IMMUNOHISTOCHEMISTRY STUDIES ON BOVINE SQUAMOUS CELL CARCINOMA MORPHOLOGICAL CHARACTERIZATION OF EPIDERMAL
CELL PROLIFERATION AND DIFFERENTIATION MARKERS AND
CHARACTERIZATION OF CYTOKERATINS
H.Vala (1), D. Fondevilla(2), T. Carvalho(3), C. Pinto(4), C. Peleteiro(3), M. Pinho(3) and L. Ferrer(2)+
1 - Escola Superior Agrária de Viseu Instituto Politécnico de Viseu, 3500 Viseu, Portugal
2 - Departament de Medicina i Cirugia Animals. Facultat de Veterinaria (UAB), 08193 Bellaterra. Barcelona, Espanha
3 – Centro Interdisciplinar de Investigação em Sanidade Animal, Faculdade de Medicina Veterinária, Rua Prof. Cid dos Santos, 1300-477 Lisboa, Portugal 4 – Serviço de Desenvolvimento Agrário de S. Miguel, Direcção Regional d o Desenvolvimento Agrário, Angra do Heroísmo, Açores
INTRODUCTION
Bovine Ocular Squamous Cell Carcinoma (OSCC) is a general designation for a group of primary neoplasias of keratinocytes arising from ocular tissues, especially the lids and particularly the third eye lid. OSCC has been diagnosed all over the world with high prevalence, being the most common bovine tumour and the one causing the most significant economic losses (Hamir & Parry, 1980; Dennis et al., 1985, Heeney & Valli, 1985; Wilcock, 1993).
In Portugal, the frequency of these tumours is particularly high in the Azores, where in S. Miguel Island a large number of cattle affected with OSCC is rejected for consumption at slaughter. OSCC is the second most frequent neoplasia after urinary bladder tumours, representing 21% of all cases of rejection due to neoplasia (Pinto et al, 1996). Several reasons have been advanced to explain this situation namely the fact that animals stay in pasture all year around, with a prolonged exposition to day light and benefiting from few shelters. The ingestion of toxic plants present the pasture could also give rise to photosensitazation problems, either primary or due to hepatic toxicity, that could generate predisposing conditions to the development of OSCC.
Although being a frequent tumour its genesis has not been fully investigated, namely what are the phenotypical changes evident in neoplastic keratinocytes when compared
with normal ones, especially in what concerns the capacity and the orientation of the differentiation process.
OBJECTIVES
The objectives of the present project are to use immunohistochemical techniques with the purpose of:
confirming the origin of neoplastic cells and their degree of differentiation using cytokeratin markers for non differentiated and for well differentiated epithelia; establishing how the neoplastic changes affect the differentiation of the epithelial cells using markers for:
involucrin, marker for keratinocytes of the stratum spinosum and the stratum granulosum of normal keratinized epithelia;
filaggrin, marker for keratinocytes of the granular cell layer of normal keratinized epithelia;
calmodulin, marker of the oral epithelia and hyperproliferative keratinocytes; relate the presence of the above mentioned markers with the degree of differentiation of the neoplasias;
evaluating of the degree of malignancy using cell division markers, such as Ki67. evaluating the possibility of using immunohistochemistry as a system for diagnosis and decision making regarding rejection at slaughter.
MATERIAL AND METHODS
Samples and routine histopathological diagnosis
Nineteen tumours of the eye were collected at the Matadouro Frigorífico e Industrial of Ponta Delgada, S. Miguel Island, Azores, from Holstein Friesian cattle, age varying from 4 to 10 years old (mean age 7,4). The anatomic origin of the neoplasias was the following: eyelids - eight (42,1%), third eyelid - eight (42,1%) and three very exuberant and extensive neoplasias (15,9%) involving the whole of the ocular region. Samples were fixed in 10% formalin, included in paraffin and 5µm sections were stained for routine histopathological diagnosis with Haematoxylin and eosin.
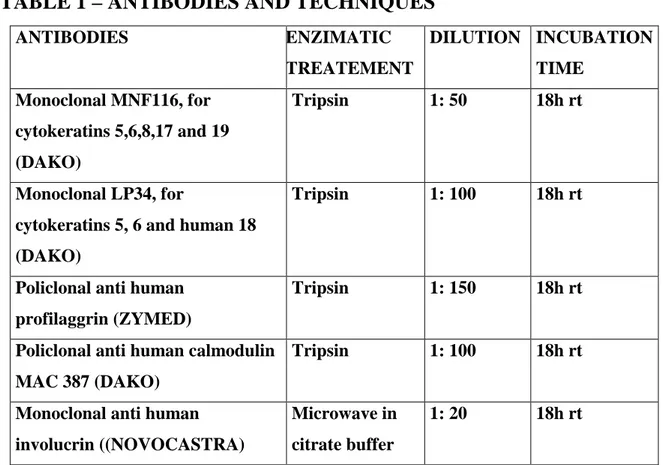
Indirect technique was applied to tissue sections fixed to glass slides with Vectabond.(Vector). Primary antibodies, pre treatments applied, dilution and incubation time are showed in the annex table:
Secondary antibody used was Multilink by DAKO, at a dilution of 1:100, and final marking was obtained with Avidin/Biotin Complex also by DAKO, peroxidase presence being revealed by the chromogen diaminobenzidine using hydrogen peroxide as substrate. Background staining was obtained with Mayer's Haemalumen.
TABLE 1 – ANTIBODIES AND TECHNIQUES
ANTIBODIES ENZIMATIC TREATEMENT DILUTION INCUBATION TIME Monoclonal MNF116, for cytokeratins 5,6,8,17 and 19 (DAKO) Tripsin 1: 50 18h rt Monoclonal LP34, for
cytokeratins 5, 6 and human 18 (DAKO)
Tripsin 1: 100 18h rt
Policlonal anti human profilaggrin (ZYMED)
Tripsin 1: 150 18h rt
Policlonal anti human calmodulin MAC 387 (DAKO)
Tripsin 1: 100 18h rt
Monoclonal anti human involucrin ((NOVOCASTRA) Microwave in citrate buffer 1: 20 18h rt RESULTS Histopathological diagnosis
All tumours sampled revealed to be OSCC varying in their degree of differentiation and infiltration of the surrounding tissues according to Table 2.
OSCC were classified as well differentiated if a considerable number of keratin pearls were formed in the centre of concentric lamellae of keratinised cells. Moderately differentiated OSCC presented a reduced number of keratin pearls and poorly
differentiated tumours showed only reduced areas of keratinisation, often involving single individual cells.
Immunohistochemistry
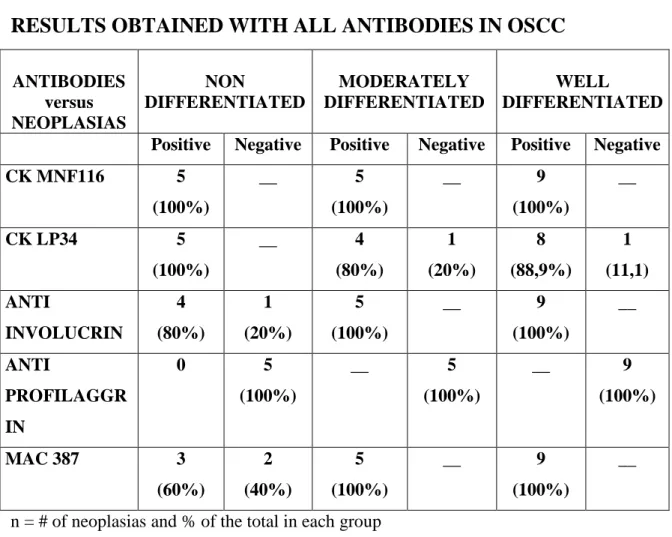
The results obtained are resumed in Table3.
TABLE 2 – DISTRIBUTION OF OSCC REGARDING THE
DEGREE OF DIFFERENTIATION AND INVASIVENESS
NOT DIFFERENTIATED MODERATELY DIFFERENTIATED WELL DIFFERENTIATED TOTAL NOT INVASIVE 3 3 (15,8%) MODERATELY INVASIVE 1 2 3 (15,8%) VERY INVASIVE 5 4 4 13 (68,4%) TOTAL 5 (26,3%) 5 (26,3%) 9 (47,4%) 19
TABLE 3 – SUMMARY OF THE IMMUNOHISTOCHEMISTRY
RESULTS OBTAINED WITH ALL ANTIBODIES IN OSCC
ANTIBODIES versus NEOPLASIAS NON DIFFERENTIATED MODERATELY DIFFERENTIATED WELL DIFFERENTIATED Positive Negative Positive Negative Positive Negative CK MNF116 5 (100%) __ 5 (100%) __ 9 (100%) __ CK LP34 5 (100%) __ 4 (80%) 1 (20%) 8 (88,9%) 1 (11,1) ANTI INVOLUCRIN 4 (80%) 1 (20%) 5 (100%) __ 9 (100%) __ ANTI PROFILAGGR IN 0 5 (100%) __ 5 (100%) __ 9 (100%) MAC 387 3 (60%) 2 (40%) 5 (100%) __ 9 (100%) __
Comments on these results are:
Most antibodies, with the exception of the anti-profilagrin, reacted with the various types of OSCC;
The antibody marking the less differentiated epithelia, MNF 116, was positive in all neoplasias independently of their degree of differentiation.
The antibody marking the more differentiated epithelia, LP34, was positive in all neoplasias except for one moderately and one non differentiated OSCC.
The anti involucrin antibody was positive in all but one non differentiated OSCC.
The anti calmodulin antibody was positive for all but two non differentiated OSCC.
One of the non differentiated OSCC negative for LP34 was also negative for MAC387, but strongly positive for involucrin.
None of the other cases of OSCC marking negatively were coincident.
Detailed results of the MNF116 and LP34 antibodies, regarding the intensity of the staining and the percentage of marked cells the are presented in Tables 4 and 5 (results concerning other antibodies are still being evaluated).
Comments on these results are:
Both antibodies for well differentiated and less differentiated epithelia marked positively the large keratinocytes in the OSCC, namely the large cells with homogenous eosinophilic cytoplasm with large vesicular nuclei, one or more prominent nucleoli and clear intercellular bridges.
Only MNF 116 marked the small basal type cells located at the periphery of the tumour trabecullae or cords. These showed basophilic cytoplasm and condensed nucleus.
Results obtained with LP34 could be useful in identifying cases of poorly differentiated OSCC, allowing for a more substantiated decision whenever regarding total or partial rejection of the carcass. In fact, as may be evaluated from the results of the present work, simple tumour morphology may be insufficient to corroborate the classification of a OSCC, as negative marking was obtained in a moderately differentiated tumour and also in a well differentiated tumour.
BIBLIOGRAPHY:
Dennis MW, Lueker DC, Kainer RA (1985). Host response to bovine ocular squamous cell carcinoma. Am J Vet Res. Sep;46(9):1975-9.
Hamir AN, Parry OB (1980). An abattoir study of bovine neoplasms wíth particular reference to ocular
squamous cell carcinoma in Canada. Vet Rec. Jun 28;106(26):551-3.
Heeney J L; Valli VEO (1985). Bovine ocular squamous cell carcinoma: an epidemiological perspective. Can J Comp Med. 49: 21-26.
Itoiz, M. E.; Conti, C. J.; Lanfranchi, H. E.; Mamrack, M.; Klein-Szanto, A. J. P.
Immunohistochemical detection of filaggrin in preneoplasic and neoplastic lesions of the human oral mucosa. American Journal of Pathology 1985; 119: p. 456-461.
Klein-Szanto, A. J. P.; Barr, R. J.; Reiners, J. J.; Mamrack, M. D. (1984). Filaggrin distribution in
keratoacanthomas and squamous cell carcinoma. Arch Pathol Lab Med. 108: 888-890.
Kvedar, J. C.; Fewkes, J.; Baden, H. P. Immunologic detection of markers of keratinocyte
differentiation. Arch Pathol Lab Med 1986; 110: p. 183 - 188.
Ohnishi, T.; Watanabe, S. Immunohistochemical characterization of keratin expression in clear cell
acanthoma. British Journal of Dermatology 1995; 133: 186 - 193.
Pinto C ; Medeiros F ; Freitas F; Cabral A; Oliveira V; Rebimbas F (1996). Neoplasias em bovinos
de S. Miguel – Açores. I-Estudo retrospectivo e situação actual. Veterinária Técnica. 6(1): 30-37.
Rice RH, Qin Q, Pilato A, Rubin AL (1992). Keratinocyte differentiation markers: involucrin,
transglutaminase, and toxicity. J Natl Cancer Inst Monogr. (13):87-91.
Simon, M. Green, H. Participation of membrane-associated proteins in the formation of the cross-linked
envelope of the keratinocyte. Cell 1984; 36: p. 827 - 834.
Smith, S. A.; Dale, B. A. Immunologic localization of filaggrin in human oral epithelia and correlation
with keratinization. The Society for Investigative Dermatology, Inc 1986; 86: p. 168-171.
Vigneswaran N, Peters KP, Hornstein OP, Haneke E (1989). Comparison of cytokeratin, filaggrin and
involucrin profiles in oral leukoplakias and squamous carcinomas. J Oral Pathol Med. Aug;18(7):377-90.
Wilcock BP (1993). The eye and ear, in Jubb, K. V. F.; Kennedy, P. C. and Palmer, N. (Eds). Pathology of domestic animals (4ª Edição), San Diego. Academic Press, Inc. Vol. I, p. 512- 515.
AKNOWLEDGEMENTS
The authors wish to thank the histopathology technicians of both Faculdade de Medicina Veterinária, Lisbon and Facultad de Veterinaria, Barcelona.
They also wish to thank João Orvalho for assistance in the macroscopy photography digitalisation.