Protective Role of the Interleukin 33 rs3939286 Gene Polymorphism in the Development of Subclinical Atherosclerosis in Rheumatoid Arthritis Patients.
Texto
Imagem
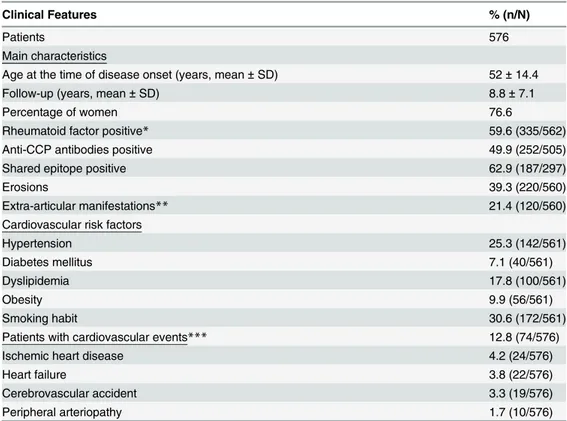

Documentos relacionados
Além das implicações éticas para os seres humanos, como os riscos à saúde de trabalhadores, salários baixos, precarização e até mesmo a existên- cia de
This review provides examples of the fact that different procedures for the measurement of atherosclerosis in mice may lead to interpretation of the extent of atherosclerosis
Therefore, the aim of the present study was to evaluate the risk factors associated with the presence of subclinical atherosclerosis, assessed by CAC, in a sample of
Resumo: Diante da presente atuação na proteção de direitos humanos, tanto por parte dos Sistemas Internacionais de Direitos Humanos quanto das Comunidades Econômicas Regionais,
O presente trabalho tem como principal objetivo investigar a participação das pessoas nos programas organizados e geridos pela Fundação INATEL, mais
No terceiro capítulo são apresentados dois estudos: o primeiro referente à influência da capacidade funcional e das variáveis demográficas no DCL e nas ABVD em idosos com DCL e
In this study, we found a signi fi cant increase in plasma uric acid (P=0.002) and allantoin (P=0.025) in participants of the Brazilian Longitudinal Study of Adult Health
Objectives: To identify the frequency of subclinical atherosclerosis in HIV-infected patients compared to control subjects; to analyze associations between atherosclerosis