Freedom and Fluorofil - in two storage
The aim of this study was to evaluate the microtensile bond strength (µTBS) of two substrates (enamel and dentin) considering two study factors: type of composite resin
The aim of this study was to evaluate the in vitro antibacterial and biofilm inhibition properties of glass ionomer restorative cements. Ketac Nano, Vitremer, Ketac Molar Easymix