r e v b r a s r e u m a t o l . 2017;57(4):286–293
ww w . r e u m a t o l o g i a . c o m . b r
REVISTA
BRASILEIRA
DE
REUMATOLOGIA
Original
article
Analysis
of
four
serum
biomarkers
in
rheumatoid
arthritis:
association
with
extra
articular
manifestations
in
patients
and
arthralgia
in
relatives
Flávia
R.
Nass
a,
Thelma
L.
Skare
b,
Isabela
Goeldner
a,
Renato
Nisihara
a,c,∗,
Iara
T.
Messias-Reason
a,
Shirley
R.R.
Utiyama
daUniversidadeFederaldoParaná,LaboratóriodeImunopatologia,Curitiba,PR,Brazil bHospitalUniversitárioEvangélicodeCuritiba,UnidadedeReumatologia,Curitiba,PR,Brazil cUniversidadePositivo,DepartamentodeMedicina,Curitiba,PR,Brazil
dUniversidadeFederaldoParaná,DepartamentodeAnálisesClínicas,Curitiba,PR,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory: Received17June2015 Accepted18November2015 Availableonline22March2016
Keywords:
Rheumatoidarthritis Biomarkers
Rheumatoidfactor AntiCCp
Anticitrulinatedvimentin
a
b
s
t
r
a
c
t
Objectives:ToevaluatethefrequencyoffourserumbiomarkersinRApatientsandtheir relativesandidentifypossibleassociationswithclinicalfindingsofthedisease.
Methods:Thiswasatransversalanalyticalstudy.Anti-cycliccitrullinatedpeptide(anti-CCP), anti-mutatedcitrullinatedvimentin(anti-MCV)andIgA-rheumatoidfactor(RF)were deter-minedbyELISAandIgM-RFbylatexagglutinationin210RApatients,198relativesand92 healthycontrolsfromSouthernBrazil.Clinicalanddemographicdatawereobtainedthrough chartsreviewandquestionnaires.
Results:AhigherpositivityforallantibodieswasobservedinRApatientswhencompared torelativesandcontrols(p<0.0001).IgA-RFwasmorefrequentinrelativescomparedto controls(14.6%vs.5.4%,p=0.03,OR=2.98;95%CI=1.11–7.98)whereasanti-CCPwasthe mostcommonbiomarkeramongRApatients(75.6%).Concomitantpositivityforthefour biomarkerswasmorecommoninpatients(46.2%,p<0.0001).Relativesandcontrolswere mostlypositiveforjustonebiomarker(20.2%,p<0.0001and15.2%,p=0.016,respectively). Noassociationwasobservedbetweenthenumberofpositivebiomarkersandageofdisease onset,functionalclassortobaccoexposure.Inseronegativepatientspredominateabsence ofextraarticularmanifestations(EAMs)(p=0.01;OR=3.25;95%CI=1.16–10.66).Arthralgia waspresentinpositiverelatives,regardlessthetypeofbiomarker.
Conclusions:AhighernumberofbiomarkerswaspresentinRApatientswithEAMs.Positivity ofbiomarkerswasrelatedtoarthralgiainrelatives.Thesefindingsreinforcethelinkbetween distinctbiomarkersandthepathophysiologicmechanismsofAR.
©2016ElsevierEditoraLtda.ThisisanopenaccessarticleundertheCCBY-NC-ND license(http://creativecommons.org/licenses/by-nc-nd/4.0/).
∗ Correspondingauthor.
E-mail:renatomitsu@yahoo.com.br(R.Nisihara). http://dx.doi.org/10.1016/j.rbre.2016.03.001
Análise
de
quatro
marcadores
sorológicos
na
artrite
reumatoide:
associac¸ão
com
manifestac¸ões
extra-articulares
no
paciente
e
artralgia
em
familiares
Palavras-chave: Artritereumatoide Biomarcadores Fatorreumatoide AntiCCP
Antivimentinacitrulinada
r
e
s
u
m
o
Objetivos: AvaliarafrequênciadequatromarcadoressorológicosempacientescomARe familiareseidentificarpossíveisassociac¸õescomachadosclínicosdadoenc¸a.
Métodos: Estudo analítico transversal. Determinaram-se os níveis de anticorpos antipeptídeocitrulinadocíclico(anti-CCP),anticorposantivimentina citrulinada-mutada (anti-MCV)efatorreumatoide(FR)IgAporElisaedeFR-IgMporaglutinac¸ãoemlátexem 210pacientescomAR,198parentese92controlessaudáveisdosuldoBrasil.Coletaram-se dadosclínicosedemográficospormeiodarevisãodeprontuáriosequestionários. Resultados: Observou-semaiorpositividadeparatodososanticorposempacientescom ARemcomparac¸ãocomosfamiliaresecontroles(p<0,0001).OFR-IgAeramaisfrequente emfamiliaresquandocomparadoscomoscontroles(14,6%versus5,4%,p=0,03,OR=2,98; IC95%=1,11a7,98).Oanti-CCPfoiobiomarcadormaiscomumentrepacientescomAR (75,6%).Apositividadeconcomitanteparaosquatrobiomarcadoresfoimaiscomumnos pacientes(46,2%,p<0,0001).Familiaresecontroleserampositivosemsuamaioriapara apenasumbiomarcador(20,2%,p<0,0001e15,2%,p=0,016,respectivamente).Nãofoi obser-vadaassociac¸ãoentreonúmerodebiomarcadorespositivoseaidadedeiníciodadoenc¸a, classefuncionalouexposic¸ãoaofumo.Empacientessoronegativos,predominouaausência demanifestac¸õesextra-articulares(MEA)(p=0,01;OR=3,25;IC95%=1,16a10,66).Aartralgia estavapresenteemfamiliarespositivos,independentementedotipodebiomarcador. Conclusões:UmamaiorquantidadedebiomarcadoresestavapresenteempacientescomAR comMEA.Apositividadedosbiomarcadoresestavarelacionadacomaartralgiaem famil-iares.Essesachadosreforc¸amaligac¸ãoentreosdiferentesbiomarcadoreseosmecanismos fisiopatológicosdaAR.
©2016ElsevierEditoraLtda.Este ´eumartigoOpenAccesssobumalicenc¸aCC BY-NC-ND(http://creativecommons.org/licenses/by-nc-nd/4.0/).
Introduction
Rheumatoidarthritis(RA)isachronicinflammatorydisease thatoccursin0.2–1%oftheworldpopulation.1Itsprevalence infirstdegreerelativesofRApatientsisof2–4%,characterizing them as a risk group for RA.2–5 In this context, investiga-tion of serological markers in healthy relatives may be of value to identify earlier cases of RA6,7 and to understand the pathophysiologic mechanisms underlying the disease process.
Rheumatoidfactor (RF)is a classical serological marker used for RA diagnosis, with IgM-RF being the most com-monisoform.In contrast,IgA-RFhasbeen associatedwith erosivearthritisand seems tobe abetter indicatorof dis-ease severity than IgM-RF or IgG-RF.8,9 Antibodies against citrullinated peptides (ACPAs), such as anti-cyclic citrul-linated peptide (anti-CCP) and anti-mutated citrullinated vimentin (anti-MCV) are considered highly specific mark-ersfor RA.10 Anti-CCP has both diagnostic and prognostic value and can be detected before clinical manifestations of the disease.11 Recently, anti-MCV has also been pro-posed as a diagnostic marker for early arthritis, with the samespecificitybuthighersensitivityofanti-CCP.12In addi-tion, anti-MCV has been detected in healthy relatives of RA patients13 suggesting it as a novel prognostic marker forRA.
PositivityformorethanoneRAserologicalmarkerinthe samepatientmayindicateworseprognosisor,inthecaseof relatives,earlydiseaseonset.14,15Infact,positivityformore thanoneautoantibodyhasbeenreportedamongunaffected relativesofRApatients,especiallyinfamilieswithmultiple cases.16,17
Todate,therearenoreportsconsideringthesimultaneity ofthesebiomarkersinRApatientsandtheirrelativesinthe Brazilianpopulation. Thus,inthepresentwork we investi-gatedthefrequencyofanti-CCP,anti-MCV,IgA-RFandIgM-RF inRApatientsandtheirrelatives,andtriedtoidentify possi-bleassociationsbetweenthesimultaneityofthesebiomarkers andclinicalfindingsordiagnosisofRA.
Methods
Thiswasatransversalandanalyticalstudyapprovedbythe localEthics CommitteeinResearch. Informedconsentwas obtainedfromallsubjects.
Subjects
288
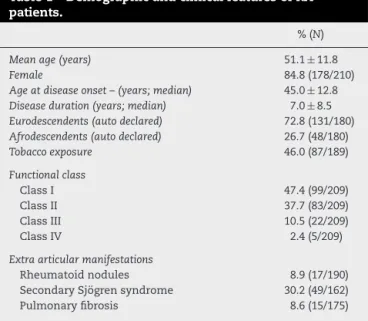
rev bras reumatol.2017;57(4):286–293Table1–DemographicandclinicalfeaturesofRA patients.
%(N)
Meanage(years) 51.1±11.8
Female 84.8(178/210)
Ageatdiseaseonset–(years;median) 45.0±12.8
Diseaseduration(years;median) 7.0±8.5
Eurodescendents(autodeclared) 72.8(131/180)
Afrodescendents(autodeclared) 26.7(48/180)
Tobaccoexposure 46.0(87/189)
Functionalclass
ClassI 47.4(99/209)
ClassII 37.7(83/209)
ClassIII 10.5(22/209)
ClassIV 2.4(5/209)
Extraarticularmanifestations
Rheumatoidnodules 8.9(17/190)
SecondarySjögrensyndrome 30.2(49/162)
Pulmonaryfibrosis 8.6(15/175)
consecutivelyenrolledinthestudyfromAugust2007toApril 2009.DemographicandclinicalfeaturesfromRApatientsare showninTable1.
Atotalof198 relativeswere investigated(61.1%female, 38.9% male; mean age 36.8 years; range 7–91 years, 94% first-degree), representing altogether 78 families (2.54 rela-tives/family).Additionally, sera from 92 healthy volunteers from the same geographicalarea,matched forgenderand agewithpatients,wereusedascontrols(82.6%female,17.4% male;meanage45.8years;range23–81years).Noneofcontrols hadfamilialRAcases.
Autoantibodiesevaluation
Anti-CCP and IgA-RF were assessed by enzyme-linked immunosorbentassay(ELISA)fromINOVADiagnostics(San Diego, USA). Anti-MCV was determined using the ORGEN-TEC Diagnostika ELISA kit (Mainz, Germany). IgM-RF was measured using latex agglutination test (BioSystems S.A., Barcelona,Spain).Cut-offvalueswereestablishedaccording tothemanufacturer’sspecifications.
Datacollection
Demographicand clinical datafrom patientsand relatives wereobtainedthroughmedicalrecordreviewandindividual standardquestionnairesappliedbyarheumatologistatthe timeofenrollment (Tables1and 2).InRApatientsthe fol-lowingextraarticularmanifestations(EAM)weretakeninto account: rheumatoid nodules, serositis, pericarditis, valvu-lar disease, vasculitis, lung fibrosis, pneumonitis, Felty’s syndrome and secondary Sjögren syndrome. Steinbrocker functional classificationwasappliedinorder toaccessthe extentofphysicaldisabilityinRApatients.Diagnosisof sec-ondarySjogren’ssyndromefollowedtheAmericanEuropean criteria.19Clinicalfollow-upofthepositiverelativeshasbeen carriedoutforthemediantimeof7years(range2–8;IQR=6–7 years).
Positivity (
%
)
Relatives Patients
P=.004 P=.03
80
70
60
50
40
30
20
10
0
Anti-CCP FR-IgM FR-IgA Anti-MCV Controls
Fig.1–Positivityofbiomarkersinthestudiedgroups. Patients:anti-CCP+vs.IgA-RF+:p=0.03;OR=1.62;95%
CI=1.06–2.48.Allothercomparisons:p=ns.Relatives:
anti-CCP+vs.IgA-RF+:p=0.004;OR=0.34;95%
CI=0.16–0.70.Allothercomparison:p=ns.Controls:all
comparisonsp=ns.RelativesIgA-RF+vs.ControlsIgA-RF+: p=0.03;OR=2.98;95%CI=1.11–7.98.Allother
comparisons:p=ns.ns,notsignificant.
Statisticalanalyses
StatisticalanalyseswereperformedusingGraphPadPrism4.0 (GraphPadSoftwareInc.,LaJolla,USA)andStatistic5.5 (Stat-Soft Inc.,Tulsa,USA).Comparisonsbetweenautoantibodies levels weredoneapplyingnonparametric Kruskal–Wallisor Mann–Whitneytests.Chi-squareandFisher’sexacttestswere applied for the analysesof the positivity between groups. Spearman’stestwasusedforcorrelationanalysis.The signif-icancelevelwassetat0.05.
Results
PrevalenceofbiomarkersinRApatients,relativesand
controls
TheprevalenceofeachbiomarkerinRApatients,their rela-tivesandcontrolscanbeseenonFig.1,whereitcanbenoted thatthemostprevalentbiomarkerinRApatientsisantiCCP andthemostcommoninrelativeswasIgARF.
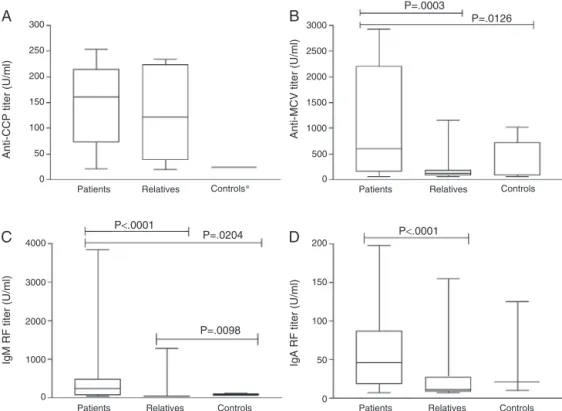
Themediantiterofeachbiomarkerwasstudiedinthe3 groupsandresultsareshowedinFig.2whereitispossibleto seethatrelativeshadanintermediatevaluebetweenpatients andcontrols.
In RA patients a significant correlation was observed among the titers of the four investigated biomarkers, but thecorrelationbetweenanti-MCVandanti-CCP(r=0.73),and IgM-RF and IgA-RF (r=0.71) were both stronger compared to other associations (anti-MCV/IgM-RF, r=0.38; anti-MCV/ IgA-RF, r=0.41; IgM-RF/anti-CCP, r=0.46; IgA-RF/anti-CCP, r=0.47).
r
e
v
b
r
a
s
r
e
u
m
a
t
o
l
.
2
0
1
7;
5
7(4)
:286–293
289
0(n=28) 1(n=15) 2(n=28) 3(n=42) 4(n=97)
Femalegender 26/28(92.8) 9/15(60) 25/28(89.3) 42/42(100) 76/97(78.4) 0.0006a
Ageatdiseaseonset(median) 18–75(41.5;IQR27.2–48.7) 24–69(46.0;IQR35.0–53.0) 19–69(43.5;IQR31.5–50.0) 18–83(43.5;IQR30–49.7) 16–71(46;IQR32.7–52.0) 0.50c
Age(mean) 48.2±10.4 50.3±12.9 50.1±11.9 52.9±13.79 51.7±11.35 0.55d
Diseaseduration(median/years) 1–28(7.0;IQR3.0–12.8) 1–27(4.0;IQR2.0–7.0) 1–35(7.5;IQR3.2–14.2) 1–60(7.0;IQR3.0–11.7) 1–36(6.5;IQR3.2–12.0) 0.36c
Functionalclass
ClassI 15/28(53.6) 8/15(53.3) 13/28(46.4) 22/42(52.4) 41/96(42.7) 0.75a
ClassII 10/28(35.7) 6/15(40) 11/28(39.3) 17/42(40.5) 39/96(40.6) 0.99a
ClassIII+IV 3/28(10.7) 1/15(6.7) 4/28(14.3) 3/42(7.1) 16/96(16.7) 0.53a
Eurodescendent 18/24(75) 10/13(76.9) 17/26(65.4) 28/38(73.7) 58/79(73.4) 0.91a
Afrodescendent 6/24(25) 3/13(23.1) 9/26(34.6) 10/38(26.3) 20/79(25.3) 0.90a
Tobaccoexposure 13/25(52) 7/12(58.3) 12/26(46.1) 16/39(41.02) 39/87(44.8) 0.82a
Nodules 2/24(8.33) 0/13(0) 4/28(14.28) 1/37(2.7) 10/88(11.36) 0.32a
Sjögrensyndrome 1/19(5.2) 3/11(27.2) 4/24(16.6) 16/33(48.4) 25/75(33.3) 0.009a
Lungfibrosis 1/22(4.50) 1/11(9.0) 2/24(8.3) 5/32(15.6) 6/72(8.3) 0.68a
IQR,interquartilerange.
a Chi-square.
b Fishertest.
c Kruskal–Wallistest.
290
rev bras reumatol.2017;57(4):286–293300
P<.0001
P=.0204
P=.0098
P<.0001 P=.0003
P=.0126
A
B
D
C
250
200
150
100
50
0
200
150
100
50
0 3000
2500
2000
1500
1000
500
0
4000
3000
2000
1000
0
Patients
Anti-CCP titer (U/ml)
Anti-MCV titer (U/ml)
IgM RF titer (U/ml) IgA RF titer (U/ml)
Relatives Controls∗ Patients Relatives Controls
Relatives
Patients Controls
Relatives
Patients Controls
Fig.2–Anti-CCP,anti-MCV,IgM-RFandIgA-RFinpatients,relativesandcontrols.*Analysisnotapplicable.
Associationbetweenbiomarkersandclinicalfindingsin
RApatientsandrelatives
Table2showsdemographicandclinicaldataofRApatients
andtheirassociationwiththenumberofpositivebiomarkers. EAM were detected in 34.8% (69/198) of the patients. Patientsthat were negativeforall biomarkers had alower frequency of EAM (5.8% vs. 17.8%, p=0.01; OR=3.25; 95% CI=1.16–10.66).Anincreaseintheconcomitantpositivityfor 3biomarkerswasobservedinpatientswithEAMcompared topatientswithout it (Table3).In theonebyone analysis of the different EAM, only the p value for secondary Sjö-grensyndromeremainedsignificant (p=0.01;OR=2.73;95% CI=1.24–6.08).
Table4showsassociationbetweendemographic,clinical
and serologicaldata and biomarkerspositivityinrelatives. Among the relatives 11.7% (22/197) had arthralgia, and a significantfrequencyofarthralgiawas observedinpositive relatives,regardlessofthetypeofbiomarker.Eachbiomarker presentedsignificant association with the others autoanti-bodiestested(Table4).
Follow-upofpositiverelatives
Therelativeswerefollowedprospectivelyforamediantimeof 7years(range2–8;IQR=6–7years)withbiannualevaluations. Atmoment,threepositiverelatives(twowomenandoneman, meanage43.3years)hadthediagnosisforRAconfirmed;all ofthem withhigh biomarkertiters.Amongthoserelatives, onewaspositiveforall4biomarkersandhissibling(withRA) hadhightitersofanti-CCP.Inaddition,sevenunaffected rela-tiveswithhighautoantibodytitersand somesymptomsof
thediseasearebeingfollowed.Twoofthemarepositivefor4 biomarkersconcomitantly,twofor2biomarkersandthreefor onebiomarker.Noneoftheotherpositiverelativesfortested biomarkershaddiagnosiscriterionsforRA.
NoRAcasesweredetectedamongrelativesnegativesfor autoantibodies.
Discussion
Thisisapioneerstudyinwhichfourserologicalbiomarkers wereevaluatedconcomitantlyinpatientswithRAandtheir relativesinaSouthern Brazilianpopulation.Previous stud-ieshaveshowedanincreasedpositivityforautoantibodiesin unaffectedfamilymemberswhencomparedtohealthy con-trolsubjects5,17,20,21andthepresentresultscorroboratethese findings.
AlthoughIgA-RFspecificityforRAisnotashighasanti-CCP, IgA-RFcanbedetectedseveralyearsbeforethesymptomsof thedisease,suggestingaprimaryroleofIgA-RFinthe patho-genesisofRA.14Thisisaveryinterestingobservationsince inthepresentstudy IgA-RFwasthe biomarkerwithhigher positivityamongrelatives.Inaddition,severalrelativeswere positiveforRFandanti-MCV,butthethreerelativesthathad confirmedRAwereall positiveforanti-CCP,reinforcing the highspecificity(99%)andpositivepredictivevalue(99%)for thisautoantibody.Supportingthisobservation,anti-CCPwas theonlybiomarkerwithnosignificantlydifferenttiterswhen patientsandrelativeswerecompared(Fig.2A).
Table3–Extraarticularmanifestationsaccordingtothenumberofpositivebiomarkers.
Numberofpositivebiomarkers WithEAM(n=69)
N(%)
WithoutEAM(n=129)
N(%)
p
4(n=90) 34(49.3) 56(43.4) ns
3(n=39) 19(27.5) 20(15.5) 0.05
2(n=28) 9(13.1) 19(14.7) ns
1(n=14) 3(4.3) 11(8.5) ns
Nobiomarker(n=27) 4(5.8) 23(17.8) 0.01
EAM,extraarticularmanifestation;ns,notsignificant.
Note:3biomarkerswithEAMvs.3biomarkerswithoutEAM;p=0.059;OR=1.26;95%CI=0.70–2.27.NobiomarkerwithEAMvs.Nobiomarker
withoutEAM;p=0.0179;OR=3.25;95%IC=1.16–10.66.Fishertest.
Table4–Associationbetweendemographic,clinicalandserologicaldataandbiomarkerspositivityintheRApatients relatives.
IgM-RF+(n=16) IgM-RF−(n=182) p
Euro-descendent 14/16 140/182 0.53a
Afrodescendent 2/16 42/182 0.53a
Tobaccoexposure 3/16 54/182 0.56a
Female 13/16 108/182 0.11a
Meanage(years) 40.3±12.3 36.5±15.6 0.34c
Arthralgia 6/16 16/178 0.004a
Anti-CCP+ 5/16 6/182 0.0006a
IgA-RF+ 6/16 23/182 0.01a
Anti-MCV+ 5/16 11/182 0.004a
IgA-RF+(n=29) IgA-RF−(n=169) p
Euro-descendent 24/29 130/169 0.63a
Afrodescendent 5/29 39/169 0.63a
Tobaccoexposure 9/29 48/169 0.77b
Female 17/29 104/169 0.76b
Meanage(years) 40.6±15.4 36.1±15.3 0.15c
Arthralgia 6/29 16/165 0.08b
Anti-CCP+ 4/29 7/169 0.059a
IgM-RF+ 6/29 10/169 0.007b
Anti-MCV+ 7/29 9/169 0.0006b
Anti-CCP+(n=11) Anti-CCP−(n=187) p
Euro-descendent 9/11 145/187 1.00a
Afrodescendent 2/11 42/187 1.00a
Tobaccoexposure 3/11 54/187 1.00a
Female 9/11 112/187 0.37a
Meanage(years) 39.3±15.9 36.6±15.4 0.57c
Arthralgia 4/11 18/183 0.02a
IgM-RF+ 5/11 11/187 0.0006a
IgA-RF+ 4/11 25/187 0.05a
Anti-MCV+ 5/11 11/187 0.0006a
Anti-MCV+(n=16) Anti-MCV−(n=182) p
Euro-descendent 12/16 142/182 0.75a
Afrodescendent 4/16 40/182 0.75a
Tobaccoexposure 4/16 53/182 1.00a
Female 14/16 107/182 0.03a
Meanage(years) 33.5±16.9 37.1±15.2 0.37c
Arthralgia 6/16 22/178 0.0006a
Anti-CCP+ 5/16 6/182 <0.0001b
IgM-RF+ 5/16 11/182 0.004a
IgA-RF+ 7/16 22/182 0.002b
a Fishertest.
b Chi-square.
292
rev bras reumatol.2017;57(4):286–293thisoutcomemaybetheepitopespreadingphenomenonthat causesincreasingantibodylevelsjustpriortotheflareof clin-icaldisease.23
Somestudieshavesuggestedthatanti-MCVhasa com-parablevaluetoanti-CCPforRAdiagnosis,withevenhigher sensitivity.24,25 Thiswas notthe case inthe present study whentheprevalence,sensitivityandspecificityofanti-MCV werelowerthanthoseofanti-CCP.
A strong correlation between RA biomarkers has been previouslydisclosed.PoulsomandCharles26showedagood correlationbetweenanti-MCVandanti-CCPandbetween anti-MCVandIgM-RF,butnotbetweenanti-CCPandIgM-RF.We foundsignificantcorrelationbetweenallmarkers,especially between anti CCP and anti MCV and between RF IgA and IgM.ThispatternwaspreviouslydescribedinfirstRAdegree relatives27andmaybeassociatedwiththepresenceof com-monepitopes.
OurcasuisticpresentedahighprevalenceofEAMs(34.8%) that may be explained by geneticand environmental fac-tors that influence the immune reactions leading to their development.28Weobservedpositivityforahighernumberof biomarkersamongpatientswithEAMs,inspecialsecondary SS.
Thepresent resultsfor relativesofRA patients demon-stratedthatserologicalscreeningisfeasibleandusefultool forthescreeningofthoseindividualsatrisk,asemphasized byotherauthors.17,21,22Thehighprevalenceofarthralgiain relativespositivetoanyofthefourbiomarkershighlightsthis hypothesis,sincesomeoftheserelatives(three)havealready evolvedtofullblownRA.AlltherelativeswhodevelopedRA presentedhighbiomarkerstiters;onewaspositiveforallfour biomarkersandhad oneextra siblingpositive foranti-CCP. Amongtheothertwo,onewaspositiveforbothanti-CCPand IgM-RF,whilethe otherwasonlypositiveforanti-CCP.The majorityofrelativeswithautoantibodieswhodidnotdevelop thediseasesofarwerepositiveforjustonebiomarker. How-everthisdoesnotruleoutthegreatriskforthemtodevelop thedisease.Itispossiblethattheymayposteriorly serocon-verttotwo,threeorevenmorebiomarkersasshowninthe patientswithestablishedRA.
Finally, the number of biomarkers was higher in RA patients with EAMs but it was not linked to duration of thediseaseand/orthefunctionalclass.Ontheother hand, seronegative patientspresentedless EAMs. Highfrequency ofarthralgiawasobservedinpositiverelatives,regardlessof thebiomarkertype.Futurestudiesmayhighlightthe fascinat-inglinkbetweenthepresenceofdistinctbiomarkersandthe pathophysiologicmechanismsofAR.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
r
e
f
e
r
e
n
c
e
s
1. AlamanosY,VoulgariPV,DrososAA.Incidenceand
prevalenceofrheumatoidarthritis,basedonthe1987
AmericanCollegeofRheumatologycriteria:asystematic
review.SeminArthritisRheum.2006;36:182–8.
2.HarneyS,WordsworthBP.Geneticepidemiologyof
rheumatoidarthritis.TissueAntigens.2002;60:465–73.
3.FiresteinGS.Evolvingconceptsofrheumatoidarthritis.
Nature.2003;5:356–61.
4.HemminkiK,LiX,SundquistJ,SundquistK.Familial
associationsofrheumatoidarthritiswithautoimmune
diseasesandrelatedconditions.ArthritisRheum.
2009;60:661–8.
5.GoeldnerI,SkareTL,MessiasReasonIT,NisiharaRM,Silva
MB,UtiyamaSRR.Anti-cycliccitrullinatedpeptideantibodies
andrheumatoidfactorinrheumatoidarthritispatientsand
relativesfromBrazil.Rheumatology.2010;49:1590–3.
6.SilmanAJ,MacGregorAJ,ThomsonW,HolliganS,CarthyD,
FarhanA,etal.Twinconcordanceratesforrheumatoid
arthritis:resultsfromanationwidestudy.BrJRheumatol.
1993;32:903–7.
7.MacGregorAJ,SniederH,RigbyAS,KoskenvuoM,KaprioJ,
AhoK,etal.Characterizingthequantitativegenetic
contributiontorheumatoidarthritisusingdatafromtwins.
ArthritisRheum.2000;43:30–7.
8.TuressonC,MattesonEL.Vasculitisinrheumatoidarthritis.
CurrOpinRheumatol.2009;21:35–40.
9.Nell-DuxneunerV,MacholdK,StammT,EberlG,HeinzlH,
HoeflerE,etal.Autoantibodyprofilinginpatientswithvery
earlyrheumatoidarthritis–afollow-upstudy.AnnRheum
Dis.2010;69:169–74.
10.BartoloniE,AlunnoA,BistoniO,BizzaroN,MiglioriniP,
MorozziG,etal.Diagnosticvalueofanti-mutated
citrullinatedvimentinincomparisontoanti-cyclic
citrullinatedpeptideandanti-viralcitrullinatedpeptide2
antibodiesinrheumatoidarthritis:anItalianmulticentric
studyandreviewoftheliterature.AutoimmunRev.
2012;11:815–20.
11.FaridSS,AziziG,MirshafieyA.Anti-citrullinatedprotein
antibodiesandtheirclinicalutilityinrheumatoidarthritis.
IntJRheumDis.2013;16:379–86.
12.MathssonL,MullazehiM,WickMC,SjöbergO,Van
VollenhovenR,KlareskogL,etal.Antibodiesagainst
citrullinatedvimentininrheumatoidarthritis:higher
sensitivityandextendedprognosticvalueconcerningfuture
radiographicprogressionascomparedwithantibodies
againstcycliccitrullinatedpeptides.ArthritisRheum.
2008;58:36–45.
13.RazaK,MathssonL,BuckleyCD,FilerA,RönnelidJ.
Anti-modifiedcitrullinatedvimentin(MCV)antibodiesin
patientswithveryearlysynovitis.AnnRheumDis.
2010;69:627–8.
14.Rantapää-DahlqvistS,JongBAW,BerglinE,HallmansG,
WandellG,StenlundH,etal.Antibodiesagainstcyclic
citrullinatedpeptideandIgArheumatoidfactorpredictthe
developmentofrheumatoidarthritis.ArthritisRheum.
2003;48:2741–9.
15.NielenMM,VanSchaardenburgD,ReesinkHW,VandeStadt
RJ,VanderHorst-BruinsmaIE,deKoningMH,etal.Specific
autoantibodiesprecedethesymptomsofrheumatoid
arthritis:astudyofserialmeasurementsinblooddonors.
ArthritisRheum.2004;50:380–6.
16.KolfenbachJR,DeaneKD,DerberLA,O’DonnellC,Weisman
MH,BucknerJH,etal.Aprospectiveapproachtoinvestigating
thenaturalhistoryofpreclinicalrheumatoidarthritis(RA)
usingfirst-degreerelativesofprobandswithRA.Arthritis
Rheum.2009;61:1735–42.
17.ArlestigL,MullazehiM,KokkonenH,RocklövJ,RönnelidJ,
DahlqvistSR.Antibodiesagainstcycliccitrullinatedpeptides
ofIgG,IgAandIgMisotypeandrheumatoidfactorofIgMand
rheumatoidarthritisfamiliesfromnorthernSweden.Ann
RheumDis.2012;71:825–9.
18.ArnettFC,EdworthySM,BlochDA,McShaneDJ,FriesJF,
CooperNS,etal.TheAmericanRheumatologyAssociation
1987revisedcriteriafortheclassificationofrheumatoid
arthritis.ArthritisRheum.1988;31:315–24.
19.VitaliC,BomardieriS,JonssonR,MoutsopoulosHM,
AlexanderEL,CarsonsSE,etal.Classificationcriteriafor
Sjögren’ssyndrome:arevisedversionoftheEuropeancriteria
proposedbytheAmericanEuropeanconsensusgroup.Ann
RheumDis.2002;61:554–8.
20.Ioan-FacsinayA,WillemzeA,RobinsonDB,PeschkenCA,
MarklandJ,VanderWoudeD,etal.Markeddifferencesinfine
specificityandisotypeusageoftheanti-citrullinatedprotein
antibodyinhealthanddisease.ArthritisRheum.
2008;58:3000–8.
21.YoungKA,DeaneKD,DerberLA,HughesAustinJM,Wagner
CA,SokoloveJ,etal.Relativeswithoutrheumatoidarthritis
shoereactivitytoanti-citrullinatedprotein/peptide
antibodieswhichareassociatedwitharthritis-relatedtraits:
studiesoftheetiologyofrheumatoidarthritis.Arthritis
Rheum.2013;65:1995–2004.
22.BarraL,ScinoccaM,SaundersS,BhayanaR,RohekarS,
RacapéM,etal.Anti-citrullinatedproteinantibodiesin
unaffectedfirst-degreerelativesrelativesofrheumatoid
arthritispatients.ArthritisRheum.2013;65:1439–47.
23.VanderWoudeD,Rantapaa-DahlqvistS,Ioan-FacsinayA,
OnnekinkC,SchwarteCM,VerpoortKN,etal.Epitope
spreadingoftheanti-citrullinatedproteinantibodyresponse
occursbeforediseaseonsetandisassociatedwiththe
diseasecourseofearlyarthritis.AnnRheumDis.2010;69:
1554–61.
24.DejacoC,KlotzW,LarcherH,DuftnerC,SchirmerM,Herold
M.Diagnosticvalueofantibodiesagainstamodified
citrullinatedvimentininrheumatoidarthritis.ArthritisRes
Ther.2006;8:R119.
25.SvardA,KastbomA,SoderlinMK,Reckner-OlssonA,SkoghT.
AcomparisonbetweenIgG-andIgA-classantibodiestocyclic
citrullinatedpeptidesandtomodifiedcitrullinatedvimentin
inearlyrheumatoidarthritisandveryearlyarthritis.J
Rheumatol.2011;38:1265–72.
26.PoulsomH,CharlesPJ.Antibodiestocitrullinatedvimentin
areaspecificandsensitivemarkerforthediagnosisof
rheumatoidarthritis.ClinRevAllergyImmunol.2008;34:4–10.
27.KimSK,BaeJ,LeeH,KimJH,ParkSH,ChoeJY.Greater
prevalenceofseropositivityforanti-cycliccitrullinated
peptideantibodyinunaffectedfirst-degreerelativesin
multicaserheumatoidarthritis-affectedfamilies.KoreanJ
InternMed.2013;28:45–53.
28.GibofskyA.Overviewofepidemiology,pathophysiology,and
diagnosisofrheumatoidarthritis.AmJCare.