www.jped.com.br
ORIGINAL
ARTICLE
Empirical
antimicrobial
therapy
for
late-onset
sepsis
in
a
neonatal
unit
with
high
prevalence
of
coagulase-negative
Staphylococcus
夽
,
夽夽
Roberta
Maia
de
Castro
Romanelli
a,b,∗,
Lêni
Márcia
Anchieta
a,c,
Ana
Carolina
Bueno
e
Silva
a,
Lenize
Adriana
de
Jesus
d,
Viviane
Rosado
d,e,
Wanessa
Trindade
Clemente
d,faUniversidadeFederaldeMinasGerais(UFMG),FaculdadedeMedicina,DepartamentodePediatria,BeloHorizonte,MG,Brazil bUniversidadeJosédoRosárioVellano,FaculdadedeCiênciasMédicas,BeloHorizonte,MG,Brazil
cUniversidadeFederaldeMinasGerais(UFMG),HospitaldasClínicas,UnidadeNeonataldeCuidadosProgressivos,Belo
Horizonte,MG,Brazil
dUniversidadeFederaldeMinasGerais(UFMG),HospitaldasClínicas,ComissãodeControledeInfecc¸ãoHospitalar,
BeloHorizonte,MG,Brazil
eUniversidadeFederaldeMinasGerais(UFMG),FaculdadedeMedicina,ProgramadePós-Graduac¸ãoemCiênciasdaSaúde,
BeloHorizonte,MG,Brazil
fUniversidadeFederaldeMinasGerais(UFMG),FaculdadedeMedicina,DepartamentodePropedêuticaComplementar,
BeloHorizonte,MG,Brazil
Received13October2015;accepted18January2016 Availableonline22April2016
KEYWORDS
Sepsis; Neonate; Staphylococcus; Oxacillin; Vancomycin
Abstract
Objective: Theaimofthisstudywastocomparetwodifferentempirictreatmentsfor late-onsetneonatalsepsis,vancomycinandoxacillin,inaneonatalintensivecareunitwithahigh prevalenceofcoagulase-negativeStaphylococcus.
Methods: Across-sectionalstudywasconductedinanneonatalintensivecareunitfrom2011 to2014.Data fromthemedicalrecordsofat-risk newbornswere collecteddaily. Infections weredefinedaccordingtotheNationalHealthSurveillanceAgencycriteria.Dataanalysiswas performedusinganinternalprogram.
Results: TherewasasignificantreductioninthenumberofStaphylococcusaureusinfections (p=0.008),withoutendocarditis,meningitis, orlowerrespiratory tractinfection,aswellas areductioninthefrequencyofdeathsrelatedtoS.aureusinfection.Therewereno signifi-cantchangesintheincidenceofGram-negativebacterialorfungalinfections.Anincreasein coagulase-negativeStaphylococcusinfectionswasobserved(p=0.022).However,therewasno
夽
Pleasecitethisarticleas:RomanelliRM,AnchietaLM,SilvaAC,deJesusLA,RosadoV,ClementeWT.Empiricalantimicrobialtherapy
forlate-onsetsepsisinaneonatalunitwithhighprevalenceofcoagulase-negativeStaphylococcus.JPediatr(RioJ).2016;92:472---8.
夽夽
StudyconductedatFacultyofMedicine,UniversidadeFederaldeMinasGerais,BeloHorizonte,MG,Brazil.
∗Correspondingauthor.
E-mail:rmcromanelli@gmail.com(R.M.C.Romanelli). http://dx.doi.org/10.1016/j.jped.2016.01.008
measured increaseinrelatedmorbidityandmortality.There wasareductioninthemedian numberofdaysoftreatmentwithoxacillinfrom11.5to6days(p<0.001)andanincreaseof onedayinthemediannumberofdaysoftreatmentwithvancomycin(p=0.046).
Conclusions: Modificationoftheempirictreatmentregimenforneonatallate-onsetsepsiswith useofoxacillinshowedasignificantreductioninS.aureusinfections,aswellasareductionin thefrequencyofinfectionswithmajororgansysteminvolvementandmortalityduetoinfection withthismicroorganism.Asaresult,oxacillincanbeconsideredasaneffectivetreatmentfor late-onsetsepsis,makingitpossibletoavoidbroad-spectrumantibiotics.
©2016PublishedbyElsevierEditoraLtda.onbehalfofSociedadeBrasileiradePediatria.Thisis anopenaccessarticleundertheCCBY-NC-NDlicense(http://creativecommons.org/licenses/ by-nc-nd/4.0/).
PALAVRAS-CHAVE
Sepse;
Recém-nascido; Staphylococcus; Oxacilina; Vancomicina
Terapiaantimicrobianaempíricaparasepsetardianaunidadeneonatalcomalta prevalênciadeStaphylococcuscoagulasenegativo
Resumo
Objetivo: Comparar dois períodos com diferentes esquemas empíricos para tratamentode sepseneonataltardia,incluindovancomicinaouoxacilinarespectivamente,emUnidade Neona-taldereferênciacomaltaprevalênciadeStaphylococcuscoagulasenegativo.
Métodos: Estudotransversal,realizadoemUnidadeNeonataldereferência,de2011a2014.A coletadedadosfoirealizadadiariamenteporvigilânciaativaemprontuárioderecém-nascidos derisco.Asinfecc¸õesforamnotificadasconformecritériosdefinidospelaAgênciaNacionalde VigilânciaSanitária.Obancodedadoseaanáliseforamrealizadosemprogramainterno. Resultados: Ocorreu reduc¸ão significativa da notificac¸ão de infecc¸ões por Staphylococcus aureus (p=0,008), sem notificac¸ões de endocardite, meningite e infecc¸ões de vias aéreas inferiores,alémdereduc¸ãonafrequênciadeóbitospelomicro-organismoesemalterac¸ão sig-nificativanasincidênciasdeinfecc¸õesporbactériasGramnegativasefungos.Houveaumento deinfecc¸õesS.coagulasenegativo(p=0,022),massemaumentodemorbidadeemortalidade. Ocorreureduc¸ãonamedianadotempodeusodeoxacilina,de11,5para6dias(p<0,001),com aumentodemedianadeumdiadeusodevancomicina(p=0,046).
Conclusões: Amodificac¸ãodoesquemaempíricocomutilizac¸ãodeoxacilinareveloureduc¸ão significativadasinfecc¸õesporS.aureus,alémdareduc¸ãonafrequênciadeinfecc¸ãodefoco profundoemortalidadepelomicro-organismo.Considera-sequeoxacilinapodeserutilizada comoesquemadetratamentodesepseneonataltardia,evitando-seousodeantibióticosde largoespectro.
©2016PublicadoporElsevierEditoraLtda.emnomedeSociedadeBrasileiradePediatria.Este ´
eumartigoOpenAccesssobumalicenc¸aCCBY-NC-ND(http://creativecommons.org/licenses/ by-nc-nd/4.0/).
Introduction
The mostprevalentmicroorganismsin late-onsetneonatal sepsis described in international literatureare coagulase-negative Staphylococcus (CoNS).1---6 While rates of
laboratory-confirmed sepsis due to these microorgan-isms vary from30% to60%,1 higher rates have also been
reported. These microorganisms account for 77.9% of late-onset neonatal sepsis in industrialized countries and 46.5%indevelopingregions.2However,infectionscausedby
thesecommensalmicroorganismsareoftenquestioneddue todifficultiesinconfirmationanddifferencesinnotification criteria.1,7
ItshouldalsobeconsideredthatCoNSareminimally inva-sivemicroorganismsthatcolonizeinfantsafterbirthandare normallypresentinthemicrobiomesofdifferentbodysites. They have the beneficial effect of stimulating the innate immuneresponseandimproving thedefense againstother pathogens.2 However, defense mechanisms can be
inade-quateinnewborninfants,increasingtheirsusceptibilityto
infection by these microorganisms.3 On the other hand,
althoughtheyareconsideredtobemicroorganisms respon-siblefor sepsis in newborninfants, theypresent insidious evolutionandlowmorbidityandmortality.2,5,8---11
It is known that the resistance profile of CoNS can exceed90%forisoxazolylpenicillin.3Asaresult,vancomycin
hasbeenconsidered thestandard treatment.1,12 However,
vancomycin restriction as empiric therapy for late-onset neonatalsepsishasbeenindicatedinliterature.9---11,13
The objectiveof this study was to compare, epidemi-ologically,twodifferentempirictreatmentsfor late-onset sepsisinaneonatalintensivecareunit(NICU)withahigh prevalence of CoNS andoxacillin-sensitive Staphylococcus aureus.
Methods
Gerais(HC/UFMG),fromJanuary 2011toDecember2014. HC/UFMG,auniversityhospital,isaresourceformanaging highobstetricriskinBeloHorizonteandinthestateofMinas Gerais.
The target population consisted of allnewborn infants in the NICU considered at risk for late-onset sepsis. Risk factorsincludedweightunder1500g,presenceofacentral venouscatheter(CVC),useofmechanicalventilation(MV), surgery, and treatment with antimicrobial agents. These at-riskinfantswere followeddailyby specializedHospital InfectionControl Commission(HICC) staff.Datawere col-lected by active surveillance, review of medical records, anddiscussionwiththecareteam.Infectionswerereported accordingtothenationallegislation,14 withinfection
crite-riadefinedbytheAgênciaNacionaldeVigilânciaSanitária (ANVISA),15 based onthe National Healthcare Safety
Net-work(NHSN).7,16
The inclusion criteria considered all newborn infants treatedwithoxacillinor vancomycinasempirictreatment forlate-onsetsepsis,reportedafter48hoflife,asdefined byANVISA.15 ‘‘Sepsis’’and‘‘bloodstreaminfection’’were
usedsynonymously.
Asamplesizewascalculatedconsideringtheprevalence oflaboratory-confirmedCoNSandS.aureusinfectionsata rateofalmost30%and15%respectively,basedona popula-tionof300patientsatriskperyear.
An internal program of HICC was used for statistical analysis.Descriptive analysisincludedfrequencyof at-risk patients, frequency of patients with hospital-associated infections(HAI),frequencyofHAI,cumulativeincidenceof HAI(numberof HAIper100 patientsat risk)andHAI inci-dencedensity(numberofHAIper1000 patient-days).The incidence density of HAI was also stratified according to weightrangeandtopographyofinfection.Densityof infec-tionofdevice-associatedinfectionswasmeasured,including infections associated toCVC, MV, and indwellingurethral catheter(IUC).
The frequency of microorganisms has been described in groups (CoNS, S. aureus, gram-negative bacteria and fungi) and by frequency of antimicrobial agents used for empiricorspecificStaphylococcusspp. coverage(oxacillin andvancomycin).Ingeneral,theseantimicrobialagentsare empiricallyor specificallyusedfor treatment ofinfection withStaphylococcus spp. Otherantimicrobial agentswere notdescribed,astheyarenotrelevanttothisstudy.
Microbiological isolation in patient samples was per-formed by automated method (VITEK®2, BioMérieux Inc,
USA) and susceptibility testing by disk diffusion agar (Kirby---Bauer).Thesensitivityprofileofmicroorganismswas definedaccordingtothehospital’sHICCandbasedonthe ClinicalandLaboratoryStandardsInstitute(CLSI).
Forcomparativeanalysis,twoperiodsweredefined: Jan-uary2011toDecember2012(Period1)andJanuary2013to December2014(Period2).DuringPeriod1,vancomycinwas usedintheempirictreatmentregimenforlate-onsetsepsis, whileoxacillinwasusedduringPeriod2.
Mortality(deathsovertotalnumberofpatientsat risk) and lethality (deaths over total number of patients with HAI),consideringCoNSorS.aureus,werecomparedinboth periods.Deathwasconsideredassociatedwithinfectionif itoccurredduringorwithin15daysoftreatment withthe studyantibiotic.
Morbiditywasdefinedasinfectionwithmajororgan sys-tem involvement such as endocarditis, lower respiratory tractinfection(pneumonia or trachealor bronchial infec-tion),or centralnervoussysteminfectionwithisolationof thesemicroorganisms,aswellasnumberofdaysof antimi-crobialtreatmentwithoxacillinorvancomycin.
Statisticalanalysiswasperformedbyastatisticianusing SPSS®(SPSSInc.,version15.0,USA)andEpiInfo®v.7.0(CDC,
USA).Descriptiveanalysisincluded frequency,percentage, mean,standarddeviation,medianandrange.Comparative analysiswasperformedusingX2orFishertestfor
categori-calvariablesandStudent’st-testortheMann---Whitneytest forquantitativevariables,accordingtovarianceanalysisby Levine’stest.Oddsratiowasusedtocalculatetherelative measureofeventsbetweenthetwoperiodsofstudyand95% confidenceinterval(95%CI)asameasureoftheprecisionof theeventestimated.Statisticalsignificancewasconsidered whenp<0.05.
Otherpreventiveandinfection-reducingstrategieshave beencontinuouslyperformedintheNICUbyHICCstaff.This study was approved by the Institutional Review Board of ResearchEthicsCommitteeofUFMG.
Results
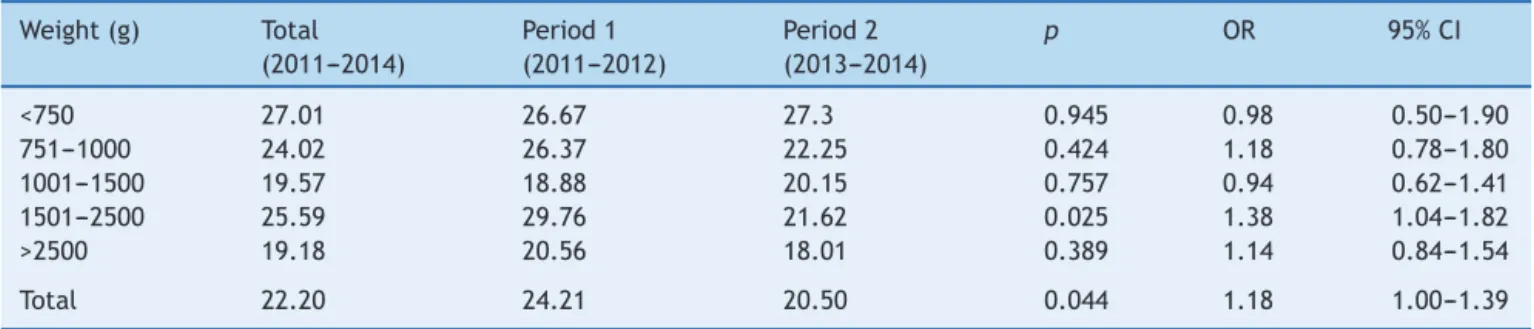
Throughoutthestudyperiod,1229at-riskpatientswere fol-lowed,totaling26,260patientdays.Atotalof367patients had583episodesofHAI,withanincidencedensityof22.20 HAI episodes per 1000 patient days. Table 1 shows HAI incidencedensityperbirthweightrange,withasignificant reductionininfectionsamongpatientsrangingfrom2500g
Table1 Incidencedensityaofhealthcare-associatedinfectionsbybirthweightrange,NeonatalIntensiveCareUnit,HC/UFMG,
2011---2014.
Weight(g) Total Period1 Period2 p OR 95%CI
(2011---2014) (2011---2012) (2013---2014)
<750 27.01 26.67 27.3 0.945 0.98 0.50---1.90
751---1000 24.02 26.37 22.25 0.424 1.18 0.78---1.80
1001---1500 19.57 18.88 20.15 0.757 0.94 0.62---1.41
1501---2500 25.59 29.76 21.62 0.025 1.38 1.04---1.82
>2500 19.18 20.56 18.01 0.389 1.14 0.84---1.54
Total 22.20 24.21 20.50 0.044 1.18 1.00---1.39
Table2 Frequencyofmicroorganismsisolatedinhealthcare-associatedinfectionsbypatientat-risk,NeonatalIntensiveCare Unit,HC/UFMG,2011---2014.
Microorganism Total Period1 Period2 p OR 95%CI
2011---2014 2011---2012 2013---2014
n(%) n(%) n(%)
CoagulasenegativeStaphylococcus 117(9.52) 40(7.37) 77(11.22) 0.028 0.63 0.41---0.95 Staphylococcusaureus 42(3.42) 27(4.97) 15(2.19) 0.012 2.34 1.18---4.67
Gramnegativebacillus 91(7.40) 47(8.66) 44(6.41) 0.167 1.38 0.88---2.17
Fungi 14(1.14) 6(1.10) 8(1.17) 0.865 0.95 0.27---3.13
to1501gduringPeriod2.Therewassignificantreductionin thetotalnumberofreportedinfections(p=0.044;OR:1.18; 95%CI1---1.39)(Table1).
Atotalof296(54.81%)and355(52.98%)ofpatientsatrisk duringPeriod1andPeriod2weremale,respectively.There wasnostatisticaldifferenceinregardstogender(p=0.564; OR:1.08;95%CI0.85---1.36),but19newbornshadnodefined gender.
The cumulativeincidence of HAI was47.44% of at-risk patients,withasignificant reductionfromthefirsttothe secondstudyperiod:from53.8%to42.4%(p<0.001).
Considering device-associated infections, there wasno significantdifferenceintheincidencedensityofinfections bydevice-daybetweenthetwoperiodsofstudy.Densityof CVC-associatedbloodstreaminfectionwas17.29infections per1000CVCdaysandvariedfrom15.39to19.09(p=0.117; OR:0.81;95%CI0.62---1.06).Densityofventilator-associated pneumoniawas1.93infectionsper1000MVdaysandvaried from2.09to1.79(p=0.789;OR:1.17;95% CI0.38---3.62). Density of IUC-associatedurinary tractinfectionwas 6.05 per1000IUCdaysandvariedfrom8.46to4.33(p=0.243; OR:1.96;95%CI0.62---6.16).
Frequency of episodesofHAI treatedwithboth antibi-otics was compared between the two periods and the implementationofthenewprotocolwasconsidered effec-tive. There was a significant decrease in the use of vancomycin, from175 to97 episodesof HAI treated with vancomycin (p<0.001; OR: 1.98; 95% CI 1.52---2.60), and therewasasignificantincreasefrom30to132episodesof lateHAI treatedwithoxacillin(p<0.001;OR:4.68;95%CI 3.07---7.17).
Consideringthefrequencyofmicroorganismsisolatedin casesofHAIinat-riskpatients,therewasasignificant reduc-tioninHAIduetoS.aureuswhenoxacillinwaspartofthe initial empiricregimen(p=0.012), andan increasein HAI due to CoNS (p=0.028), but no significant changes were observed in the ratio of gram-negative bacteria or fungi (Table2).AllS.aureusisolatedfrompatientsampleswith HAI associated infectionsin Period 1 wereoxacillin sensi-tive.DuringPeriod2,onlyonesamplepresentedanoxacillin resistance profileat sensitivity test.This reveals that the prevalenceofoxacillin-resistantS.aureusatthisNICUwas calculatedas2.4%(n=1/42).
Table3shows HAIbytopography,considering S.aureus
and CoNS associated infections. No infections with major organsysteminvolvement,suchasendocarditis,lower respi-ratorytractinfection,orcentralnervoussystem infection with isolation of S. aureus, were noted in Period 2; only conjunctivitis,skininfection,andbloodstreaminfectionby thismicroorganismwerereported.
Mortality and lethality were calculated by comparing cases of death due to HAI caused by S. aureus or CoNS betweenthe two periods (Table 4). Mortality was consid-ered to be the number of deaths associated with these infectionsper total numberof deaths. Lethalitywas con-sideredtobethenumberofdeaths associatedwiththese infectionspertotalnumberofcasesofHAIbyeach microor-ganism.There werenosignificant changes,but therewas only onedeath associated withS. aureus during Period 2 (whenempirictherapyincludedoxacillin),comparedtofive deathsinPeriod1.Nostatisticaldifferencewasobservedin mortalityassociatedwithCoNSinfection,withfewer asso-ciateddeaths.
Morbiditywasalsoevaluatedconsideringthedurationof antimicrobialtreatment.Therewasasignificantreduction indurationoftreatmentwithoxacillininPeriod2withthe mediantimereducingfrom11.5to6days(p<0.001). Dura-tionof treatment withvancomycin increasedby just one day,rangingfromeightdaysinPeriod1toninedaysinPeriod 2(p=0.046).
Discussion
Aftertheintroductionof empirictreatment withoxacillin inthetherapeutic regimenfor late-onset sepsis,a signifi-cantreductionincasesofHAIcausedbyS.aureussensitive tooxacillin(p=0.012)wasobserved.Theincidencedensity ofinfectionsalsosignificantlydecreased,anditwasmainly evidentinpatientswithweightrangingfrom1501to2500g. A previous study17 already showed a greater number of
patientsinthisweightrangeatriskforlaboratory-confirmed bloodstreaminfections,probablyduetotheprofileofthis NICU,asitisacenteroffetalmedicine.Thesepatientshave prolongedhospitalizationsandtheyareatriskofrequiring surgery,CVC,andMVthatmayincreaseriskofinfection.
Despitetheincreaseincasesofinfectionassociatedwith CoNS,noincreaseinmortalityandevaluatedmorbiditywas observed, indicating that this microorganism can be con-sideredless pathogenic andpresentsinsidious progression whencomparedtoS.aureusasreportedinliterature.1,2,12
It also shouldbe pointed out that CoNS infection may bequestionedanddiagnostic criteriarequireat least two blood cultures with the same microorganism.7,15 In
addi-tion,otherstudiesdemonstratethatitispossibletofollow clinicaloutcomes, waitfor cultureresults, andalso post-poneadministrationofantimicrobialagentsinpatientswith suspectedCoNSinfection.9---11,18
Table3 Healthcare-associatedinfectionsbyS.aureusandcoagulasenegativeStaphylococcus(CoNS)bytopography,Neonatal IntensiveCareUnit,HC/UFMG,2011---2014.
Topography Microorganism Period1 Period2 p OR 95%CI
n(%) n(%)
Conjuntivitis CoNS 5(38.5) 2(22.2) 0.648 2.19 0.23---20.0
S.aureus 3(23.1) 2(22.2) 1 1.05 0.10---12.44
Endocarditis CoNS --- --- --- ---
---S.aureus 3(100) 0 --- ---
---Necrotizingenterocolitis CoNS --- 1(6.7) --- ---
---S.aureus --- 0 --- ---
---Intra-abdominalinfection CoNS --- 1(50) --- ---
---S.aureus --- 0 --- ---
---Vascularinfection CoNS --- 3(100) --- ---
---S.aureus --- 0 --- ---
---Skinorsofttissueinfection CoNS 0 2(3.2) --- ---
---S.aureus 1(2.0) 1(1.6) 1 1.22 0.0---20.0
Bronchialortrachealinfection CoNS 0 --- --- ---
---S.aureus 1(50.0) --- --- ---
---Pneumonia CoNS 0 --- --- ---
---S.aureus 1(8.3) --- --- ---
---Urinarytractinfection CoNS 1(7.1) 2(13.3) 1 0.5 0.02---8.61
S.aureus 0 0 --- ---
---Centralnervoussysteminfection CoNS 0 2(28.6) --- ---
---S.aureus 3(15.8) 0 --- ---
---Bloodstreaminfection(sepsis) CoNS 34(21.38) 64(38.1) 0.001 0.44 0.26---0.74
S.aureus 15(9.4%) 12(7.1) 0.58 1.34 0.58---3.20
CoNS-associated infections. However, no statistical dif-ferences in percentages of infection associated with gram-negative bacteria and fungi were detected. It was considered that there was no negative interference in flora profile and, consequently, to patient exposure pro-file.Furthermore,anoverallstatisticalreductionofHAIwas achieved.Itshouldbeemphasizedthatasmallernumberof infectionsisdirectly relatedtoloweruseofantimicrobial agents.AsreportedbyPintoetal.,6criteriatoreducesepsis
includedsignificantlydecreasingtheuseofvancomycinand
carbapenemaswellasthenumberofantimicrobialagents used,althoughthoseauthorsdidnotpresentafloraprofile. As published in other reports,9,11 the present study
revealedthatempirictreatmentwithoxacillineven signif-icantly reduced the duration of treatment (p<0.001), in more than 50% of days,with increase in only one day of vancomycininPeriod2.The reductionindaysofoxacillin usecanlikelybeassociatedwiththeappropriateempiricor specificuse,sinceoxacillinhasbetterantimicrobialactivity and therapeutic efficacyin sensitive S. aureus infections.
Table4 MortalityandlethalityofpatientswithStaphylococcusspp.hospital-associatedinfections,NeonatalIntensiveCare Unit,HC/UFMG,2011---2014.
Mortality/lethality Total Period1 Period2 p OR 95%CI
2011---2014 2011---2012 2013---2014
n(%) n(%) n(%)
Generalmortality
CoagulasenegativeStaphylococcus/patient atrisk
3(0.24) 1(0.18) 2(0.29) 0.63 0.63 0.06---6.95
Staphylococcusaureus/patientatrisk 6(0.49) 5(0.92) 1(0.15) 0.08 6.32 0.74---53.91
LethalityassociatedtocoagulasenegativeStaphylococcus CoagulasenegativeStaphylococcus/totalof
infections
3(0.82) 1(0.56) 2(1.07) 1 0.52 0.02---7.32
S.coagulasenegative/totalofinfectionsby agent
3(2.56) 1(2.5) 2(2.6) 1 0.96 0---14.16
LethalityassociatedtoStaphylococcusaureus
Itisimportanttohighlight thatS.aureushasan oxacillin-sensitiveprofileinthepresentNICU,supportingthechange ofempirictherapy.
In a national study by Bentlin et al.,19 which
investi-gatedpractices relatedtoprevalence of late-onset sepsis inprematureinfants,theauthorsnotedthatcentersusing empirictherapy withoxacillinandaminoglycosideshave a lowerincidenceoflate-onsetsepsis,probablyduetomore effectivetreatment.
Considering the recommendation of isoxazolyl peni-cillinsassociatedwithgentamicinasempirictreatmentfor late-onset neonatal sepsis, a study by Chiu et al.9 also
showed a significant reduction in the use of vancomycin andincreaseduseofoxacillinwithoutsignificantdifference in neonatal morbidity and mortality as assessed by inci-denceoflate-onsetsepsis,meningitis,anddeaths.However, those authors did not compare patients with laboratory-confirmed S. aureus and CoNS-associated HAI as in this study,whichshowed asignificant reductioninepisodesS. aureus-associated infections, withas well as fewer cases ofinfectionswithmajororgansysteminvolvementsuchas endocarditis,lowerrespiratorytractinfection,andcentral nervoussysteminfection.
Inadditiontoastatisticalreductionintheoverall num-ber of HAI cases as well as HAI due to susceptible S. aureus,fewerinfectionswithmajororgansystem involve-mentwerealsoobserved,reducedmortalityassociatedwith thismicroorganismwasnoted,andasignificantreductionin daysoftreatmentdurationandhospitalizationwas demon-strated.Thesefindingscanbeconsideredadequatetoavoid morbidity associated with sepsis and exposure to other adverseevents.AsreportedbyKaufman,20accuracyin
diag-nosisandreductionof theuseofantimicrobial agentsare importanttopreventinfectionsingeneraland,specifically, neonatalmortalityrelatedtosepsis.
A multicenterstudy with348 NICUs11 evaluated
imme-diate use of vancomycin (<1 day) and delayed use (1---3 days)inthefirstepisodeoflate-onsetsepsisassociatedwith CoNS. The authors reportedthat there wasnodifference in the rates of mortality at 7 or 30 days after the initi-ationof treatmentor at discharge.Furthermore,patients whostartedtheuseofvancomycinimmediatelyhada sig-nificantly longerduration of treatment, witha medianof twoadditionaldays.Inanothermulticenterstudyconducted by the same group,18 there was no differencein
mortal-itywhen comparingthenewborns withpossible,probable or confirmed infection by CoNS. Additionally, the authors reportedthatpatientswithinfectionsassociatedwithCoNS hadsignificantly lowermortality thanpatients with nega-tivebloodcultures,consideringthathighermortalitycould beattributedtoothermicroorganismsnotisolatedinblood cultures.Itmustbeconsideredthatanantimicrobialagent maybeusedunnecessarily,aspossibleorprobableinfections byCoNSareoverestimated.
Cottenetal.,21 inamulticentercohort,foundina
mul-tivariateanalysisthatprolongeddurationofinitialtherapy in dayswasassociated withdeathof extremely lowbirth weightinfants, especiallywhentreatment with antimicro-bialagentsexceededfivedays,withincreasedoddsperday ofantimicrobialuse.Itmustalsobeconsideredthat mortal-ityincreaseswhenempirictherapyisinadequate,afactor whichcanincreasetreatmentduration.
ApreviousstudyinthesameNICUrevealedhighmortality ofpatientswithsepsiswithreportedlaboratory-confirmed
S. aureus.5 In thisstudy, a reduction in the frequency of
deathsduetoS.aureus-associatedHAIfromPeriod1(five deaths)toPeriod2(onedeath) wasnoted.Thisreduction tendedtowardsignificance(p=0.08).Theonlydeath asso-ciatedwithS.aureusinPeriod2wasthatofapatientwith severalmalformationsandpotentialtrisomy13.Duetothe clinicalseverityofthepatient’scondition,testswere per-formed to screen for infection. However, treatment with oxacillinwasinitiatedwhenthebloodcultureresultswere available,twodaysaftertesting.Thedeathmaybe associ-atedwiththeunderlyingdiseaseanddelayintheuseofan appropriateantimicrobialagent.
The present studyidentified justoneand twocasesof deathduetoCoNS-associatedHAI,respectively, inPeriods 1and2,withoutstatisticalsignificance.Literaturealso dis-closesthat mortalityassociated withinfectionby CoNS is relatedto 1% of cases.5,10 Karlowicz et al.10 investigated
causes of fulminant late-onset sepsis and observed that evenwhen cases of CoNS infection were associated with death, patients had other comorbidities or cultures grew more than one microorganism, suggesting contamination. Makhouletal.8assessedriskfactorsforearlymortalityafter
late-onsetneonatalsepsisandtheyreportedthatpatients withinfections caused byCoNS had alower risk of death whencomparedtopatientswithinfectionscausedbyother microorganisms.
InastudybyHemelsetal.,13 cefazolinassociatedwith
gentamicinwas used as empiric treatment for late-onset neonatalsepsisduetoCoNS.Clinicalresponsewasobserved in87%oftreated patientsandsensitivitytocefazolinwas observed in 88% of the strains analyzed. No difference in length of stay (0.77 days) or mortality (p=0.33) was observedamongtreatedpatients withsepsisdueto sensi-tiveorresistantstrainsofCoNS.Thoseauthorssuggestthat other beta-lactam antibiotics may be useful in the treat-mentofHAIinnewborninfants,decreasingtheneedforthe useofvancomycin.
Consideringrational useof antibiotics,vancomycinuse should be restricted in units with high prevalence of S. aureus resistant to vancomycin, in cases without clinical or laboratory response within 48---72h despite use of a beta-lactam such as oxacillin,and defined cases of CoNS resistant to oxacillin without clinical response and which is not considered to be commensal.1,22 These
recommen-dations follow the guidelines of the Centers for Disease ControlandPrevention,inordertoavoidsignificantincrease instrainsofEnterococcusspp.andotherbacteriaresistant to vancomycin.23 The restriction of utilization of
antimi-crobialagentsbasedonstewardshipisimportanttoreduce interferenceinmicrobiome,selectionofresistant microor-ganismsandatopicmanifestations,andothercomplications inprematureinfants,suchasenterocolitisanddeath.24
to the epidemiological profile of each NICU, which must be associated with other effective infection prevention practices, in order to avoid the use of broad-spectrum antimicrobialagents.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
References
1.BlanchardAC,QuachC,AutmizguineJ.Staphylococcal infec-tionsininfants:updatesandcurrentchallenges.ClinPerinatol. 2015;42:119---32,ix.
2.Dong Y, Speer CP.The roleof Staphylococcusepidermidisin neonatalsepsis:guardingangelorpathogenicdevil?IntJMed Microbiol.2014;304:513---20.
3.Venkatesh MP, Placencia F, Weisman LE. Coagulase-negative staphylococcalinfectionsintheneonateandchild:anupdate. SeminPediatrInfectDis.2006;17:120---7.
4.Stoll BJ, Hansen N. Infections in VLBW infants: studies fromtheNICHDNeonatalResearchNetwork.SeminPerinatol. 2003;27:293---301.
5.RomanelliRM,AnchietaLM,MourãoMV,CamposFA,LoyolaFC, Jesus LA, et al.[Notification ofhealthcare associated infec-tionsbasedoninternationalcriteriaperformedinareference neonatalprogressivecareunityinBeloHorizonte,MG].RevBras Epidemiol.2013;16:77---86.
6.PintoMC,BuenoAC,VieiraAA.Implementationofaprotocol proposedbytheBrazilianNationalHealthSurveillanceAgency forantibioticuseinverylowbirthweightinfants.JPediatr(Rio J).2013;89:450---5.
7.CenterforDiseasesControlandPrevention(CDC).Bloodstream
infection event (central line-associated bloodstream
infec-tion and non-central line-associated bloodstream infection).
[cited05Sep2015].Availablefrom:http://www.cdc.gov/nhsn/
PDFs/pscManual/4PSCCLABScurrent.pdf.
8.Makhoul IR,Sujov P,Smolkin T, Lusky A, ReichmanB, Israel NeonatalNetwork.Pathogen-specificearlymortalityinverylow birthweightinfantswithlate-onsetsepsis: anationalsurvey. ClinInfectDis.2005;40:218---24.
9.ChiuCH,MichelowIC,CroninJ,RingerSA,FerrisTG,Puopolo KM. Effectiveness of a guideline to reduce vancomycin use in the neonatal intensive care unit. Pediatr Infect Dis J. 2011;30:273---8.
10.Karlowicz MG, Buescher ES, Surka AE. Fulminant late-onset sepsisin aneonatalintensive careunit,1988---1997,andthe impact of avoiding empiric vancomycin therapy. Pediatrics. 2000;106:1387---90.
11.Ericson JE, Thaden J, Cross HR, Clark RH, Fowler VG Jr, Benjamin DK Jr, et al. No survival benefit with empiri-calvancomycintherapyforcoagulase-negativestaphylococcal
bloodstreaminfectionsininfants.PediatrInfectDisJ.2015;34: 371---5.
12.Marchant EA, Boyce GK, SadaranganiM, LavoiePM. Neona-talsepsis due to coagulase-negativestaphylococci. ClinDev Immunol.2013;2013:586076.
13.HemelsMA,vandenHoogenA,Verboon-MaciolekMA,FleerA, KredietTG.Aseven-yearsurveyofmanagementof coagulase-negativestaphylococcalsepsis intheneonatalintensivecare unit:vancomycin may not be necessary as empiric therapy. Neonatology.2011;100:180---5.
14.Brasil.MinistériodaSaúde.Portaria2616/98,de12demaiode 1998.Regulamentaasac¸õesdecontroledeinfecc¸ãohospitalar nopaís.Brasília:MinistériodaSaúde;1998.
15.Brasil. Ministério da saúde. Agência Nacional de Vigilância Sanitária. Critérios diagnósticos de infecc¸ão relacionados à assistênciaasaúde---Neonatologia.Brasília:MinistériodaSaúde; 2013.
16.CenterforDiseasesControlandPrevention.CDC/NHSN surveil-lance definitions for specific types of infections. [cited 08 Dec 2015]. Available from: http://www.cdc.gov/nhsn/PDFs/ pscManual/17pscNosInfDefcurrent.pdf.
17.Romanelli RM, Anchieta LM, Mourão MV, Campos FA, Loy-ola FC, Mourão PH, et al. Risk factors and lethality of laboratory-confirmed bloodstream infection caused by non-skin contaminant pathogens in neonates. J Pediatr (Rio J). 2013;89:189---96.
18.Jean-BaptisteN,BenjaminDKJr,Cohen-WolkowiezM,Fowler VGJr,LaughonM,ClarkRH,etal.Coagulase-negative staphy-lococcalinfectionsintheneonatalintensivecareunit.Infect ControlHospEpidemiol.2011;32:679---86.
19.BentlinMR,RugoloLM,FerrariLS,BrazilianNeonatalResearch Network (Rede Brasileira de Pesquisas Neonatais). Practices relatedtolate-onsetsepsisinverylow-birthweightpreterm infants.JPediatr(RioJ).2015;91:168---74.
20.KaufmanDA.Less is more:decreasing antibiotic daysinthe NICU.JPediatr(RioJ).2013;89:424---5.
21.CottenCM,TaylorS,StollB,GoldbergRN,HansenNI,Sánchez PJ, et al. Prolonged duration of initial empirical antibiotic treatment is associated with increased rates of necrotizing enterocolitisanddeathforextremelylowbirthweightinfants. Pediatrics.2009;123:58---66.
22.LiuC,BayerA,CosgroveSE,DaumRS,FridkinSK,GorwitzRJ, et al. Clinicalpractice guidelines bythe InfectiousDiseases SocietyofAmericaforthetreatmentofmethicillin-resistant
Staphylococcusaureusinfectionsinadultsand children.Clin InfectDis.2011;52:e18---55.
23.CenterforDiseasesControlandPrevention(CDC). Recommen-dations for preventing the spreadof vancomycin resistance recommendations ofthe HospitalInfectionControl Practices Advisory Committee (HICPAC). [cited 02 Sep 2015]. Avail-able from: http://www.cdc.gov/mmwr/preview/mmwrhtml/ 00039349.htm.