ContentslistsavailableatSciVerseScienceDirect
Industrial
Crops
and
Products
j o u r n al hom ep a g e :w w w . e l s e v i e r . c o m / l o c a t e / i n d c r o p
Fractioning
and
chemical
characterization
of
barks
of
Betula
pendula
and
Eucalyptus
globulus
Isabel
Miranda,
Jorge
Gominho
∗,
Inês
Mirra,
Helena
Pereira
CentrodeEstudosFlorestais,InstitutoSuperiordeAgronomia,UniversidadeTécnicadeLisboa,TapadadaAjuda,1349-017Lisboa,Portugal
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received17February2012 Receivedinrevisedform3April2012 Accepted14April2012
Keywords: BetulapendulaRoth. EucalyptusglobulusLabill. Bark
Particlesize Chemicalcomposition
a
b
s
t
r
a
c
t
Thecompositionofbirch(BetulapendulaRoth.)andeucalypt(EucalyptusglobulusLabill.)barkswas studiedaftergrindingandfractioningintodifferentparticlessizes.
Therewasasignificantdifferenceinthefractionationofbothbarksinrelationtotheyieldoffines(5.9% and28.3%ofparticlesunder0.450forbirchandeucalypt,respectively)andofcoarserparticlesover2mm (70.7%and41.4%).
Thechemicalcompositionofbirchandeucalyptbarks,asamassweighedaverageofallgranulometric fractionswas,respectively:ash2.9%and12.1%;totalextractives17.6%and6.5%(hydrophilicextractives weredominant),lignin27.9%and28.8%andholocellulose49.8%and62.6%.Birchbarkcontaineda con-siderableamountofsuberin(5.9%)whereaseucalyptbarkcontainedaverysmallamount(<1%).The carbohydratecompositiondifferedbetweenbirchandeucalyptbarks,i.e.,respectively,glucose47.0% and68.4%,andxylose33.8%and23.2%oftotalneutralmonosaccharides.
Ashelementalcompositionwasdifferentinbothspecies.Birchbarkcontainedinrelationtoeucalypt bark,inthe0.250–0.450mmfraction,moreN(0.69%vs.0.26%)andP(0.075%vs.0.001%),andlessCa (0.39%vs.0.62%),K(0.24%vs.0.31%)andMg(0.07%vs.0.15%).HighconcentrationofZnwasfoundin birchbark(217mg/kgvs.11mg/kgineucalyptbark).
Aftergrindingandgranulometricseparation,extractiveswerepresentpreferentiallyinthefinest frac-tionwithanenrichmentindichloromethaneandethanolsolublesespeciallyinthecaseofbirchbark. Eucalyptbarkhadahighcontentofcelluloseandhemicellulosesespeciallyinthecoarserfraction.The fibrouscharacterofthisfractionshowsitspotentialasafibersource.
© 2012 Elsevier B.V. All rights reserved.
1. Introduction
Barkrepresentsasubstantialproportion oftheaboveground
totalbiomassoftrees.Duringindustrialprocessingfortimberorfor
pulping,barkisremovedfromthelogsandconstitutesanimportant
millresidualmaterialthatisusuallyburntforenergyproduction.
Inaddition tobeingconsidereda valuablesolid biofuel,barkis
alsoscrutinizedformoreadded-valueproductsthatwillconsider
potentialspecificchemicalcompositionorproperties(Demirbas,
2010).
Barkvalorization,namelyifenvisagedwithinabiorefinery
plat-form,thereforerequiresacarefulexaminationofcompositionand
processing characteristics.Barks areusually rich in extractives,
includingorganicsolventandwatersolubles,andin
polypheno-lics,and theyalsocontaina highamountof inorganicmaterial
(FengelandWegener,1984;Pereiraetal.,2003).Structurallybarks
∗ Correspondingauthor.
E-mailaddress:[email protected](J.Gominho).
arecomplextissuesandtheirsampling,characterizationand
pro-cessinghavedifficultiesthatarenotfound,e.g.inwoodprocessing.
Hardwoodspeciesarepresentlythemostimportantsourceof
woodforpulpproduction(Pattetal.,2006).Whitebirch(Betula
pendula) is thedominant pulpwood species in Northern
Euro-peancountries(especially inFinlandand Russia)and eucalypts
(mainlyEucalyptusglobulus)aredominantinPortugalandSpain,
in Southern Europe. The pulpwood consumption in Europe in
2010ofbirchandeucalyptwas,respectively,18425millionm3and
13708millionsm3,correspondingto12.5%and9.3%ofthetotal
pulpwoodconsumption(CEPI,2010).
Inbothcasesconsiderableamountsofbarkaremadeavailable
fromlogdebarking, and areseparated inthemillas aresidual
productandusedasfuel.
InE.globulustreesattheageusedforpulping(9–13yearsin
temperateclimates)barkhasathicknessintherangeof3–16mm
andcorrespondsto7–20%ofo.d.massofthestemdependingon
siteandgenetics(ascompiledinPereiraetal.,2010).
TherearefewstudiesonB.pendulabarkcontent.Jensen(1948)
reported3.4%outerbarkinbirchlogs.Bhat(1982)referredthatbark
thicknessisstronglyassociatedwithstemdiameterandreported
0926-6690/$–seefrontmatter © 2012 Elsevier B.V. All rights reserved. http://dx.doi.org/10.1016/j.indcrop.2012.04.024
ameanvalueof16.6mmforthedoublebarkthicknessoftrees
aged65–95years.Repola(2008)reportedadoublebarkthickness
atbreastheightbetween2.5and38.7cmintreeswithinanage
rangeof7and132years.Foryoungtreeswith1–16yearsofage
and0.3–24.0cmstemdiameter,Trockenbrodt(1991)reportedbark
thicknessvaluesbetween7.1and26.1mm.
Somestudieshave characterizedeucalypt barkanatomically
(Quilhóetal.,1999,2000)andchemically(Sakai,2001;Bargatto,
2010)aswellasbirchbark(Bhat,1982;Trockenbrodt,1991;Harkin
and Rowe,1971). Howeverlittle is known ontheirfractioning
behaviorandonthechemicalcharacteristicsofdifferentfractions.
Theuseoffractioningisusuallyinvolved inbiomassprocessing
andmaybeusedforselectiveenrichmentofspecificcomponents
bytakingadvantageofthebiomasschemicalandstructural
het-erogeneity(Mirandaetal.,2012;Silvaetal.,2011).However,this
isspeciesspecificanddependsonthespecificbarkcharacteristics,
asrecentlyshownforPiceaabies,PinussylvestrisandQuercuscerris
(Mirandaetal.,2012;Senetal.,2010).
Thispaperstudiesthechemical composition ofthebarks of
thesetwo importantEuropeanhardwoods, silver birch(B.
pen-dula)andTasmanianbluegum(E.globulus),obtainedasresidual
by-productsofcommercialdebarkinginpulpmills,after
fractiona-tionintodifferentparticlesizes.Summativechemicalanalysisand
inorganiccompositionwere determinedfor eachgranulometric
fraction,aswellasbulk density.Theobjectiveistoanalyzethe
potentialoftrituratingandparticlefractioningasabiomass
pre-treatmentstepforaselectivecomponentenrichmentwithinabark
valorizationchainforenergy,compositematerialsandchemicals.
2. Materialsandmethods
2.1. Sampling
Barksfrombirch(B.pendula Roth)and eucalypt(E. globulus
Labill.)wereobtainedbyindustrialstemdebarkinginpulpmills,
andwereprovidedbySödra(Sweden)andCelbi(Portugal),
respec-tively.Thebulkbarksampleswereair-driedatambientconditions
andanyvisiblewoodchipswereremoved.
2.2. Fractioning
Thebarkswerefractionatedusingaknifemill(RetschSM2000)
withan outputsieve of 10mm×10mm and screened using a
vibratorysievingapparatus(RetschAS200basic).Thefollowing
sievemesh sizeswereused: 80(0.180mm),60(0.250mm),40
(0.450mm),20(0.850m),15(1.0mm)and10(2.0mm).Themass
ofthefractionretainedoneachsievewasweighedandthe
corre-spondingsevenmassfractionsyieldsweredetermined.
2.3. Bulkdensity
Thebulk densityof thegranulatedsampleswasdetermined
for each sieve fraction using a cylindrical container (29.8mm
height×28.1mmdiameter)astheratioofthemasssampleinthe
containertothevolumeofthecontainer.Thedeterminationwas
madeintriplicatesamples.
2.4. Microscopicobservations
Thedifferentgranulometricfractionsofthebarksampleswere
observedmicroscopicallyaftercelldissociationbymacerationin
a1:1glacialaceticacid:hydrogenperoxidesolution,andstaining
withastrablue.
2.5. Chemicalcharacterization
Chemicalsummativeanalysisincludeddeterminationof ash,
extractives soluble in dichloromethane, methanol, ethanol and
water,suberin,Klasonandacidsolublelignin,andholocellulose,
aswellasthemonomericcompositionofpolysaccharides.
Thegranulometricfractions withparticlesizeover 40 mesh
werecarefullygroundpriortochemicalanalysisinordertoobtain
particlesthatpassedthroughthe40meshsieve.
Theunextractedbarksampleswereusedtodetermine1%NaOH
solubles.
AshcontentwasdeterminedaccordingtoTAPPIStandardT15
os-58using2.0gsamplesastheresidueofovernightincineration
at450–500◦C.
The alkaline lixiviation with1% NaOH was carried out in a
stirredglassreactorwithrefluxusing1.0gofmaterialwitha1:50
solid:liquidratio,at100◦Cduring1h.
SolventextractionwasperformedinaSoxtecextractor
succes-sivelywithdichloromethane,methanol,ethanolandwaterduring
1.5hwitheachsolvent,andtheextractivessolubilizedbyeach
sol-ventweredeterminedusingthemassdifferencefromthemassof
thesolidresidueafterdryingat105◦Candreportedasapercentage
oftheoriginalsamples.
Suberin content was determined on 1.5g of extractive-free
material by refluxing with100ml of a 3% NaOCH3 solution in
CH3OH during 3h (Pereira, 1988a). The sample was filtrated,
washed withmethanol, again refluxed with100ml CH3OH for
15minandfiltrated.ThecombinedfiltrateswereacidifiedtopH
6with2MH2SO4andevaporatedtodryness.Theresiduewas
sus-pendedin50mlwaterandthealcoholysisproductsrecoveredwith
dichloromethaneinthreesuccessiveextractions,eachwith50ml
dichloromethane.Thecombinedextractsweredriedover
anhy-drousNa2SO4andevaporatedtodryness.Thesuberinextracts,that
includethefattyacidandfattyalcoholmonomersofsuberin,were
quantifiedgravimetrically,andtheresultsexpressedinpercentof
theinitialdrymass.
Klason and acid-soluble lignin, and carbohydrates contents
weredeterminedon0.35gofextractedanddesuberinized
sam-ples.Sulfuricacid(72%,3.0ml)wasaddedtothesampleandthe
mixtureplacedinawaterbathat30◦Cfor1hafterwhichthe
sam-plewasdilutedtoaconcentrationof3%H2SO4andhydrolyzedfor
1hat120◦C.Thesamplewasvacuum-filteredthroughacrucible
andwashedwithboilingpurifiedwater.Klasonligninwas
deter-minedasthemassofthesolidresidueafterdryingat105◦Cand
acid-solubleligninwasdeterminedonthefiltratebymeasuring
theabsorbanceat206nmusingaUV/VISspectrophotometer.
Kla-sonligninandacid-solubleligninwerereportedaspercentageof
theoriginalsampleandcombinedtogivethetotallignincontent.
Thepolysaccharideswerecalculatedbasedontheamountofthe
neutralsugarmonomersreleasedbytotalhydrolysis,after
deriva-tizationasalditolacetatesandseparationbygaschromatography
withamethodadaptedfromTappi249cm-00.Thehydrolyzed
car-bohydrateswerederivatizedasalditolacetatesandseparatedbyGC
(HP5890Agaschromatograph)equippedwithaFIDdetector,using
heliumascarriergas(1ml/min)andafusedsilicacapillarycolumn
S2330(30m×0.32mmi.d.×0.20mfilmthickness).Thecolumn
programtemperaturewas225–250◦C,with5◦C/minheating
gra-dient,andthetemperatureofinjectoranddetectorwas250◦C.For
quantitativeanalysistheGCwascalibratedwithpurereference
compoundsandinositolwasusedasaninternalstandardineach
run.
Theholocellulosecontentwasdeterminedonthe
extractive-freesamplesbythechloritemethod(Rowell,2005):1gofsample
wasplacedinanErlenmeyerflask(300ml)and32mlofdistilled
water wasadded.While slowlyshaking, 0.750gof NaClO2 and
Table1
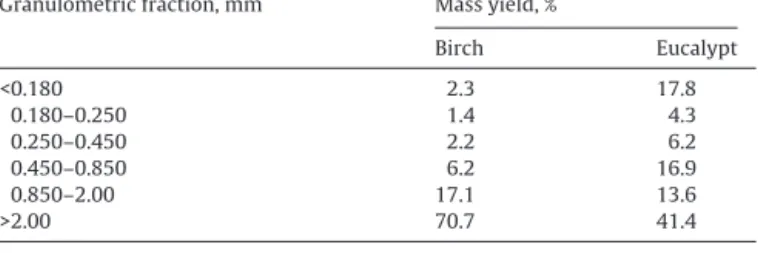
Massyields(%oftotaldrymass)ofthedifferentgranulometricfractionsaftermilling ofbarks.
Granulometricfraction,mm Massyield,%
Birch Eucalypt <0.180 2.3 17.8 0.180–0.250 1.4 4.3 0.250–0.450 2.2 6.2 0.450–0.850 6.2 16.9 0.850–2.00 17.1 13.6 >2.00 70.7 41.4
glassandboiledat70–80◦Cfor60min.Again,0.750gofNaClO2
and0.3mlofaceticacidwereaddedandboiled3times.After
cool-ing,thesamplewasfilteredusingafilterflaskandwashedwith
coldwaterandacetone50%untilfreeofacid.Theinsolubleportion
wasdriedat105◦Cfor4h,cooledinadesiccatorandweigheduntil
constantweight.Theashcontentoftheobtainedholocellulosewas
determinedbyincinerationandtheash-freeholocellulosecontent
wascalculatedaspercentofdrymassoftheinitialsample.
2.6. Ashcomposition
Nitrogen wasdetermined by the Kjeldahl method (Jackson,
1958)inaTecatorequipment(Herdon,VA,USA).Aftera
hydrochlo-ricdigestionoftheash(MartiandMu ˜noz,1957),phosphoruswas
determined by molecularabsorption spectrometryin a Hitachi
U-2000VIS/UVequipment,andalltheothermineralswere
deter-minedbyatomicabsorptionspectrophotometerinaPyeUnicam
SP-9apparatus(Cambridge,UK)equippedwithagraphitefurnace
GF95.
3. Resultsanddiscussion
3.1. Barkfractioning
Thebarksofbirch(B.pendulaRoth.)andeucalypt(E.globulus
Labill.)weremilledandthemassyieldsobtainedforthedifferent
granulometricfractionsaresummarizedinTable1.
Therewasasignificantdifferenceinthefractionationofboth
barks.Forbirchbarktheyieldoffineswaslow,i.e.only5.9%were
particlesunder0.450mm,andthemajorfractioncorrespondedto
thelargestparticles,i.e.70.7%ofparticlesover2mm.Thisgrinding
behaviorwithlittleformationoffineswasalsofoundforconifer
barksofPinuspinaster(Vázquezetal.,1987), P.sylvestrisand P.
abies(Mirandaetal.,2012).
Onthecontrary,inthecaseofeucalyptbarkasignificantamount
offineswasobtainedwith17.8%ofparticlesunder0.180mmand
thefractionswiththelargerparticlesshowedcomparativelylower
yields(e.g.41%forparticlesover2mm).
Theseresultsshowthatthemillingprocessappliesdifferently
todifferentbarksand thebarkstructuralfeaturesinfluencethe
grindingbehavior.Barksaremadeupofdifferenttissues–phloem,
peridermandrhytidome–invaryingextentandcellular
charac-teristics.
Inthecaseofeucalyptbark,thereisnorhytidome,sinceolder
peridermsaresheddedoutbythetreeandonlyathinperiderm
coversthephloem(Quilhóetal.,2000).Thephloemisrather
uni-formandischaracterizedbytangentiallayersofaxialparenchyma
cellsthatareinterspersedbyregionsoffibers,representing,
respec-tively,50%and28%ofthetotalcross-sectionalarea(Quilhóetal.,
1999,2000).Itis therefore understandable thatfracture occurs
preferentiallybythefragileandthinwalledparenchymaregions,
leaving thefibrous bundles in the coarserfractions. This could
betrackedmacroscopicallysincethe>2mmfractionhadaclear
Fig.1. Eucalypt(EucalyptusglobulusLabill.)bark.(a)Granulometricfractionwith >2mmparticles,observedunderthemagnifyingglass;(b)microscopicobservations ofdissociatedcellsobtainedfromthe1to2mmgranulometricfraction.
fibrousaspect(Fig.1a)whichwasfurtherobservedmicroscopically
byanalyzingthecelltypesafterdissociation(Fig.1b).
Birchbarkis markedlydifferentin structure. Itis composed
of twodistinct zones,thephloemandanimportant outerbark
(rhytidome)consistingofnumeroustightlypackedperidermsthat
includephellemlayersofthickandthin-walledcorkcells(Bhat,
1982).Thephloemhasagreatproportionofscelenchymaandthe
proportionofsclereidspredominatesoverthatoffibers.In
conse-quencethecoarserfractionofbirchdifferedmacroscopicallyfrom
thatofeucalyptbarkshowinganaspectofirregularlymoreorless
cubicparticlesofphloemandofflakesshapedparticlesfromthe
rhytidome(Fig.2a).Microscopically,aggregatesofsclerifiedcells
andofthephellemtissuecouldbeobserved(Fig.2b).
3.2. Bulkdensity
Table2givesthebulkdensityofthedifferentbark
granulomet-ricfractions.Bothbarksdifferedsignificantlyindensity,withmean
valuesof277kg/m3and169kg/m3forbirchandeucalypt,
respec-tively.Inthecaseofbirchbarkthereisacleartrendofincreasing
bulkdensitywiththedecreaseofparticledimensioninagreement
withtheoverallideathatsmallparticlesmaybebettercompacted
thanlargerparticles.
The eucalypt barkgranulates did not showa cleartrend of
Fig.2. Birch(BetulapendulaRoth.)bark.(a)Granulometricfractionwith>2mm particles,observedunderthemagnifyingglass;(b)microscopicobservationsof dissociatedcellsobtainedfromthe0.180to0.250mmgranulometricfraction.
betweenfractionsweresmaller.Theelongatedformoftheeucalypt
barkfractionsdoesnotallowaneasycompactionofparticles.
Verylittleinformation existsonbarkbulkdensity. Similarly
tothedifferencefoundhereforeucalyptandbirchbarks,abulk
densityof250kg/m3wasreportedfor0.120–0.50mmsizedbark
particlesofE.globulus(SarinandPant,2006).
Itisobviousthatbulkdensitiesofgranulatedbarksaremuch
lowerthanthecorrespondingparticledensity.Birchandeucalypt
barkshavebasicdensitiesranging,respectively,from452kg/m3to
559kg/m3(Bhat,1982;LambandMarden,1968),andfrom374to
454kg/m3(QuilhóandPereira,2001).Thisdifferencebetweenthe
Table2
Bulkdensity(kg/m3)forthedifferentgranulometricfractionsofthebirchand euca-lyptbarks.
Granulometricfraction,mm Bulkdensity,kg/m3
Birch Eucalypt <0.180 311.3 160.8 0.180–0.250 305.1 155.5 0.250–0.450 300.7 189.9 0.450–0.850 271.0 192.1 0.850–2.00 255.6 153.2 >2.00 239.0 181.2 Mean 276.9±28.9 169.4±18.5 Table3
Ashcontent(%oftotaldrymass)ofthedifferentgranulometricfractionsaftermilling ofbarks.
Granulometricfraction,mm Ash,%oftotaldrymass
Birch Eucalypt <0.180 3.4 23.1 0.180–0.250 4.5 14.7 0.250–0.450 2.5 15.9 0.450–0.850 2.6 7.9 0.850–2.00 2.1 6.7 >2.00 2.2 4.3 Mean 2.9±0.9 12.1±7.1
soliddensityofbothbarksisinaccordancewiththeir
correspond-ingstructuralfeatures,asdiscussedpreviously.
3.3. Ashcontentandcomposition
Theashcontentof thebirchand eucalyptbarksamples
col-lectedatthemillaftermillingand separationintothedifferent
granulometricfractionsisreportedinTable3.Thewholebiomass
ofbirchandeucalyptbarkshadanashcontentof2.9%and12.1%,
respectively,determinedasamassweighedaverageofallfractions.
ForB.pendulabark,Werkelinetal.(2005)reportedasimilarash
contentof3.8%andSaarelaetal.(2005)reportedvaluesbetween
1.0%and1.9%.Ashcontentforotherbirchspecieswasreportedas
1.7%forBetulaalleghaniensisand1.8%forBetulapapyrifera(Corder,
1976).
Thebirchbarkfractionsshowedhigherashcontentinthefiner
particles (4.5%in the 0.180–0.250mm fractionand 2.2%in the
>2mmfraction).Itisknownthatashtendstoaccumulateinthe
finersizedfractionduringbiomassprocessingduetothesmallsize
andbrittlenessofinorganicmaterial(Bridgemanetal.,2007;Liu
andBi,2011).Howevertheextentofmineralaccumulationinbark
fractionsdependsonthespecies(Mirandaetal.,2012).
TheashcontentofE.globulusbarkwasreportedas4.7%(Vázquez
etal.,2008)and4.5%(Akyuzetal.,2003)andintherangeof1.6–3.5% (Pereira, 1988b).Thepresent eucalyptbarksamplehada much
highermineralcontentwhichshowsthatthebarkobtainedinthe
millcontainedconsiderablecontaminationwithsoilparticlesand
otherextraneousfinematerial.Theamountofincrustatedsand
granuleswasnotoriousbyadirectmacroscopicobservationofthe
sample.Duetotheirsmallsize,theseparticleswereretainedin
thefinergranulometricfractionsafterthemillingandscreening.
AsshownbyTable3,thisoccurredespeciallyinthefinestparticles
(e.g.23.1%ashcontentinparticlessizebelow0.180mm)whilethe
largerdimensionparticlesshowedanashcontentsimilartothe
valuesreportedintheliteratureforeucalyptbark(4.3%in>2mm
particles).
Asimilaroccurrenceofsubstantialaccumulationofminerals
wasalsofoundforpinebarkobtainedatthemill(Mirandaetal.,
2012).Thisclearlyshowstheimportanceofbarkcleaningduring
fieldandmillhandlinginordertoavoidunsuitablecontaminations
duringsubsequentbarkvalorizationsteps.
The occasional problem of excessive sand and soil in the
eucalypt stem raw-material for thepulping industry is already
identified,dependingonthecaretakenbycontractorsduringfield
treeharvestingandhandlingsincethesoftandmoisteucalyptbark
allowseasyincrustationofthemineralparticles.
Twoofthebarkgranulometricfractions(0.250–0.450mmand
<0.180mm)werecharacterizedinrelationtomineralcomposition,
asshowninTable4.
Theconcentrationsofthemajorelementsweredifferentinboth
species.Birchbarkcontainedinrelationtoeucalyptbarkinthe
Table4
Elementalconcentrationsinbarksamplesofbirchandeucalyptdeterminedinthe 0.250–0.450mmand<0.180mmgranulometricfractions.
Birch Eucalypt 0.250–0.450mm <0.180mm 0.250–0.450mm <0.180mm N(%) 0.69 0.50 0.26 0.19 Ca(%) 0.393 0.303 0.623 0.181 Mg(%) 0.072 0.049 0.154 0.120 Na(%) 0.013 0.011 0.070 0.080 K(%) 0.242 0.169 0.308 0.230 P(%) 0.075 0.022 0.001 0.001 Cu(mg/1000g) 3.37 1.86 4.70 4.08 Zn(mg/1000g) 216.77 152.58 11.39 6.45 Ni(mg/1000g) 1.77 1.65 5.37 3.68 Cr(mg/1000g) 2.18 1.72 9.38 3.01 Pb(mg/1000g) 2.15 1.43 2.02 1.47
vs.0.001%),butlessCa(0.39%vs.0.62%),K(0.24%vs.0.31%)andMg
(0.07%vs.0.15%).
HighconcentrationofZnwasfoundintheashesofbirchbark
with217mg/kgincomparisonwith11mg/kgineucalyptbark.
Therewereonlysmalldifferencesinthemineralcomposition
ofboth fractions (Table 4)withtheexceptionof Ca
concentra-tionwhichdecreasedsubstantiallyinthefinestfractionofeucalypt
bark.
Thevaluesareintherangeofthosereportedforbirchbarks
byWerkelinetal.(2005),Saarelaetal.(2005)andReimannetal. (2007)andforeucalyptbarksbyPereira(1988b)andDamindaSilva etal.(1983).
3.4. Alkalineextraction
Alkalineextractionwith1%NaOH solubilized51.7% of birch
barkand43.3% ofeucalyptbark.Theseresultsarein therange
foundrecentlyforthealkalineextractionofspruceandpinebarks
(Mirandaetal.,2012)butarehigherthanthe1%NaOHextractives
reportedforE.globulusbarkbyVázquezetal.(2008)andbyPereira
(1988b),respectively,at26.6%and20.4–30.6%.HarkinandRowe (1971)alsoreportedforB.alleghaniensisand B.papyrifera28.4%
and25.1%,respectively.
Thehigheramountsofsubstancesremovedbyalkaline
lixivi-ationofbirchbarkincomparisontoeucalyptbarkareinrelation
withthepresenceofhigheramountsofextractivesandsuberinas
wellasofhemicelluloses.Suchcompoundsarepresentinhigher
contentsinthebirchbark,asdiscussedbelow.
3.5. Summativechemicalcomposition
Thebirchandeucalyptbarkswerechemicallycharacterizedand
thecompositioncalculatedasamassweighedaverageofall
gran-ulometricfractionsisshowninTable5.
Thereareconsiderabledifferencesbetweenbirchandeucalypt
barksinrelationtoextractives,suberinandholocellulosecontents.
Eucalyptbarkcontainslessextractivesthanbirchbarkbutmore
celluloseandhemicellulosesexpressedasholocellulose.
As regards the total extractives content, birch bark has
approximatelythree timesmore extractivesthaneucalypt bark
(respectively, 17.6% and 6.5%). Polar compounds extracted by
ethanol and water, which include especially phenolics and
polyphenolics,correspondedtoasignificantproportionofthetotal
content(60.8%inbirchand83.0%ineucalyptofthetotal
extrac-tives).Waxes and other non-polar compoundsthat are soluble
indichloromethane andmethanol makeuptheremaining bark
extractives,representing31.8%ofthetotalextractivesinbirchbark,
and16.9%ineucalyptbark.
Table5
Summativechemicalcomposition(%o.d.material)ofthebirchandeucalyptbarks, calculatedasmassweighedaverageofallgranulometricfractions.
Birch Eucalypt Ash 2.9 12.1 Extractives 17.6 6.5 Dichloromethane 5.1 0.9 Methanol 0.5 0.2 Ethanol 5.5 1.3 Water 5.2 4.1 Suberin 5.9 0.98a Lignin 27.9 34.1 Klason 26.4 26.6 Acidsoluble 1.5 7.5 Holocellulose 49.8 62.6
aSuberincontentdeterminedin40–60meshfraction.
Appreciabledifferencesarealsofoundinthecontentofsuberin.
Thebirchbarksamplecontainedaconsiderableamountofsuberin
(5.9%)whereastheeucalyptbarkcontainedaverysmallamount
(less than1%). Thisis in direct relationwiththedifferences in
theanatomicalstructureofbothbarks,i.e.withtheabsenceofa
rhytidomewithsuberizedphellemtissuesineucalyptbark.
Thebirchandeucalyptbarksamplesshowedsimilarcellwall
lignificationbuttherewasastrikingchemicaldifferencebetween
bothbarksinrelationtoholocellulosecontent:eucalyptbarkhad
muchhigherholocellulose(62.6%vs.49.8%).
Therearefewpublishedreferencesonthechemical
composi-tionofhardwoodbarks.ForthebirchbarksofB.alleghaniensisand
B.papyrifera,HarkinandRowe(1971)reported,respectively,total
extractives17.4%and22.4%,andlignin40.6%and37.8%.
ForE.globulusbark,Yadavetal.(2002)reported7.2%alcohol
extractives,15.5%waterextractives,28.0%ligninand62.2%total
carbohydrates.Vázquez etal. (2008) reported19.2% lignin and
Sakai (2001)18.6% lignin,43.2% cellulose and19.6% pentosans.
Pereira(1988b)referred6.3–8.5%extractives,16.7–21.1%Klason
lignin,48.2–54.9%celluloseand7.0–19.6%pentosans.
ThecarbohydratecompositionisgivenbyTable6inrelationto
theproportionofneutralmonosaccharides.Therearealso
consid-erabledifferencesbetweenbirchandeucalyptbarksespeciallyin
relationtoglucoseandxylosecontents.Glucoserepresented68.4%
and47.0%oftotalneutralmonosaccharides,respectively,in
euca-lyptandbirchbarksandxylose23.2%and33.8%,respectively.This
isclearlyindicativeofadifferentproportionofcelluloseand
hemi-cellulosesineucalyptandbirchbark.Takingroughlytheratioof
glucosetoxyloseasindicativeofcellulosetohemicelluloses,there
isastrikingdifference:2.96ineucalyptbarkand1.39inbirchbark.
Thechemicalcompositionofthexylansalsodiffersbetweenthe
species:theratioofarabinosetoxylosewas0.12ineucalyptbark
and0.31inbirchbark.
Vázquez et al. (2008)reported similarmonomeric
composi-tion ofpolysaccharides for E.globulusbark: a predominanceof
glucosewith70.4%andanimportantcontentofxyloseof20.8%
with3.6% ofgalactose,3.7% of arabinoseand 1.4% ofmannose.
Bargatto(2010)reportedalsothecarbohydratecompositionofE.
grandis×urophyllaandE.grandisbarks,respectively,forglucose
Table6
Carbohydratecompositionofthedifferentbarksin%oftotalneutral monosaccha-rides(40–60meshgranulometricfraction).
Monosaccharide Birch Eucalypt
Glucose 47.0 68.4 Mannose 4.1 1.9 Galactose 3.8 3.3 Rhamnose 1.1 0.4 Xylose 33.8 23.2 Arabinose 10.3 2.7
Table7
Chemicalcomposition(%oftotaldrymass)ofthedifferentgranulometricfractions aftermillingofbirchandeucalyptbarks.
Birchbark Eucalyptbark
F M C F M C Extractives 24.0 15.6 12.0 11.0 6.2 5.5 Dichloromethane 7.5 4.2 3.1 2.6 0.9 0.6 Methanol 0.5 0.5 0.2 0.4 0.3 0.2 Ethanol 8.8 5.1 4.3 1.9 1.2 1.1 Water 6.3 5.7 4.4 6.2 4.0 3.6 Suberin 7.3 6.0 7.2 – – – Lignin 27.1 28.5 27.1 29.0 29.2 22.1 Klason 26.0 27.2 25.1 26.6 26.1 18.9 Acidsoluble 1.1 1.4 2.0 2.4 3.1 3.1 Holocellulose 38.0 56.3 55.1 64.3 53.4 70.0 Granulometricfractions:fine(F,<0.180mm),medium(M,0.250–0.450mm)and coarse(C,>2mm).
76.4%and77.6%,xylose18.9%and16.9%,galactose1.8%and2.3%,
arabinose2.0%and2.2%,ramnose0.6%and0.7%.
HarkinandRowe(1971)reportedfor B.alleghaniensisandB.
papyrifera,respectively:glucose54%and53%,galactose3%and2%,
mannose1%,xylose32%and36%,arabinose8%and6%.
Itisinterestingalsotonoticethatthechemicaldifferencein
polysaccharidecompositionbetweentheB.pendulaandE.globulus
barksalsooccursinthecorrespondingwoods:xyloseandglucose
contents(%oftotal neutralmonosaccharides)represent,
respec-tively,32.6%,and61.4%inB.pendulawoodand20.0%and75.3%in
E.globuluswood(Pintoetal.,2005).
3.6. Effectofparticlesizeonbarkchemicalcomposition
Table7summarizestheresultsobtainedforthechemical
com-position of the fractions of <0.180mm (fine), 0.250–0.450mm
(medium)and >2mm(coarse)of themilledbirchand eucalypt
barks.
Aparticlesizeeffectwasobservedonthecontentand
compo-sitionofextractivesofbirchandeucalyptbark.Extractiveswere
presentpreferentiallyinthefinestfractionduetoenrichmentin
nonpolarandpolarcompounds.Theextractivescontentwas
high-estinthefine fraction, i.e.24.0% incomparison with12.0% for
thecoarserfractioninthecaseofbirchbarkand11.0%vs.5.5%
ineucalyptbark.Asregardscompositionofextractivestherewas
anenrichmentindichloromethaneandethanolsolublesinthefine
fractionespeciallyinthecaseofbirchbark.
Forthestructuralcomponents,theobserveddifferenceswere
eitherofsmallmagnitudeorwithoutaconsistentpatternof
varia-tionforbirchbark.Forinstance,suberincontentwassimilarinthe
threebirchbarkfractions.However,consideration ofthe
chem-icalcomposition basedonextractive-freematerial (ascouldbe
obtainedafterextractionofthebark)pointsoutthatsuberin
con-tentincreasesinthefinerfraction:respectively,9.6%,7.1%and8.1%
ofextractive-freematerialinthefine,mediumandcoarsefractions.
Ineucalyptbarkthelignincontentwashighestinthefine
frac-tion,i.e.29.0%incomparisonwith22.1%forthecoarserfraction
whileholocellulosecontentwashighestinthecoarsefraction.
Thedifferencesinthechemicalcompositionofthebarkfractions
areduetothedifferentanatomicaltissuesthatconditionthe
dis-tributionofsizesaftergrinding(Vázquezetal.,2001).Bridgeman
etal.(2007)reportedthatcellulose,hemicellulosesandlignintend
toremaininthelargerparticlesizedfraction.TamakiandMazza
(2010)andChundawatetal.(2007)alsoreportedcompositional
changeswithparticlesize:withincreasingparticlesizeextractives
contenttendtodecreaseandhemicellulosesandglucancontentto
increasewhilelignincontentdidnotshowcleartrends.
Theresultsshowedthatgrindingandfractionation by
parti-clesizeareunitoperationsthatmaybeusedtoselectiveenrich
fractionsinsolublematerials(thefinestfractions)forbothbarks.
Coarserfractionstendtohaveahigherholocellulosecontentand
willbethereforemoresuitableforcarbohydraterelateduses.
4. Conclusions
Structuralandanatomicalfeaturesareimportantcharacteristics
that influence bark processing, namely their grinding
behav-iorand particle fractioning.Accordingly thebarks of birchand
eucalyptshoweddifferentsizereductionpatternandparticle
char-acteristics.Thereforebiomasspre-treatmentssuchasmillingand
fractioninghavetobeadaptedtothespecificbiomasssource.
Birchandeucalyptbarkssubstantiallydifferedintheirchemical
composition,leadingtodifferentpotentialvalorizationroutes.
Birchbarkhasahighcontentofextractivesandanappreciable
amountofsuberinthatallowconsideringtheiruse.Fractionation
bysizeisadequateforaselectiveenrichmentinthesecomponents
inthefinerfractions.
Eucalyptbarkhasahighcontentofcelluloseandhemicelluloses
thatisenrichedinthecoarserfraction.Thefibrouscharacterofthis
fractionshowsitspotentialasafibersource.
Theresultsalsoshowedtheimportanceofacarefulfield
han-dling of stems in order to avoid excessive contamination with
minerals.
Acknowledgements
ThisworkwassupportedbytheEUresearchproject“AFORE
–Forestbiorefineries:Added-valuefromchemicalsandpolymers
bynewintegratedseparation,fractionationandupgrading
tech-nologies”underthe7thResearchFrameworkProgramme.Centro
deEstudosFlorestaisisaresearchunitsupportedbythenational
fundingofFCT–Fundac¸ãopara aCiência eaTecnologia
(PEst-OE/AGR/UI0239/2011).
References
Akyuz,M.,Sahin,A.,Alma,A.,Bektap,I.,Usta,A.,2003.Conversionoftreebark intobakelite-likethermosettingmaterialsbyphenolation.In:XIIWorldForest Congress,QuébecCity,Canada,September30.
Bargatto,J.,2010.Avaliac¸ãodopotencialdacascadeEucalyptusspp.paraaproduc¸ão debioethanol.Ph.D.Dissertation.UniversidadedeS.Paulo,EscolaSuperiorde AgriculturaLuizdeQueirós,Piracicaba,Brazil.
Bhat,K.M.,1982.Anatomy,basicdensityandshrinkageofbirchbark.IAWABulletin 3(3–4),207–213.
Bridgeman,T.G.,Darvell,L.I.,Jones,J.M.,Williams,P.T.,Fahmi,R.,Bridgwater,A.V., Barraclough,T.,Shield,I.,Yates,N.,Thai,S.C.,Donnison,I.S.,2007.Influenceof particlesizeontheanalyticalandchemicalpropertiesoftwoenergycrops.Fuel 86,60–72.
CEPI,2010.KeyStatistics2010.EuropeanPulpandPaperIndustry.
Chundawat,S.P.S.,Venkatesh,B.,Dale,B.E.,2007.Effectofparticlesizebased sepa-rationofmilledcornstoveronAFEXpretreatmentandenzymaticdigestibility. BiotechnologyandBioengineering96(2),219–231.
Corder,S.E.,1976.Propertiesandusesofbarkasanenergysource.ResearchPaper 31,ForestResearchLaboratory,OregonStateUniversity,Corvallis.
DamindaSilva,H.,Poggiani,F.,Coelho,L.C.,1983.Biomass,nutrientcontentand concentrationinfiveEucalyptusspeciescultivatedonlowsoilfertility.Boletim dePesquisaFlorestal,Colombo6/7,9–25.
Demirbas,A.,2010.BiorefineriesforBiomassUpgradingFacilities(GreenEnergyand Technology).Springer-Verlag,London.
Fengel,D.,Wegener,G.,1984.Wood:Chemistry,Ultraestructure,Reactions.Walter deGruyter,Berlin.
Harkin,J.M.,Rowe,J.W.,1971.Barkanditspossibleuses.ResearchNoteFPL,091.U.S. DepartmentofAgriculture.ForestService.ForestProductsLaboratory,Madison, Wisconsin.
Jackson,M.L.,1958.SoilChemicalAnalysis.Prentice-Hall,Inc.,Madison,Wisconsin. Jensen,W.,1948.Omytskiktsavfalletvidframställningavbörkfaner(Aboutthe sur-facelayerwastefromproductionofbirchveneer).Ph.D.Thesis.ActaAcad. Aboensis,Math.Phys.XVI,3.
Lamb,F.M.,Marden,R.M.,1968.BarkspecificgravitiesofselectedMinnesotatree species.ForestProductsJournal18(9),76–82.
Liu,X.,Bi,X.T.,2011.Removalofinorganicconstituentsfrompinebarksand switch-grass.FuelProcessingTechnology92(7),1273–1279.
Marti,F.,Mu ˜noz,J.,1957.FlamePhotometry.ElsevierPublishing,London. Miranda,I.,Gominho,J.,Mirra,I.,Pereira,H.,2012.Chemicalcharacterizationofbarks
fromPiceaabiesandPinussylvestrisafterfractioningintodifferentparticlesizes. IndustrialCropsandProducts36,395–400.
Patt,R.,Kordsachia,O.,Fehr,J.,2006.EuropeanhardwoodsversusEucalyptusglobulus asarawmaterialforpulping.WaterScienceandTechnology40,39–48. Pereira,H.,1988a.ChemicalcompositionandvariabilityofcorkformQuercussuber
L.WaterScienceandTechnology22,211–218.
Pereira,H.,1988b.Variabilityinthechemicalcompositionofplantationeucalypts (EucalyptusglobulusLabill.).WoodandFiberScience20(1),82–90.
Pereira,H.,Grac¸a,J.,Rodrigues,J.C.,2003.Woodchemistryinrelationtoquality.In: Barnett,J.R.,Jeronimidis,G.(Eds.),WoodQualityanditsBiologicalBasis.CRC Press,BlackwellPublishing,Oxford,pp.53–83.
Pereira,H.,Miranda,I.,Gominho,J.,Tavares,F.,Quilhó,T.,Grac¸a,J.,Rodrigues,J., Shatalov,A.,Knapic,S.,2010.In:CentrodeEstudosFlorestais(Ed.),Qualidade TecnológicadoEucaliptoEucalyptusglobulus.InstitutoSuperiordeAgronomia, UniversidadeTécnicadeLisboa,Lisboa.
Pinto,P.C.,Evtuguin, D.V.,PascoalNeto,C.,2005.Structureof hardwood glu-curonoxylans:modificationsandimpactonpulpretentionduringwoodkraft pulping.CarbohydratePolymers60,489–497.
Quilhó,T.,Pereira,H.,Richter,H.G.,1999.Variabilityofbarkstruturein plantation-grownEucalyptusglobulus.IAWAJournal20(2),171–180.
Quilhó,T.,Pereira,H.,Richter,H.G.,2000.Within-treevariationinphloemcell dimensionsandproportionsinEucalyptusglobulus.IAWAJournal21(1),31–40. Quilhó,T.,Pereira,H.,2001.Withinandbetween-treevariationofbarkcontentand wooddensityofEucalyptusglobulusincommercialplantations.IAWAJournal 22(3),255–265.
Reimann,C.,Arnoldussen,A.,Finne,T.E.,Koller,F.,Nordgulen,Ø.,Englmaier,P., 2007.Elementcontentsinmountainbirchleaves,barkandwoodunderdifferent anthropogenicandgeogenicconditions.AppliedGeochemistry22,1549–1566. Repola,J.,2008.BiomassequationsforbirchinFinland.SilvaFennica42(4),605–624. Rowell,R.M.,2005.HandbookofWoodChemistryandWoodComposites.CRCPress,
Madison,USA.
Saarela,K.-E.,Harju,L.,Lill,J.-O.,Heselius,S.-J.,Rajander,J.,Lindroos,A.,2005. Quan-titativeelementalanalysisofdry-ashedbarkandwoodsamplesofbirch,spruce andpinefromsouth-westernFinlandusingPIXE.ActaAcademiaeAboensis, SeriesB65(4),1–27.
Sakai,K.,2001.Chemistryofbark.In:Hon,D.N.-S.,Shiraishi,N.(Eds.),Woodand CellulosicChemistry.,2nded.MarcelDekker,NewYork.
Sarin,V.,Pant,K.K.,2006.Removalofchromiumfromindustrialwastebyusing eucalyptusbark.BioresourceTechnology97,15–20.
Sen,A.,Miranda,I.,Santos,S.,Grac¸a,J.,Pereira,H.,2010.Thechemicalcomposition ofcorkandphloemintherhytidomeofQuercuscerrisbark.IndustrialCropsand Products31,417–422.
Silva,G.G.D.,Guilbert,S.,Rouau,X.,2011.Successivecentrifugalgrindingandsieving ofwheatstraw.PowderTechnology208,266–270.
Tamaki,Y.,Mazza,G.,2010.Measurementofstructuralcarbohydrates,lignins,and micro-componentsofstrawandshives:effectsofextractives,particlesizeand cropspecies.IndustrialCropsandProducts31,534–541.
Trockenbrodt,M.,1991.Qualitativestructuralchangesduringbarkdevelopmentin Quercusrobur,Ulmusglabra,PopulustremulaandBetulapendula.IAWABulletin 12(1),5–22.
Vázquez,G.,Parajó,J.C.,Antorrena,G.,1987.Sugarsfrompinebarkbyenzymatic hydrolysis.Effectofsodiumchloritetreatments.WaterScienceandTechnology 21,167–178.
Vázquez,G.,González-Alvarez,J.,Freire,S.,López-Suevos,F.,Antorrena,G.,2001. CharacteristicsofPinuspinasterbarkextractsobtainedundervariousextraction conditions.HolzalsRohundWerkstoff59,451–456.
Vázquez,G.,Fontenla,E.,Santos,J.,Freire,M.S.,González-Álvarez,J.,Antorrena,G., 2008.Antioxidantactivityandphenoliccontentofchestnut(Castaneasativa) shellandeucalyptus(Eucalyptusglobulus)barkextracts.IndustrialCropsand Products28,279–285.
Werkelin,J.,Skrifvars,B-J.,Hupa,M.,2005.Ash-formingelementsinfour Scan-dinavianwoodspecies.Part1:Summerharvest.Biomass&Bioenergy 29, 451–466.
Yadav,K.R.,Sharma,R.K.,Kothari,R.M.,2002.Bioconversionofeucalyptusbark wasteintosoilconditioner.BioresourceTechnology81,163–165(Short com-munication).