ORIGINAL
ARTICLE
Correlation of disease spectrum among four Dengue serotypes:
a five years hospital based study from India
Author RajniKumaria
PostdoctoralAssociate– Singapore-MITAlliancefor ResearchandTechnology, NationalUniversityof Singapore,Singapore.
Submittedon:06/11/2009 Approvedon:08/06/2009
Correspondence to: Dr.RajniKumaria Singapore-MITAlliance forResearchand Technology, NationalUniversityof Singapore,Singapore Phone:6582282240 E-mail:rankumaria@ gmail.com
Wedeclarenoconflictof interest.
ABSTRACT
TherecognitionofDF(DHFDengue Hemorrhagic Fever)isverycomplicatedduetooccurrenceof awidespectrumofclinicalsignsandsymptomsduringacutephaseofillness.Moreover,presenceof fourserotypesfurthercomplicatestheprognosis.Toinvestigatethepredictorsofdiseaseseverityand elucidatetheprognosticmarkersamongfourdengueserotypes,thisstudywasconductedon320 inpatientshavingacutefebrileillnessclinicallysuspectedasDI,overaperiodoffiveyears.Dengue serotypeswereconfirmedbymultiplexreversetranscriptase(RT)-PCR.Eightypatientswerepositive forDIwithpresenceofDen-1,Den-2,Den-3,andDen-4in8,35,27and10patients,respectively. Thesevereclinicalmanifestations,abdominalpainandhepatomegaly,werecomparativelyhigherin Den-2patients.LiveraminotransferaseslevelswerealsohigherinDen-2patients(app.5fold).This studyclearlyindicatesthehyperendemicityofalldengueserotypes.NucleotidesequencingofEn-veloperegionrevealedthatthepresentlyemergedDen-3belongstotypeIII,havinghighhomology withgenotyperesponsiblefornumberofoutbreaksin1980s.There-emergenceofthisdeadlytype canbesuspectedtocausemoreoutbreaksinfutureandisamatterofgreatconcern.
Keywords: Denguefever,dengueinfection,serotype,clinicalspectra,livertransaminase. [BrazJInfectDis2010;14(2):141-146]©ElsevierEditoraLtda.
INTRODUCTION
Denguevirus,amemberofthefamilyflaviviri-dae,hasfourserologicallyrelatedbutgenetically distinctvirusserotypes,namely,Den-1,Den-2, Den-3 and Den-4. Dengue infection (DI) is presentlythemostimportantarboviraldisease
ofmankind.1
DImayeitherremainasympto-matic, or rise as mild infection called dengue fever (DF). In some instances, it results into denguehemorrhagicfever(DHF).2,3Atpresent,
DIisendemicinover100countriesworldwide and causes nearly 100 million cases of DF, 500,000casesofDHFand24,000deathseach year.4-7Pathophysiologicalbasisofhemorrhage
indenguepatientsstillremainspoorlyunder-stood.6ApatienthavingDIcanrapidlydevelop
life-threatening hemorrhagic form, as early as2daysaftertheonsetofillness.1,8-10Dengue
outbreaksinIndiaarenowfollowingthepat-tern like other high burdened countries (e.g., Thailand and Indonesia).11 The study during
thelastmajorDHFoutbreakin1996reported Den-2asthepredominantserotypecirculating inIndia.12
However,theextensiveresearchdur-
ingthisstudy,since2002,highlightsthepres-enceofallfourserotypes,withpredominance
ofDen-2andDen-3serotypes.13
Because all four dengue virus serotypes cause a number of illness, defining precisely which clinical manifestations and laboratory
abnormalitiesareassociatedwithdifferentse-rotypesremainsdisappointing.14Conductedto
evaluatediseasetreatment,thecurrentstudy,a firstofitskindfromthisgeographicalregion, wasplannedtorecognizetheclinicalandbio-chemicalmanifestationsoccurringduringacute stageofinfectioninpatientsinfectedwithvari- ousserotypes.Thepresentworkpresentsabet- terunderstandingofcorrelationbetweensero-typesanddiseasemanifestationsduringacute phase. Further, genotypic characterization of Den-3,thesecondmostpredominantserotype duringthisstudy,revealedthethreatassociated withthisnewlyre-emergedserotype.
METHODOLOGY
Patient selection and sample processing
clinically suspected of DI, attending the outpatient and medical wards of Lok Nayak Hospital, a tertiary care hos-pitalatDelhi,India,fromJanuary2002toNovember2006. OnthebasisofWHOinclusioncriteria,1apatienthavingan
acutefebrileillnessof2-7daysdurationwithtwoormoreof thefollowingmanifestations:severeheadache,retro-orbital pain,myalgia,arthralgia,rash,hemorrhagicmanifestations, and/or leucopenia was categorized as having DF. Patient withDHFisaprobablecaseofDF,havinghemorrhagicten-dency along with thrombocytopenia (platelets 100,000/cu. mmorless)andevidenceofplasmaleakage.Fiftynormal healthyageandsexmatchedindividuals,havingnohistory offebrileorotherillnessinthelastthreemonthswerein-cludedasnegativecontrols.
ThepriorapprovalhasbeengrantedbytheEthicalCom- mitteeoftheInstitutetoconductthisstudy.Informedcon-sentwasobtainedfromallpatients.Acasewasexcludedif routine laboratory testing suggested bacterial infection or anyviralinfectionotherthanDIoranyotherdisease.
Detection of dengue serotypes by multiplex RT-PCR
ReferencestrainsspecificforserotypeDen-1:Hawaii;Den-2: P-23085;Den-3:633798;andDen-4:642069(obtainedfrom NationalInstituteofVirology,Pune,India)wereindividu-allyinoculatedinC6/36cellscontainingLeibovitz-15media
with2%FetalBovineSerum.15-17After7days,supernatant
was separated from each culture vial at 1500 rpm for 10 minutes, 4° C and stored at -70° C, to be used as positive controls.Thepresenceofdengueserotypeswasconfirmed bymultiplexRT-PCR,usingfourserotypespecificprimers pairsdescribedearlier.13,18Serumsamplesfromallpatients
weresimilarlyprocessedfordetectionofdengueserotypes.
Amplifi cation of E-NS1 gene region by RT-PCR
AsperthemethodofDomingoet al.,19
5µlofdenguevi-ralRNAwasusedforRT-PCRmixtoamplifyE-NS1gene regionofDen-3serotypespecificstrains.Thereactionwas carriedoutintwostepsandthefirststepconsistedofRT-PCR using outer sense primer S1871DEN3 and anti-sense primer AS2622DEN3 specific for Den-3. This reaction was followed by nested PCR using internal sense primer; S2176DEN3 specific for Den-3 and a common anti-sense primerAS2504DEN.
Nucleotide sequencing of E-NS1 region of Den-3 and phylogenetic analysis
ThepurifiedDNAproducts(25-50ng)weresequencedby ABI3130xlautomatedDNAsequencer(AppliedBiosystems, USA) using dideoxy chain termination method with ABI sequencing kit following manufacturer’s instructions, us-ingprimersS2176DEN3andAS2504DEN.Sequencesthus generatedweresubmittedtoGeneBankdatabase.Sequence identity matrices and multi-sequence alignments from
DNAdatabasesweregeneratedusingBasicLocalAlignment Search Tool; available at NCBI website (http://www.ncbi. nlm.nih.gov/BLAST) and ClustalW programme (http:// align.genome.jp/).
Hematological and biochemical investigations
Blood samples were collected into an EDTA-coatedVacu-tainertube(BectonDickinsonVacutainerSystems,Franklin Lakes,N.J.),forhematologicalinvestigationsandplainVa- cutainertube(BectonDickinsonVacutainerSystems,Fran-klinLakes,N.J.),forbiochemicalinvestigations.Allthetests wereperformedasperstandardlaboratoryprocedures.
Statistical analysis
StatisticalanalysiswasperformedwithEpiInfoversion3.3.2 (updated2005,CenterforDiseaseControlandPrevention, USA).Proportionsofpatientswithabnormalclinical,hema-tologicalandbiochemicalfindingsbetweenthetwogroups were compared using Chi (χ2)/Fischer exact test. Mean valuesbetweenfourserotypegroupswerecomparedusing analysisofvariance(ANOVA).Kruskal-Wallistestwasap-plied,whenvarianceinsampleswashigh.Oddsratio(OR) for disease outcomes in serotypes was calculated to assess strengthofassociationat95%confidenceintervals.There-sultswereconsideredstatisticallysignificantwhenerrorwas lessthan5%(p<0.05).
RESULTS
Distribution pattern of dengue serotypes among dengue patients
On the basis of multiplex RT-PCR, DI was confirmed in eightypatients.Serotypiccharacterizationidentified35pa-tientsbeingpositiveforDen-2,27forDen-3,10forDen-4, and8patientspositiveforDen-1.Thus,Den-2wasdetected inmostpatients,followedbyDen-3.
Correlationofdengueserotypeswithclinicalandhem-orrhagicmanifestations
Correlation of serotypes with hematological parameters
Thrombocytopenia was the common hematological manifestationduringacutephase.Thelowestmeanplate-let counts were observed in the Den-2 infected patient group. Hematological parameters (except TLC) showed significant difference in disease group when compared with control group (Table 2). However, none of the pa-rametersshowedsignificantdifferencewithinthedisease
groups. In Den-3 infected patients, a strong correlation wasfoundbetweenhematocritandhemoglobinlevels(p value<0.05,r=0.411)andalsoplateletcountsandTLC levels(pvalue<0.05,r=0.484).Inpatientsinfectedwith Den-2,correlationwasobservedbetweenplateletcounts and hemoglobin levels (p value < 0.05, r = 0.25). How- ever,therewasnocorrelationbetweenanyofthehemato-logicalparametersinDen-1andDen-4infectedpatients inthepresentstudy.
Table 1. Correlation of Dengue serotypes with clinical and hemorrhagic manifestations
Disease Manifestations Dengue serotype groups
Den-1 Den-2 Den-3 Den-4 p Value
Abd Pain, n (%) 3 (50) 25 (71.4) 9 (33.3) 7 (70) < 0.001
Hepatomegaly, n (%) 1 (12.5) 17 (49) 5 (18.5) 2 (20) < 0.05
ROP, n (%) 4 (50) 23 (65.7) 13 (48.2) 6 (60) 0.54
Anorexia, n (%) 7 (87.5) 33 (94.3) 19 (70.4) 8 (80) 0.18
Arthalgia, n (%) 5 (40) 24 (68.5) 16 (59.3) 4 (40) 0.54
Myalgia, n (%) 4 (50) 21(60) 14 (51.9) 6 (60) 0.95
Bleeding 4 (50) 31 (88.6) 12 (44.4) 10 (100) 0.001
Malena 3 (37) 11 (31.4) 4 (14.8) 4 (40) 0.3
Hemetamesis 1 (12.5) 9 (25.7) 2 (7.4) 3 (30) 0.22
Epistaxis 0 (0) 12 (34.3) 3 (11.1) 4 (40) 0.07
Footnote: Significant p values are underlined (calculated by Chi square Test/Fisher’s Exact Test) Abd = abdominal; ROP = retro-orbital pain.
n = number of patients having manifestations.
Table 2. Correlation of the four dengue serotypes with hematological parameters
Hematological Parameters Control Group Den-1 Den-2 Den- 3 Den-4 p value
Hb 12.5 12.5 11.6 12.2 10.6
< 0.05
(mmHg) (±0.8) (±1.04) (±3.0) (±1.3) (±2.6)
TLC 6930 5900 6608 6559 6610
(cells/mm3) (±1221) (±2389) (±2037) (±3467) (±1737) 0.6
Hematocrit 43.3 38.12 (%) 37.25 39.6 37.3
(±3.8) (±3.4) (±5.9) (±3.6) (±9.1) < 0.05
Platelet count 312619 72000 66700 72900 75400
(cells/mm3) (±66150) (± 55500) (±40600) (±42000) (±39900) < 0.01
PT 14.4 14.5 16 17.2 16.6
(sec) (±0.7) (±0.7) (±1.1) (±2.2) (±1.8) < 0.001
Hb = hemoglobin; TLC = total leukocyte count.
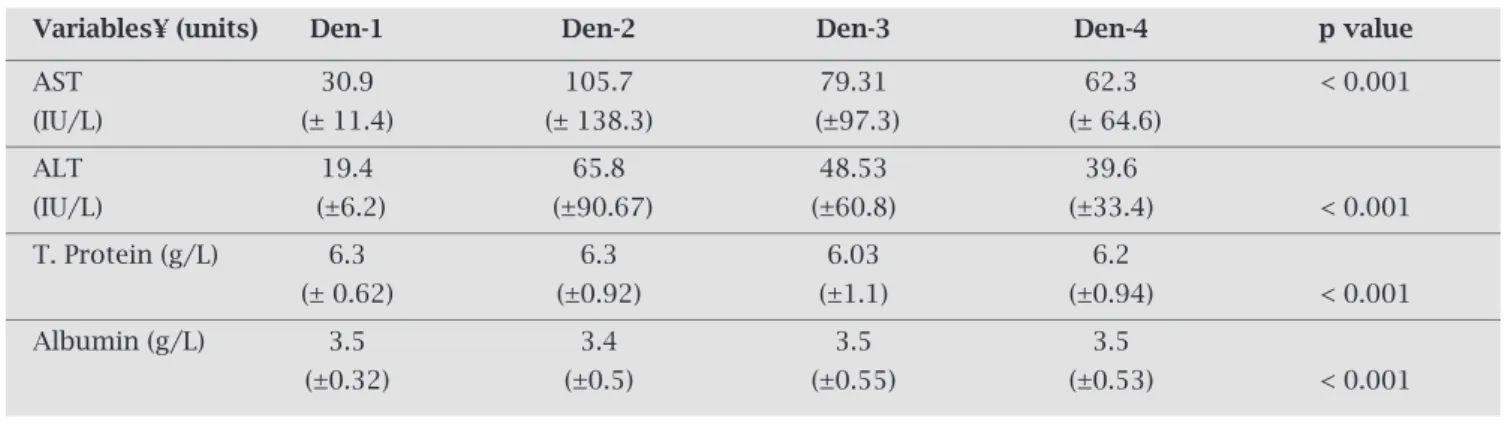
Correlation of serotypes with biochemical profi le
The elevation of liver aminotransferase levels was maxi-muminDen-2patients.Increaseofaboutfive-foldinlevels of both the aminotransferase was noticed as compared to healthycontrols.ThemeanlevelofserumalbumininDen-2 infected patients was slightly reduced, as compared to the normal reference limits. However, when compared with normal healthy controls, the mean levels of all biochemi- calparametersweresignificantlyaltered(Table3).Likehe-matological parameters, there was no significant different between biochemical parameters within serotype groups. ThefrequencyofpatientswithelevatedlevelsofSGOTwas significantly higher in Den-2 group (p < 0.05) showing a strongassociation(O.R.2.42;CI=0.89-6.64).
Genotypic characterization of Den-3
There was 99-100% nucleotide homology between the se-quences of five Den-3 strains isolated from dengue pa-tients(Accessionnumbers:EF094024,EF119833,EF119834, EF119835,EF119836).ATcompositionofthesequencesfrom thepresentstudywasbetween51-58.3%.ComparisonofE-NS1regionofthesestrainswithpublishedsequencesofother strainsofDen-3categorizedthenintosubtypeIII(Table4).
DISCUSSION
DIhasnowbecomearegularpost-monsoonphenomenon inmostpartsofIndia.13,20
MostofIndianstudiesonclini-calandlaboratoryprofileofdenguepatientshaveinvolved serologicallypositivepatients.Becauseserologicaldiagnosis demandsacollectionofsamplesfivedayspostdiseaseon-set,itthusdepictsthepatternofmanifestationsoccurringin the convalescent phase of disease.As severe complications relatedtoDHFmayoccurveryearlyindisease,itisimpor-tanttoexploreprognosticmarkersofseverediseaseduring acute phase. The hyperendemicity with co-circulation of two or more serotypes during the same time period have beenwidelysuspectedasoneofthemajorcauseofdisease severityinSouthEastAsia.8,21Variousstudiesaretryingto
establishconnectionbetweencausativeserotypeanddisease outcome.FewofwhichproposethatDen-2andDen-3may causemoreseveredisease,8,22-24
however,adefinitelinkbe-tweendistinctserotypesandseveremanifestationshasnot beenestablishedyet.
Thisstudyprovideddirectevidenceforthepresenceof all four serotypes in this geographical region, with Den-2 andDen-3astwopredominantserotypes.Theclinicalman-ifestations,suchasabdominalpainandhepatomegaly,were
Table 3. Serum levels of biochemical parameters: Comparison between four serotypes and healthy controls
Variables¥ (units) Den-1 Den-2 Den-3 Den-4 p value
AST 30.9 105.7 79.31 62.3 < 0.001
(IU/L) (± 11.4) (± 138.3) (±97.3) (± 64.6)
ALT 19.4 65.8 48.53 39.6
(IU/L) (±6.2) (±90.67) (±60.8) (±33.4) < 0.001
T. Protein (g/L) 6.3 6.3 6.03 6.2
(± 0.62) (±0.92) (±1.1) (±0.94) < 0.001
Albumin (g/L) 3.5 3.4 3.5 3.5
(±0.32) (±0.5) (±0.55) (±0.53) < 0.001
¥ = Value of each parameter is expressed as mean value ± S.D. Significant p values are underlined (calculated by comparing the four serotype groups with control group - calculated by ANOVA test/Kruskal Wallis test).
Table 4. Den-3 strains: analysis of E-NS1 gene region
Strain Year of isolation Serotype (Genotype) Gene bank accession
number
ARG051 2003 Den-3 (III) EF094024
106ARMG1 2005 Den-3 (III) EF119833
206ARMG1 2005 Den-3 (III) EF119834
306ARMG1 2006 Den-3 (III) EF119835
higherinDen-2patients,indicatingaroleincausingliver function abnormalities. Incidences of severe hemorrhagic manifestations,suchasepistaxis,malena,andhemetemesis, werehigherinpatientshavingDen-4.AsDen-4positivity remainedverylow,thereisaneedofmorestudiestovalidate thisfinding.InaretrospectivestudyfromThailand,Den-2 hasbeendocumentedasthemostfrequentserotypeamong
DHFcases(35%),followedbyDen-3(31%).24Inastudyin
Thailand,23Den-2showedassociationwithagreaterpleural
effusionindexandwithahigherpercentageofDHFcases. In congruence with the present study, Corwinet al.25 also
foundthatDen-1isassociatedwithlessseveredisease.Sev-eralreportsfromothercountriesarticulatingdifferencesin disease severity associated with distinct dengue serotypes supportourresults.Itissuggestedthatscreeningforabdom-inalpain,andhepatomegalyalongwithseverehemorrhagic manifestationsinpatientstestedpositivebymultiplexRT-PCRduringacutephaseofillnesswouldbeanassetinbetter prognosis.Hepaticdysfunctioniscommoninpatientswith
severedenguemanifestations.26Liverdysfunctionmightbe
responsibleforthedecreasedsynthesisofspecificfactorsof the intrinsic pathway, an unbalance of which may conse-quentlycausehemorrhage.26Inourfinding,meanvaluesof
boththetransaminasesweresignificantlyraisedinallfour serotype groups, when compared with control groups. El-evation of liver aminotransferase levels was maximum in Den-2 patients (app. five-fold increase). Moreover, reduc-tioninserumalbuminlevelspointstowardsriskofpleural effusion.HigherlevelsofSGOTandSGPThavebeenearlier reportedfromalimitedsampleofDen-2infectedpatients.27 However,moreinvestigationsarestillrequiredbeforeadefi- niteassociationofanyserotypewithhematologicalorbio-chemicalprofilescanbeascertained.Asmortalityisusually linkedtodelayedprovisionofsupportivetreatment,abetter understandingofclinicalandbiochemicalprofileduringthe acutephaseofillnessisextremelyimportant.
Changes in the envelope protein in particular are sug-gestedtoalterhostcellsusceptibility,therebyaffectingvirus replication and disease severity.28 The nucleotide sequence
comparison of E-NS1 region of five Den-3 strains from present study with published sequences using BLAST re-vealed that irrespective of the year in which samples were collected,allofthembelongedtogenotypeIII.Thestrains isolatedinourstudyshowed~98%homologywithsubtype-IIIofDen-3strainsisolatedduringepidemicsin1984from India,in1985fromMozambique,in1989,1991and2000
fromSriLanka,andin2000fromMartinique.28,29
Theiden-tificationofDen-3duringtheoutbreakinIndiaduring2003 anditscontinuedpresenceinlateryearsindicatesitsresur- genceinadominantformand,thus,amatterofgreatcon-cern. Constant and thorough epidemiological surveillance ismandatorytocontroltheinvasionandspreadofdengue viruses.Studiesofthesetypeswoulddefinitelybeanassetin theestablishmentofintimeeffectivecontrolstrategies.
REFERENCES
1. World Health Organization. Prevention and control of den-gueanddenguehemorrhagicfever:comprehensiveguidelines. WHORegionalpublication,SEARO1999;29. 2. GublerDJ.Dengueanddenguehemorrhagicfever.ClinMicro-biolRev1998;11:480-96. 3. MalavigeGN,FernandoS,FernandoDJ,SeneviratneSL.Den-gueviralinfections.PostGradMedJ2004;80:588-01. 4. CDC; Centers for Disease Control and Prevention. National
Center for Infectious Diseases, Division ofVector-Borne In-fectiousDiseases.Denguefeverhomepageyear.www.cdc.gov/ ncidod/dvbid/dengue.htm. 5. DengueNet-WHOsInternet-basedSystemforGlobalSurveil- lanceofDengueFeverandDengueHaemorrhagicFever(Den- gue/DHF).WeeklyEpidemiologicalRecord2002;77(36):301-304.http://www.who.int/denguenet. 6. GibbonsRV,VaughnDW.Dengue:anescalatingproblem.BM J2002;324:1563-66.
7. Sapir DG, Schimmer B. Dengue fever: new paradigms for a changing epidemiology. Emerging Themes in Epidemiology 2005, 2:1 doi:10.1186/1742-7622-2-1 online http://www.ete-online.com/content/2/1/1.
8. EndyTP,NisalakA,ChunsuttiwatSet al.Spatialandtemporal
circulationofdenguevirusserotypes:aprospectivestudyof primaryschoolchildreninKamphaengPhet,Thailand.AmJ Epidemiol2002;56:52–9.
9. GuzmanMG,KouriG.Dengueanddenguehemorrhagicfeverin Americas:lessonsandchallenges.JClinVirol2003;27:1-13. 10. World Health Organization. Dengue haemorrhagic fever:
diagnosis, treatment, prevention and control, 2nd ed.World HealthOrganization,Geneva,Switzerland.1997.
11. DengueNetinIndia.WeeklyEpidemiologicalRecord2004;79 (6):57-62.
12. SinghUB,MaitraA,BroorS,RaiA,PashaST,SethP.Partial nucleotide sequencing and molecular evolution of epidemic causingdengue2strains.JInfectDis1999;180:959-65. 13. KumariaR,ChakravartiA.Moleculardetectionandserotypic
characterization of dengue viruses by single tube multiplex RT-PCR.DiagnMicrobiolInfectDis2005;52(4):311-6.
14. BalmasedaA,HammondSN,PérezLet al.Serotype-specific
differences in clinical manifestations of dengue. Am J Trop Med.Hyg2006;74(3):449-56.
15. HarrisE,RobertsG,SmithLet al.Typingofdenguevirusesin
clinicalspecimensandmosquitoesbysingle-tubemultiplexre-versetranscriptasePCR.JClinMicrobiol1998;36(9):2634-9. 16. Igarashi A. Mosquito cell cultures and study of
arthropod-bornetogaviruses.AdvVirusRes1985;30:21-39.
17. TeshRB.Amethodfortheisolationandidentificationofden-gueviruses,usingmosquitocellcultures.AmJTropMedHyg 1979;28:1053-9.
18. ChakravartiA,KumariaR,KarP,BatraVV,VermaV.Improved Detection of Dengue Virus Serotypes From Serum Samples -EvaluationofSingleTubeMultiplexRT-PCRwithCellCul-ture.WHODengueBull2006;30:133-40.
19. DomingoC,PalaciosG,NiedrigMet al.ANewToolforthe
DiagnosisandMolecularSurveillanceofDengueInfectionsin ClinicalSamples.WHODengueBull2004;28:87-95. 20. Chakravarti A, Kumaria R. Eco-epidemiological analysis of
dengue infection during an outbreak of dengue fever, India. VirologyJ2005;2:32;1-7.(Online).
21. GrahamRR,JuffrieM,TanRet al
inYogyakarta,IndonesiaStudiesin1995-1996.AmJTropMed Hyg1999;61:412-19.
22. KalayanaroojS,NimmannityaS.Clinicalandlaboratorypres-entations of dengue patients with different serotypes.WHO DengueBulletin.2000;24:53-9.
23. Vaughn DW, Green S, Kalayanarooj S et al. Dengue viremia
titer,antibodyresponsepattern,andvirusserotypecorrelate withdiseaseseverity.JInfectDis2000;181:2-9.
24. Nisalak A, Endy TP, Nimmanniya Set al. Serotype-specific
dengueviruscirculationanddenguediseaseinBangkok,Thai-landfrom1973-1979.AmJTropMedHyg2003;68:191-202.
25. Corwin AL, Larasati RP, Bangs MJet al. Epidemic dengue
transmission in southern Sumatra, Indonesia. Trans R Soc TropMedHyg2001;95:257-65.
26. LeiHY,YehTM,LiuaHS,LinYS,ChenSH,LiuCC.Immuno-pathogenesis of DengueVirus Infection. J Biomed Sci 2001; 8:377-88.
27. HuangYH,LiuCC,WangST,LeiHY,LiuHL,LinYS.Acti-vationofcoagulationandfibrinolysisduringdenguevirus infection.JMedVirol2001;63:247-51.
28. LanciottiRS,LewisJL,GublerDJ,TrentDW.Molecularevolu-tionandepidemiologyofdengue-3viruses.JGenVirol1994; 75:65-75.
29. PeyrefitteCN,Couissinier-ParisP,Mercier-PerennecVet al.