O
r
IG
IN
A
l
A
r
T
IC
lE
ABSTRACT
Thispaperreportstheresultsofenvironmentalsurveillanceofyeastsinspecificareasoftwoterti- arylocalhospitals.FromMarch2007toFebruary2008,samplesfromtheairoftwopublichospi-talswerecollectedonamonthlybasis.Thesampleswerecollectedthroughpassivesedimentation method(dayandnightexposure)ofPetridishes.Atotalof240airsamplesfrom10hospitalenvi-ronmentswereanalyzed.Theseenvironmentspresentedsimilarcontaminationlevels,fromwhich 80fungiisolateswereisolated:Candida parapsilosis(n=34),Rhodotorula spp.(19),Trichosporon asahii (11),C. tropicalis(8),C. albicans(4),C. glabrata(1),C. guilliermondii(1),C. krusei(1)and
Saccharomycesspp.(1).Regardingthepresenceofyeastsandclimaticconditions,therewere40strains (50%)insemi-criticalareas(naturalventilation)andcriticalareas(airconditioned).Considering thepresenceofmicroorganismswithpathogenicpotential,environmentalmonitoringisnecessary topreventpossiblehospitalinfections.
Keywords:environmentalmonitoring,aircontamination,yeasts,hospitals.
[Braz J Infect Dis 2010;14(1):30-34]©ElsevierEditoraLtda. Authors
RossanaACordeiro1,2,3
RaimundaSN Brilhante1,2,3
LydiaDMPantoja2
RenatoEMoreiraFilho1,2
PatríciaRNVieira1
MarcosFGRocha1,2,4
AndréJMonteiro5
JoséJCSidrim1,2,3
1SpecializedMedical MycologyCenter, UniversidadeFederaldo Ceará,Fortaleza,Ceará, Brazil.
2PostgraduateProgram
inMedicalMicrobiology, UniversidadeFederaldo Ceará,Fortaleza,Ceará, Brazil.
3PostgraduateProgram
inMedicalScience, UniversidadeFederaldo Ceará,Fortaleza,Ceará, Brazil
4PostgraduateProgram inVeterinaryScience, UniversidadeEstadualdo Ceará,Fortaleza,Ceará, Brazil.
5DepartmentofStatistics
andAppliedMathematics, UniversidadeFederaldo Ceará,Fortaleza,Ceará, Brazil.
Submittedon:05/03/2009 Approvedon:11/13/2009
Correspondence to:
RaimundaSâmiaN. Brilhante,Prof, RuaBarãodeCanindé, 210;Montese CEP:60.425-540. Fortaleza,CE, E-mail:brilhante@ufc.br
Wedeclarenoconflict ofinterest.
INTRODUCTION
The importance of bio-aerosols has been em-phasizedinrecentdecadesduetotheireffecton humanhealth.Theyhavebeenimplicatedincon- ditionsrangingfromallergiestodisseminatedin-fectionsinsusceptiblepatients.1Differentauthors
have reported the importance of these particles astransmittersofhospitalinfections,2,3especially
thosecausedbyfungalisolates,astheyactasepi-demiologicmarkersofmicrobialcontamination.4
Fungal infections of hospital origin have been gaining importance in recent years due to their progressiveincreaseandtheirhighratesofmor-bidityandmortality.5-7
In the hospital environment, the airborne microbiota is formed mainly by filamentous fungi,especiallythosebelongingtothegenera As-pergillus, Cladosporium, Paecilomyces, Penicillium,
Scopulariopsis.8,9Yeasts have also been found, of
the generaCandida, Rhodotorula, Cryptococcus
andTrichosporon,2,7,10,11,12 although there is less
understandingabouthowtheyremainsuspended intheair.Allthementionedgenerahavebeende-scribedaspotentialhumanpathogens,8especially
theCandida genus,whichisthemaincausative
agentofhospitalfungemia.13
Various studies suggest that the distribu-tion of fungi in the air, in quantitative and qualitative terms, varies among geographic areas, and is also influenced by seasonal en-vironmental and climatic factors,14,15 such as
temperature, air humidity, time of day, wind speedanddirection,presenceofhumanactiv-ity,andtypeofventilationinclosedspaces.16
Thedifficultyofestablishingareliablemeth-odtomeasuretheexposuretofungalparticles intheairisoneofthemainimpedimentsto clarifyingtherealimpactofexposureonthe
developmentoffungalinfections.17
Despite the need to monitor bio-aerosol levelsinevaluatinghealthrisks,differencesbe- tweenautomatictechniquesandpassivesedi-mentation techniques hamper results
com-parison.18 Automated techniques, although
they are efficient in quantitative analysis, are oflimitedusebecausetheyrequireheavyand noisy equipment and need a constant power
supply.17Thepassivesedimentationtechnique
is also limited because it does not permit an adequate quantitative analysis, but it is still widelyrecommendedintheliteratureforuse asamicrobiologicalalert.7,9
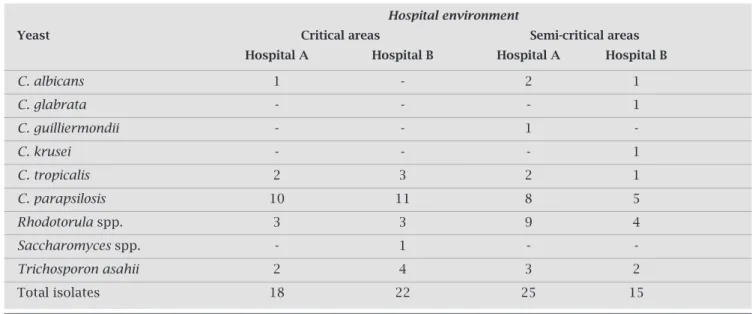
Table 1. Isolated yeasts in different areas of hospital A and hospital B from March/2007 to February/2008
Hospital environment
Yeast Critical areas Semi-critical areas Hospital A Hospital B Hospital A Hospital B
C. albicans 1 - 2 1
C. glabrata - - - 1
C. guilliermondii - - 1
-C. krusei - - - 1
C. tropicalis 2 3 2 1
C. parapsilosis 10 11 8 5
Rhodotorula spp. 3 3 9 4
Saccharomyces spp. - 1 -
-Trichosporon asahii 2 4 3 2
Total isolates 18 22 25 15
Therefore, through the passive sedimentation method, bio-aerosol monitoring in hospitals can provide informa- tionforepidemiologicalinvestigationofnosocomialinfec-tious diseases and research into airborne microorganism spreadandcontrol,aswellasforqualitycontrolmeasures.19
Thisstudyinvolvedtheenvironmentalmonitoringofyeasts inspecificareasoftwotertiarylocalhospitals.
MATERIAL AND METHODS
Ethical aspects
Thisstudywassubmittedtoanalysisoftheresearchethics committees of the two institutions and obtained approval inFebruary2007.
Hospital environments
Thepresentstudywasconductedintwotertiaryhospitals inthecityofFortaleza,Ceará,NortheastBrazil(HospitalA andHospitalB).Theairsamplingoccurredduringtwelve months(March2007toFebruary2008),and240Petridishes wereanalyzedbothdaytimeandnighttime.Ineachhospital, fivesectorswereanalyzedandgroupedaccordingtotherisk ofhospitalinfection20into:criticalareas(ICUandsurgical
ward),whichwereairconditioned,withoutnegativepres-sureandwithouttheuseofHEPAfiltering,andsemi-critical areas(infirmaries),allwithnaturalventilation.
Air sampling
Airsamplingfromdifferentareaswasperformedattwo times, one during the morning, immediately after the cleaning and disinfecting service was carried out (8 to
10a.m.),andanotheratnight(8to10p.m.),attheend of the regular shift, when there was no cleaning serv-ice.Samplecollectionwasperformedusingthepassive sedimentationmethodin150mmdiameterPetridishes containing Sabouraud dextrose agar medium (Sanofi®, France), supplemented with vancomycin (0.1 g/L) and polymyxin B (0.0075 g/L). The plates were exposed in each of the environments for two hours in each peri-od, positioned 2 m high – roughly human respiration height.14 The Petri dishes were sealed and sent to the
SpecializedMedicalMycologyCenter(CEMM),Univer-sidadeFederaldoCeará.
Fungal isolation and identification
The samples were then incubated at 28o
C for 10 days. A triage based on macroscopic characteristics was per-formed after growing the colonies on Petri dishes con- tainingSabourauddextroseagar(Sanofi®,France)toiso-late all possible yeasts in each plate. All colonies with a glabrousaspectwereculturedagaininpotatoagarslants (Himedia®,India).Theyeastisolateswereidentifiedac- cordingtotheirmorphologicalcharacteristics,biochemi-calprofile,andgrowthindifferentialculturemedia.For
identificationofCandidaspecies,thefollowingtestswere
conducted: germinative tube, microculture in cornmeal agar with Tween 80 (Difco®, USA), growth in CHRO-Magar® (Candida, Paris, France), auxonogram,
zimo-gramsubcultures,andnitrogenassimilation.21
Identifica-tionoftheRhodotorula, SaccharomycesandTrichosporon
species was based on morphological characteristics and biochemicalprofile.22
RESULTS
Thecriticalandsemi-criticalareaspresentedthesamenumber ofyeasts(40strainsperarea).Fourdifferentyeastgenerawere isolated:Candida(n=49),Rhodotorula(n=19),Trichosporon (n=11)andSaccharomyces(n=1)(Table1).
Thesemi-criticalareasofHospitalA(n=25)hadagreat-ernumberofstrainsthanthesemi-criticalareasofHospital B(n=15).Allthegenerawerepresentintheseareas,except
Saccharomyces.Therewerealsomoreyeastspresentinthese
areasatnight(n=26)thanindaytime(n=14).
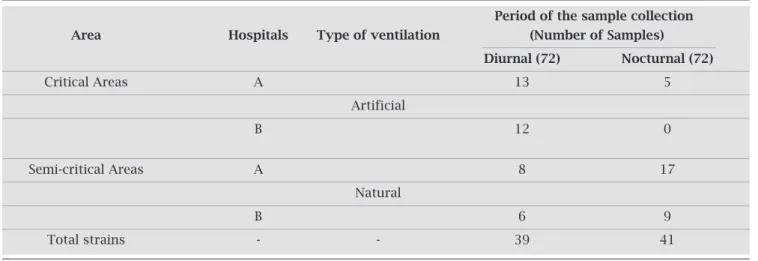
Incriticalareas,therewerefeweryeastsisolatedatHos-pitalA(n=18)thanatHospitalB(n=22).Allthespecies ofyeastswerepresent,exceptforC. glabrata,C. guilliermon-dii, andC. krusei. Hospital B showed an even distribution inrelationtothenumberorstrainsandcollectionperiod: 12and10strainsindaytimeandatnight,respectively.The distributionatHospitalAwasskewedtodaytime(n=13)in relationtothenightperiod(n=5).
Thenumberofyeastswasequallydividedaccordingto climateconditions,with40(50%)foundinareaswithnatu-ralventilation(semi-criticalareas)andthesamenumberin airconditionedareas(criticalareas).Inrelationtothepe-riodofthedaywhenthedisheswereexposed,39(48.75%) were observed in daytime samples and 41 (51.25%) were foundinnighttimesamples(Table2).
Finally,therewasnocorrelationbetweenfungaldiversity intheairbetweentherainyanddryseasons(Figure1).
DISCUSSION
In recent years, opportunistic fungal diseases have been characterized by their increased incidence, as well as the diversity of fungi isolated and infections greater severity.
YeastsoftheCandida
genusareoutstandingamongoppor-tunistic fungi.13 In the present study, other genera besides
Candidawereisolated,suchasRhodotorula, Saccharomyces
andTrichosporon.Thisisanimportantfinding,sincecontact
oftheseagentswithpatientscanstarttheinfectiousprocess,
dependingonvariousfactors,suchaspatientimmunity.11,23
Candidaisthemostimportantgenusofyeastimplicatedin
humaninfectionsandisthemajorcauseoffungemia.13Allthe
Candida
speciescancauseasimilarspectrumofdiseases,rang- ingfromthrushtoinvasivediseasessuchasarthritis,osteomy-elitis,endocarditis,endophthalmitisandmeningitis.However, theremaybedifferencesinseverityandtherapeuticoptions.24
Based on data descriptive statistical analysis, we ob-served thatC. parapsilosis (n = 34) was more frequently
isolatedthanother Candidaspecies.Thisspeciescouldbe
comingfromhumanclinicalmaterialaswellasfromenvi-ronmentalsamples.Morerecently,thisspecieshasemerged as an important nosocomial pathogen.25 Characteristics
ofC. parapsilosis that may relate to its increasing
occur- renceinnosocomialsettingsincludeskinfrequentcoloni-zation and an ability to proliferate in glucose-containing
Table 2. Number of strains isolated from different areas of hospitals A and B, regarding the type of ventilation and the period of sample collection
Period of the sample collection Area Hospitals Type of ventilation (Number of Samples)
Diurnal (72) Nocturnal (72)
Critical Areas A 13 5
Artificial
B 12 0
Semi-critical Areas A 8 17
Natural
B 6 9
Total strains - - 39 41
Figure 1: Relationship between the number of yeast strains isolated by hospital area (critical and semi-critical) and according to rainfall level in Fortaleza, Ceará from March 2007.
Number of Yeast
rainfall (mm)
mar/07 rainyapr/07 rainymay/07 rainyjun/07 Post-rainyjul/07 Dryaug/07 Drysept/07 Dryoct/07 Drynov/07 Drydec/07 Dryjan/08 Pre-rainyfeb/08 Post-rainy
rainfall 9
8
7
6
5
4
3
2
1
0
400
350
300
250
200
150
100
50
0
solutions, with a resultant increase in adherence to syn-theticmaterials.26AstudyshowedthatC. parapsilosiswas
themainisolatedagent,evidencingtheimportanceofC.
parapsilosisincandidemiaepisodesinaBraziliantertiary
carehospitalinNortheastBrazil.25
C. tropicalis(n=8)wasthesecondmostisolated
Can-dida speciesfromthecriticalandsemi-criticalareasofHos-pitalAandB,followedbyC. albicans(n=4),isolatedfrom thesemi-criticalareasofHospitalAandBandcriticalareas of Hospital A. These species are considered the principal
sourcesofcandidiasisinhumans.27Theothersspeciesof
theCandidagenusfound(C. glabrata, C. guilliermondiiand
C. krusei)occurredonlyonce(n=1).ThesethreeCandida
speciescanalsocausemucosalorsystemicinfections.24,28
Thenumberofinfectionscausedbynon-Candidayeast
species in immunosuppressed patients has increased in recent years,13 in both Brazilian and international health
facilities.Theirpresenceintheairdeservesmoreattention because they have been emerging as pathogenic fungi,29
hencethepresenceofthegeneraRhodotorula,
Saccharomy-ces,andTrichosporonstandsout.
In this context, regular environmental monitoring of hospital areas is important because the presence of bio-aerosolscanberapidlytransmittedbyair,actingasasource ofinfectiousagents.30Inparticular,yeastssuspendedinthe
aircansettledirectlyonsurgerywounds(directcontami-nation)oronthesurfaceoftheinstrumentsused,aswell asonthemedicalteam’ssurgicalgloves,andthenbetrans-mittedtothepatient.31
Accordingtotheliterature,airborneparticlescanhave manyorigins.32Inenvironmentswithartificialventilation,
theairconditioningsystem,duetocondensationtrays,has been considered an important source of microorganism distribution.Thus,inenvironmentswithartificialventila-tion,yeastsareprobablytransmittedbydropletsproduced bybio-aerosolequipment,aswellasbycoughingorsneez-ing,orevenbyroutineproceduressuchasthewithdrawal ofvascularcatheters.33Inthisenvironment,duetocontrol
of temperature and humidity, we could not observe any significantdifferencesbetweenthedayandnightperiods, exceptforthenight-timeperiodatHospitalA.
However,inenvironmentswithnaturalventilation,the principaloriginsofairparticlesarebelievedtobeventila-tors, nebulizers, air humidifiers, plant vases, some foods,
andpeoplethemselves.3Thepresenceofmoresourcesof
microorganismsinplaceswithnaturalventilationcanex- plainthegreaterdiversityofyeastspeciesfoundinsemi-criticalareasthanincriticalones.
In relation to day time period, it was established that yeasts can be transmitted in both diurnal and nocturnal periods,afactthatcanbeexplainedbythesmalltempera-turevariationbetweenthedayandnightperiods.
Therewasnosignificantrelationshipbetweenthecli-matic data and air quality. This can be explained by the lackofwell-definedseasonsinnortheastBrazil.Neverthe-less,itdisagreeswithotherstudies,whichshowthatfungal coloniesintheregionvarysignificantlywiththeseasonof theyear.15
Finally,thehospitalareasanalyzedpresentedasimilar
contaminationlevel,withtheCandidagenusbeingthemost
common,especiallyC. parapsilosis.Therefore,considering
thepresenceofthesemicroorganismswithpathogenicpo-tential,airmonitoringisessentialtohelppreventhospital infections.Amongthepreventivemeasuresthatshouldbe consideredaremorefrequentmechanicalremovalofdust, better cleaning and maintenance of air conditioners and otherequipmentthatproducebio-aerosols,correctionof roomhumidity,useofprotectiveclothingbyhospitalper-sonnel, and better staff training regarding the threats of infectionbyairbornemicroorganisms.
ACKNOWLEDGEMENTS
The authors thank CAPES (Coordination Office for Uni-versityImprovement)fortheresearchgrant.Thisworkwas supportedbytheNationalScientificandTechnologicalRe-searchCouncil(CNPq),Process473881/2008-0.
REFERENCES
1. GangneuxJP,BousseauA,CornilletA,Kauffmann-Lacroix C. Control of fungal environmental risk in French hospi-tals.JMycolMédicale2006;16:204-11.
2. DacarroC,PiccoAM,GrisoliP,RodolfiM.Determination ofaerialmicrobiologicalcontaminationinscholasticsports environments.JApplMicrobiol2003;95:904-12.
3. AfonsoMSM,SouzaACS,TippleAFV,MachadoEA,Lucas EA.Condicionamentodearemsalasdeoperaçãoecontrole deinfecção–umarevisão.RevEletrEnf2006;8:134-43. 4. Brasil,MinistériodaSaúdeResolução.n.176,24out.2000.
Agência Nacional de Vigilância Sanitária. Orientação téc-nicasobrepadrõesreferenciaisdequalidadedoarinterior emambientesclimatizadosartificialmentedeusopúblicoe coletivo2000.(Of.El.N.370/2000).
5. PfallerMA.Nosocomialcandidiasis:emergingspecies,res-ervoir, and modes of transmission. Clin Infect Dis 1996; 22:89-94.
6. Colombo AL. Epidemiology and treatment of hematog-enouscandidiasis:aBrazilianperspective.BrazJInfectDis 2000;4:113-8.
7. CentenoS,MachadoS.Assessmentofairbonemycoflorain criticalareasoftheprincipalhospitalofCumaná,stateof Sucre,VenezuelaInvestClin2004;45:137-44.
8. RainerJ,PeintnerU,PöderR.Biodiversityandconcentra- tionofairbonefungiinahospitalenvironment.Mycopath-ologia2001;149:87-97.
9. Sanca S, Asan A, Otkun MT, Ture M. Monitoring Indoor Airborne Fungi and Bacteria in the Different Areas of Trakya University Hospital, Edirne, Turkey. Indoor Built Environ2002;11:285-92.
10. KrajewskaK,Krajewska-KulakE,LukaszukC,RolkaH,Lach J,KarczewskiJ.Occurrenceoffungalpathogensinthedelivery roomsofahospitalobstetricsdepartment.GinekolPol2004; 75:451-6.
11. PiniG,FaggiE,DonatoR,FanciR.IsolationofTrichosporonin ahematologyward.Mycoses2005;48:45-9.
12. WangCY,WuHD,HsuehPR.Nosocomialtransmissionofcryp-tococcosis.NEnglJMed2005;352:1271-2.
13. MorettiML.Aimportânciacrescentedasinfecçõesfúngicas.Rev PanamInfectol2007;9:8-9.
14. Pei-ChihW, Huey-Jen S, Chia-Yin L. Characteristics of indoor andoutdoorairbonefungiatsuburbanandurbanhomesintwo seasons.TheScioftheTotalEnviron2000;253:111-8.
15. HuangCY,LeeCC,LiFC,MaYP,SuHJJ.Theseasonaldistribu- tionofbio-aerosolsinmunicipallandfillsites:a3-yrstudy.At-mospEnvironm2002;36:4385-95.
16. ButtnerMP,StetzenbachLD.Monitoringairbonefungalspores in an experimental indoor environment to evaluate sampling methodsandtheeffectsofhumanactivityonairsampling.Appl EnvironMicrob1993;59:219-26.
17. TavoraLGF,GambaleW,Heins-VaccariEM,et al.Comparative performanceoftwoairsamplersformonitoringairbornefungal propagules.BrazJMedBiolRes2003;36:613-6.
18. EduardW,HeederikD.Methodsforquantitativeassessmentof airbonelevelsofnoninfectiousmicroorganimsinhighlycontam-inatedworkenvironments.AmIndHygAssocJ1998;59:113-27. 19. LiC,HouP.Bio-aerosolcharacteristicsinhospitalcleanrooms.
TheScioftheTotalEnviron2003;305:169-76.
20. Brasil,MinistériodaSaúde.CoordenaçãodeControledeInfecção Hospitalar.ManualdeControledeInfecçãoHospitalar.Brasília, DistritoFederal.1985.
21. GautretP,RodierMH,Kauffmann-LacroixC,JacqueminJL.Case reportandreview.OnychomycosisduetoCandidaparapsilosis. Mycoses2000;43:433-5.
22. HoogGS,GuarroJ,GeneJ,FigueirasMJ.AtlasofClinicalFungi. Baarn/Delft: Centraalbureau voor Schinmelculture/Universitat RoviraiVirgilli,2.ed.1126pp.2000.
23. PletinexM,LegeinJ,VandenplasY.Fungemiawith
Saccharomy-ces boulardii ina1-year-old-girlwithprotacteddiarrhea.JPediatr
GastroenterolNutr1995;21:113-5.
24. PfallerMA,YuWL.Antifungalsusceptibilitytesting.Newtech-nology and clinical applications. Infect Dis Clin N Am 2001; 15:1227-61.
25. MedranoDJA,BrilhanteRSN,CordeiroRA,RochaMFG,Rab-enhorstSHB,SidrimJJC.Candidemiainabrazilianhospital:the importanceofCandidaparapsilosis.RevInstMedTropSãoPaulo 2006;48:17-20.
26. Weems JJ.Candida parapsilosis: epidemiology, pathogenicity, clinicalmanifestationsandantimicrobialsusceptibility.ClinIn-fectDis1992;14:756-66.
27. CalderoneRA,FonziWA.VirulencefactorsofCandidaalbicans. TrendsMicrobiol2001;9:327-35.
28. GeigerAM,FoxmanB,SobelJD.Chronicvulvovaginalcandidia-sis:characteristicsofwomenwithCandida albicans,Candida
gla-brata,andno-Candida.GenitourinMed1995;71:304-7.
29. NguyenMH,PeacockJE,MorrisAJet al.Thechangingfaceof candidemia:emergenceofnon-Candida albicans speciesandan-tifungalresistance.AmJMed1996;100:617-23.
30. AlbertiC,BouaklineA,RibauadPet al .Relationshipbetweenen-vironmentalfungalcontaminationandtheincidenceofinvasive aspergillosisinhematologypatients.JHospInfect2001;48:198-206.
31. OwersKL,JamesE,BannisterGC.Sourceofbacterialsheddingin laminarflowtheatres.JHospInfect2004;58:230-2.
32. MoscatoU.Hygienicmanagementofairconditioningsystems. AnnIg2000;12:155-74.