The
Brazilian
Journal
of
INFECTIOUS
DISEASES
w w w . e l s e v i e r . c o m / l o c a t e / b j i d
Brief
communication
Comparison
among
the
BED
capture
enzyme
immunoassay
test
and
AxSYM
avidity
index
assay
for
determining
recent
HIV
infection
and
incidence
in
two
Voluntary
Counselling
and
Testing
Centres
in
Northeast
Brazil
Daniela
Medeiros
Salustiano
a,b,∗,
Kledoaldo
Oliveira
de
Lima
b,c,
Ana
Maria
Salustiano
Cavalcanti
d,
Ricardo
Sobhie
Diaz
e,
Heloisa
Ramos
Lacerda
baTheHerbertdeSouzaCounsellingandTestingCentre,CabodeSantoAgostinho,PE,Brazil
bPost-graduationinTropicalMedicine,UniversidadeFederaldePernambuco,PE,Brazil
cThePaulistaCounsellingandTestingCentre,Paulista,PE,Brazil
dDepartmentofVirology,LaboratórioCentraldePernambuco(LACEN-PE),Recife,PE,Brazil
eRetrovirologyLaboratory,UniversidadeFederaldeSãoPaulo,SP,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received24October2013
Accepted17March2014
Availableonline26April2014
Keywords: HIV
Recentinfection
Avidityindexassay
BED-CEIA
a
b
s
t
r
a
c
t
TheaimsofthisstudyweretocomparetheautomatedAxSYMavidityassayindexwith
theBEDcaptureenzymeimmunoassaytestandtocalculatetheHIV-1incidenceusingthe
BEDcaptureenzymeimmunoassayandAxSYMavidityassayindexalgorithmswithina
populationseekingtheVoluntaryCounsellingandTestingCentresintwomunicipalities
intheMetropolitanRegionofRecife,NortheastofBrazil.Ananalysiswasconductedin365
samplesthattestedpositiveforHIVinfectionfromfrozenserumcollectedduringtheperiod
2006–2009.Therewasasimilarproportionofmalesandfemales;mostpatientswere
hetero-sexual(86%)withamedianageof29years.Ofthe365samples,102(28%)and66(18.1%)were
identifiedasrecentinfectionsbyBEDcaptureenzymeimmunoassayandAxSYMavidity
assayindex,respectively.TheHIV-1totalincidenceintheBEDcaptureenzyme
immunoas-sayandAxSYMavidityassayindexalgorithmswere:0.79(95%CI:0.60–0.98)and0.34(95%
CI:−0.04to0.72),respectively.Incidencewashigheramongmen.Therewasgood
agree-mentbetweenthetests,withakappaof0.654andaspecificityof95.8%.AxSYMavidity
assayindexmaybehelpfulinimprovingthequalityoftheestimatesofrecentHIVinfection
andincidence,particularlywhenusedinacombinedalgorithmwithBEDcaptureenzyme
immunoassay.
©2014ElsevierEditoraLtda.Allrightsreserved.
∗ Correspondingauthorat:Av.Prof.MoraesRêgo,s/n,BlocoA,TérreodoHospitaldasClínicasdaUniversidadeFederaldePernambuco,
50670-420,CidadeUniversitária,Recife,PE,Brazil.
E-mailaddress:danisalustiano@yahoo.com.br(D.M.Salustiano).
http://dx.doi.org/10.1016/j.bjid.2014.03.001
Brazilhasreported656,701 casesofAIDS, andinrecent
yearstherehasbeenanincreaseintherateofcasesinthe
NorthandNortheastregionsofthecountry,whencompared
totheSouthandSoutheastregions.Furthermore,the
North-easternstateofPernambucoanditscapitalRecifepresentthe
highestratesofnewcasesofAIDSofanystateorstatecapital
inNortheastBrazil.1
ThegrowinginterestinestimatingHIV-1incidencehasled
tothe developmentof a varietyoflaboratory assays. This
methodologyis usedonlyforepidemiologicalinvestigation
purposes,andisreferredtobytherecentlyintroducedterm
RITA (RecentInfectionTestingAlgorithm),used todescribe
laboratoryassaysthatareabletoidentifyatransitionalperiod
ofrecentseroconversionofHIVinfection.2
In 1998, an adaptation of a diagnostic test for HIV-1
that would revolutionize epidemiological methodsthrough
itsabilitytoidentifyrecentHIV-1infectionwasdescribedin
theliterature.3Thetestdifferentiatedrecentinfections(less
thansixmonths)fromestablishedinfections(morethansix
months),duetothekineticsofantibodiesaftertheonsetof
HIV-1infection.ThistestallowedtoestimateHIV-1incidence
fromcross-sectionalstudies,usingasingleserumsampleper
patient.
TheBED capture enzymeimmunoassay(BED-CEIA), the
assayforHIV-1 incidence,haspeptidesasantigensderived
from the immunodominant region of gp41, containing
sequencesofsubtypesB,EandD,whicharerepresentativeof
multiplesubtypesofHIV-1.ThetimeatwhichHIVinfection
occurredcanbeestimated,giventhattheratiobetweenthe
totalIgGandanti-HIVIgGvariesaccordingtotheprogression
ofthedisease.4Thistechniquehasbeenwidelyusedin
con-junctionwithmathematicaltoolsforestimatingtheincidence
ofHIVinfection.5
Another method for distinguishing between recent and
establishedHIVinfectionsbasedonantibodyavidity(affinity)
wasdescribedin2002.6Thismethodologyisbasedonthe
prin-ciplethattheantibodyavidityproducedintheinitialphaseof
theinfection(withinsixmonthsofseroconversion)islow,in
contrasttothatobservedinestablishedinfections(morethan
sixmonths).6Besidesthesimplicityandautomatednatureof
thetechnique,intheory,itwouldalsobeabletodetectahigh
proportionofsubtypesofHIV-1andwouldnotbeaffectedby
thestageoftheinfection.Theassociationofthetwotestshas
beenusedtoconfirmthepositiveresultsofBED,resembling
resultsofincidencedataofcohorts thathavedocumented
seroconversion.7
Inviewoftheimportanceofcomprehendingthedynamics
oftheHIVepidemic,theaimofthisstudyweretocomparethe
magnitudeofagreementbetweentheautomatedtestAxSYM
avidity assay index (Ax-AI) with the BED-CEIA test and to
calculatetheHIV-1incidenceusingBEDandAx-AIwithina
populationseekingVoluntaryCounsellingandTesting
Cen-tres(VCTs)intwomunicipalitiesoftheMetropolitanRegion
ofRecife,inNortheastBrazil.
FrozensamplesfrompatientsdiagnosedwithHIV
infec-tion during the period 2006–2009 were obtained from two
VCTsinthemunicipalitiesofCabodeSantoAgostinhoand
Paulista, in the Metropolitan Region of Recife – Northeast
Brazil.Threehundredandeighteenpatientswerediagnosed
asHIV-infectedattheVCTinCabodeSantoAgostinho,and
aliquotsfrom206(64.8%)ofthemwereavailableforfurther
testing.AtthePaulistaVCT,179casesofHIVinfectionwere
detected,butonly159(94%)hadserumavailablefortesting.
Thusatotalof365serumsampleswereanalyzed.
Samples from patientswith previoususe of
antiretrovi-raltherapy(ART),infectedthroughverticaltransmission,and
thosewithpreviousdiagnosisofHIVinfectionoccurringmore
than sixmonthspriortothecollectionofthesamplewere
excluded.
Datarelatingtotheindividuals’gender,age,and
sociode-mographicdatawereretrievedfromthedatabankoftheHIV
testresultinformationsystem(SIREX-HIV)attheVCTs.This
studywasapprovedbytheResearchEthicsCommitteeofthe
HospitalAgamenonMagalhães(protocolnumber518/2009).
For theAx-AIthesamplewas defrosted,twoaliquotsof
20Lwereobtainedanddiluted1:10.Onealiquot(PBSaliquot)
wasdilutedwith180Lofphosphate-bufferedsaline(PBS)and
theother(Galiquot)wasdilutedwith180Lof1Mguanidine
solution(G)(Sigma–Aldrich).Afterdilution,allsampleswere
vortex-stirredandincubatedfor10minatroomtemperature.
Thetwoaliquotsofeachsampleweretestedusingthe
auto-matedAxSYMHIV-1/2gOassay(AbbottDiagnosticsDivision,
Wiesbaden-Delkenheiem,Germany),inaccordancewiththe
manufacturer’sinstructions.6
Additionofadenaturingagent(guanidine)breaks
hydro-genbridges,whichhelpsdeterminingthesecondarystructure
of the antibodies and leads to a residual effect from the
antigen–antibody interaction. This procedure has greater
effectonrecentantibodies,whicharelow-avidityantibodies.8
ThisprocedureresultsinanS/COratiothatislowerfortheG
aliquotthanforthePBSaliquot.Theavidityindex(AI)is
calcu-latedafterobtainingtheS/COratiosforthePBSandGaliquots
asfollows:6,9
AI=GuanidinealiquotS/CO
PBSaliquotS/CO .
Acutoffvalueof0.8distinguishesaninfectionoflessthan
sixmonths6,10fromalong-terminfection(adurationofmore
thansixmonths).
TheBED-CEIAtest(CalypteBiomedicalCorporation,
Ore-gon, USA) is an IgG-capture ELISA technique based on
detectingincreasingproportionsofspecificanti-HIV-1IgG
fol-lowingseroconversion.Anti-HIVIgGandnon-specificIgGsare
capturedinthesolidphaseofthemicroplate.After
incuba-tionandwashing,peptidesfromtheimmunodominantregion
ofgp41 thatcontainsequences ofthe subtypesB,Eand D
wereadded.Afterfurtherincubationandwashing,a
strepta-vidinconjugatewasadded,followedbyanincubationperiod
of90min.Theresponseisascertainedaftertheadditionof
tetramethylbenzidine(TMB)withaspectrophotometer
(wave-length450nm).4,11
Following the immunoassays and reading of the
spec-trophotometer,thenormalizedopticaldensities(ODns)were
obtained,asfollows:
ODnofthecontrols= medianODofthecontrols
medianODofthecalibrator
ODnofthesample= ODofthesample
Thetests were interpreted as follows: all samples with
ODn≤1.2 were tested in triplicate to confirm whether it
was a recent or long-term infection. Confirmation
inter-pretation: samples with OD>0.8 were characterized as
long-termseroconversionandthosewithOD≤0.8asrecent
seroconversion.12
HIV-1 incidence and 95% confidence interval (95% CI)
for the BED-CEIA and Ax-AI algorithms were calculated
using the spreadsheet providedbythe South African
Cen-tre for Epidemiological Modelling and Analysis (SACEMA)
(http://www.incidence-estimation.com/page/tools-for-incidence-from-biomarkers-for-recent-infection).13
Thefollowingparameterswerereportedonthespreadsheet:
the Estimated Mean Duration of Recent Infection (MDRI),
whichwas180daysfortheAx-AIand155daysforthe
BED-CEIA;4 the estimatedFalse Recent Rate (FRR%),which was
5.6%,14and10.6%fortheBED-CEIAandAx-AI,respectively;7,14
numberofHIV-positiveindividuals;numberofHIV-negative
individuals and number of recent HIV-1 infections.
Inci-denceestimateswereexpressedasthenumberofnewHIV
infectionsper100person-years.
Sensitivity,specificity,positiveandnegativepredictive
val-ues,accuracy,likelihoodratiosforpositiveandnegativetests,
andkappawereobtainedbycomparingtheresultsofAx-AI
tothoseofBED-CEIA,usingtheMSOfficeExcelversion2010
(Microsoft Corporation)and the SPSS forWindows version
12.0.Significantassociationsbetweenthevariableswere
con-sideredwhenp-valueswerelessthan0.5.
Thestudy population consistedof177 men(48.5%)and
188women(51.5%),ofwhom53(28.2%)werepregnant.The
medianagewas29years.Otherepidemiologicandbehaviour
dataarelistedinTable1.Ax-AIandBED-CEIAresultswere
comparedusing 365samples. One hundred and two
sam-pleswereidentifiedasrecentinfectionbyBED-CEIA,and77
samplesbyAx-AI.Forthestatusoflong-terminfection,the
BED-CEIAandAx-AIidentified263and288samples,
respec-tively.Bothtestswereconcordantin66samplesclassifiedas
recentinfectionand252asalong-terminfection.
WhencomparedtotheBED-CEIA,withacutoffof0.8,the
Ax-AIshowed64.7%sensitivityofand95.8%specificity,with
ratesofpositivepredictivevalues(PPV)and negative(NVP)
greaterthan85%.Theagreementbeyondchancebetweenthe
twotechniqueswasgood(kappa=0.654,p<0.001).
BED-CEIAandAx-AIresultsforsixpatientswhose
sero-conversiondatewasequaltoorlessthanayear,confirmedby
anegativeassayforHIVfollowedbyapositiveassaywithin
12months,demonstrateddisagreementinonlytwopatients
theclassificationstatusofrecentinfection.Inoneofthese
casesseroconvertedaftersevenmonthsandBED-CEIAODn
wasborderline(atthecutoffthreshold)(datanotshown).
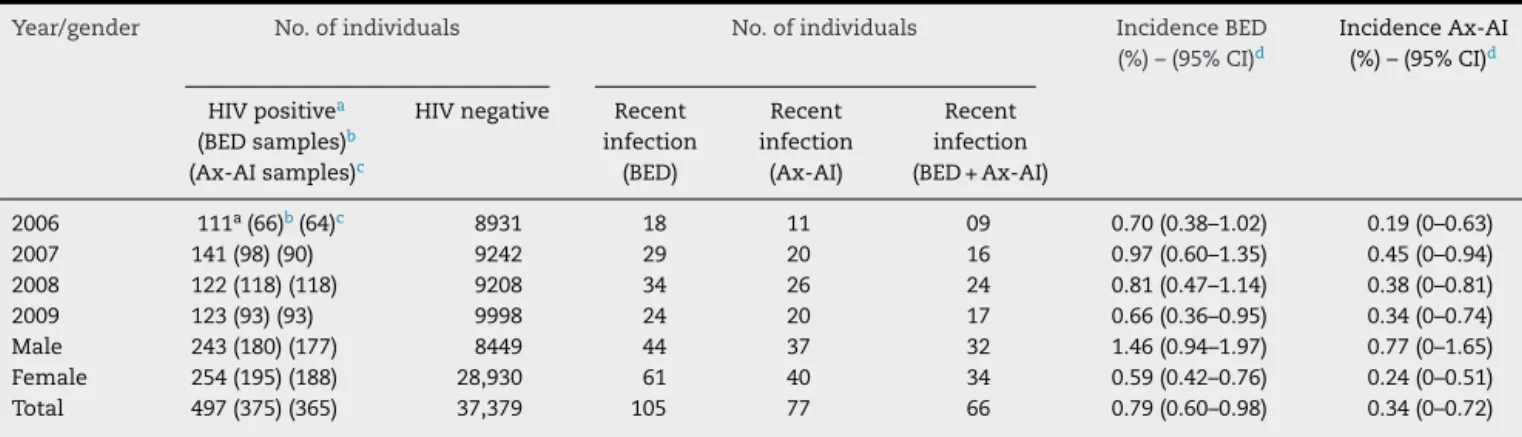
GlobalHIV-1incidencesaccordingtoBED-CEIAandAx-AI
were0.79(95%CI:0.60–0.98)and0.34(95%CI:0–0.72),
respec-tively. Thus,the incidencerate estimatedbythe BED-CEIA
was significantly higher (p>0.0001).The number ofrecent
infectionsamongHIVpositiveindividuals,fortheBED-CEIA,
Ax-AIandBED+Ax-AIwere 105,77 and66 (p=0.004).Only
theAx-AIdemonstratedasignificantincreasingtendencyof
HIVincidencewithtime (p=0.02). Theincidenceestimates
variedbetween0.66and0.97%/yearand0.19and0.45%/year
for the BED-CEIA and Ax-AI, respectively. All methods for
estimatingHIV-1incidenceshowedsignificantlyhigherrates
Table1–Demographicsandbehaviouraldataofpatients newlydiagnosedasHIV-positiveattwoVCTsin
Pernambuco–Brazil,2006–2009.
Variables HIV-positivepatients
Gender
Male 177(48.5)
Female 188(51.5)
Pregnant
Yes 53(28.2)
No 135(71.8)
Medianage(years) 29
Sexualorientation
Heterosexual 304(86)
MSMa 50(14)
Risksituationb
Bloodtransfusion 14
Sexworker 08
HIV+partner 26
Schooling
≤8years 231(63.3)
>8years 123(33.7)
Notinformed 11(3.0)
Employmentsituation
Employed 69(18.9)
Self-employed 42(11.5)
Unemployed 61(16.7)
Student 25(6.8)
Housewife 117(32.1)
Retired 09(2.5)
Notinformed 42(11.5)
a MSM,menwhohavesexwithmen.
b Notexpressedinpercentagesincemostpatientsdidnotpresent
thisrisk.
for men than for women (p<0.0001), with a difference of
approximatelythreefold(Table2).
The present study demonstrates that estimates of HIV
incidenceinapopulation seekingVCTsintwo
municipali-tieswithintheMetropolitanRegionofRecife,capitalcityof
Pernambuco,NortheastBrazil,mayvaryaccordingtothe
diag-nosticalgorithmused,witharateof0.79%/yearwhenusing
theBED and0.34%/yearwiththeAx-AI,excludingthoseon
HAART. TheBED-CEIA methodology has been widely used
to measurethe incidence ofHIV-1 incross-sectional
stud-ies,mainlyduetoitscommercialavailability.However,many
questions remain regardingits accuracy,witha suggestion
ofoverestimation,despitetheconsensualuse ofcorrection
factors.2TheWHOWorkingGrouponHIVIncidenceAssays
systematicallyreviewedtheaccuracyofserologicaltestsfor
recent infectionswithHIV.Across 13different assays,
sen-sitivity todetect recent infections ranged from 42 to100%
(median89%).Specificityfordetectingestablishedinfections
wasbetween49.5and100%(median86.8%)andwashigher
forinfectionsofdurationslongerthanoneyear(median98%,
range 31.5–100.0).Serologicalassayshavereasonable
sensi-tivity fordetectingrecentHIVinfection,but are vulnerable
tomisclassifyingestablishedinfectionsasrecent,potentially
leadingtobiasesinincidenceestimates.15Theuseofa
Table2–AnnualHIV-1incidenceestimatedbyusingtheBED-CEIA,Ax-AI,andBED-CEIA+Ax-AIalgorithms.
Year/gender No.ofindividuals No.ofindividuals IncidenceBED
(%)–(95%CI)d
IncidenceAx-AI
(%)–(95%CI)d
HIVpositivea
(BEDsamples)b
(Ax-AIsamples)c
HIVnegative Recent
infection (BED)
Recent infection
(Ax-AI)
Recent infection
(BED+Ax-AI)
2006 111a(66)b(64)c 8931 18 11 09 0.70(0.38–1.02) 0.19(0–0.63)
2007 141(98)(90) 9242 29 20 16 0.97(0.60–1.35) 0.45(0–0.94)
2008 122(118)(118) 9208 34 26 24 0.81(0.47–1.14) 0.38(0–0.81)
2009 123(93)(93) 9998 24 20 17 0.66(0.36–0.95) 0.34(0–0.74)
Male 243(180)(177) 8449 44 37 32 1.46(0.94–1.97) 0.77(0–1.65)
Female 254(195)(188) 28,930 61 40 34 0.59(0.42–0.76) 0.24(0–0.51)
Total 497(375)(365) 37,379 105 77 66 0.79(0.60–0.98) 0.34(0–0.72)
a Atotalof10HIV-positivepatientswereexcludedfromtheincidencecalculation:eighthadpresentedapreviouspositiveserologicaltest
forHIV,onewasverticaltransmission,andanotherwasundergoingantiretroviraltreatment.Threeoftheseindividualswerefromthe2006
cohort,twowerefromthe2007cohort,threewerefromthe2008cohort,andtwofromthe2009cohort.
b TotalnumberofavailablealiquotsforBED-CEIAtesting.
c TotalnumberofavailablealiquotsforAx-AItesting.
dResultsareexpressedastheincidence/100people/year(95%CI).
combinationwithanavidityassaywascitedasthemost
com-monlyusedalgorithm.2
Thepresent study tested this strategy inindividuals in
themetropolitanareaofRecife,NortheastBrazil,wherethe
mostprevalentsubtypesBare(around57%),F(around37.7%)
andC(around3.1%).16Asignificantdifferencewasobserved
betweenincidenceratesestimatedbyBED-CEIA(0.79%/year)
andAx-AI(0.34%/year)(p<0.00001).
One major difficulty when discussing estimates of HIV
incidenceisthedifferenceamongpopulationsstudied.Most
studieshaveatargetpopulationofgroupsatrisk,especially
sexworkers,menwhohavesexwithmen(MSM)andinjected
drugusers.Thisresearchevaluatedamixedpopulationfrom
twoVCTsintheBrazilianstateofPernambuco,whichwere
linkedtotheBrazilianMinistryofHealth’sNationalSTDand
AIDSProgramme.Inthisregion,thoseattendingtheVCTs
con-sistofthreemaingroups:peoplefromthegeneralcommunity,
usuallyoflowriskinfection,pregnantwomenfromwithinthe
community,andindividualswithbehaviourpatterns
associ-atedwithanincreasedriskofinfection(e.g.sexworkersand
MSM).Thus,onelimitationofthisstudyamongotherstudies
inBrazilistheheterogeneityofthepopulationstudiedplusthe
factthattheaccuracyoftheestimatesofincidenceby
sero-logicalmethodsdependsontheaccuracyofthemeanRITA
durationandtheRITAFRR,whicharethemeasuresevaluated
forpopulationswithcharacteristicsdifferentfromthoseofthe
presentstudy.17
Therearedataillustratingthattheaviditytestpresentsa
betterperformanceincorrectly classifyingindividuals with
along-termviralload,whiletheBED-CEIAhasgreater
ten-dency to classify them erroneously as recent infections.18
Furthermore,ithasbeenshownthatAI-Axsuffersless
inter-ference from the HIV subtype and antibody titres intheir
performance.6
The incidence estimates presented herein were about
threefoldhigherformenthanforwomen.TheBrazilian
epi-demicisconcentratedwithincertaingroups,especiallyyoung
MSM.1 However inthepresent study,thisgroup accounted
for only14%, thus, underrepresented inthe population of
VCTsstudied.InBrazil,therehavebeenseveralstudies,which
applied serological methods to estimate HIV-1 incidence,
especially inthe South and Southeast. Althougha
consid-erableportion ofthese studiesincluded individuals witha
higherriskforHIVinfection,suchasprisoners,cocaineand
injected drugsusers,MSMand high-riskheterosexualmen
andwomen,incidencedataforbothsexesshowahigherrate
formen,asobservedbySchechteretal.19andAlvesetal.,20
wheretheestimatedincidenceratewasbetween1.9and2%
forwomenand2.8and2.7%formenand,respectively.
Giventhelargenumberofdiagnostictestsperformedfor
HIVinfection(132million/year)andbloodbankscreening(101
million/year),aswellasrecognizingthehugemarketinvolved,
theimportanceofapplyingtheRITAmethodologyshouldbe
emphasized inorder toprovide abettermap ofthe spread
oftheepidemicandallowadequatestructuringofpreventive
strategies.Toourknowledge,thisisthefirststudyinBrazil
toapplytheconceptofthedualtestingalgorithmto
deter-mineestimatesofHIVincidence,basedonthemostrecent
recommendationsoftheWHO.2,14
Funding
ProgramaNacionaldeCooperac¸ãoAcadêmica–Ac¸ãoNovas
Fronteiras(Procad–NF)–Coordenac¸ãodeAperfeic¸oamento
dePessoaldeNívelSuperior(CAPES),MinistériodaEducac¸ão,
Brasil.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
r
e
f
e
r
e
n
c
e
s
1.BrazilianMinistryofHealthy.Boletimepidemiológico
AIDS-DST.AnoI,n◦
01.2013.Availablefrom:
2. WorldHealthOrganization(WHO),Switzerland.Whenand
howtouseassaysforrecentinfectiontoestimateHIV
incidenceatapopulationlevel.UNAIDSWorkingGroupon
GlobalHIV/AIDSandSTISurveillance.Availablefrom:
http://www.who.int/diagnosticslaboratory/hivincidence may13 final.pdf[cited23.07.12].
3. JanssenRS,SattenGA,StramerSL,etal.Newtestingstrategy todetectearlyHIV-1infectionforuseinincidenceestimates andforclinicalandpreventionpurposes.JAMA.
1998;280:42–8.
4. ParekhBS,KennedyMS,DobbsT,etal.Quantitativedetection ofincreasingHIVtype1antibodiesafterseroconversion:a simpleassayfordetectionrecentHIVinfectionand estimatingincidence.AIDSResHumRetroviruses. 2002;18:295–307.
5. HargroveJW,HumphreyJH,MutasaK,etal.ImprovedHIV-1 incidenceestimatesusingtheBEDcaptureenzyme immunoassay.AIDS.2008;22:511–8.
6. SuligoiB,GalliC,MassiM,etal.Precisionandaccuracyofa procedurefordetectingrecentHumanImmunodeficiency Virusinfectionsbycalculatingtheantibodyavidityindexby anautomatedimmunoassay-basedmethod.JClinMicrobiol. 2002;40:4015–20.
7. BrausteinSL,NashD,KimAA,etal.Dualtestingalgorithmof
BED-CEIAandAxSYMavidityindexassaysperformsbestin
identifyingrecentHIVinfectioninasampleofRwandansex
workers.PLoSONE.2011;6:e18402,
http://dx.doi.org/10.1371/journal.pone.0018402. 8. MurphyG,ParryJV.Assaysforthedetectionofrecent
infectionswithhumanimmunodeficiencyvirustype-1. Eurosurveillance.2008;13:4–10.
9. SuligoiB,MassiM,GalliC,etal.IdentifyingrecentHIV infectionsusingtheavidityindexandanautomatedenzyme immunoassay.JAcquirImmuneDeficSyndr.2003;32:424–8.
10.LoshenS,Bätzing-FeigenbaumJ,PoggenseeG,etal. ComparisonoftheHumanImmunodeficiencyVirus(HIV) type1-SpecificImmunoglobulinGcaptureenzyme-linked immunosorbentassayandtheavidityindexmethodfor identificationofrecentHIVinfections.JClinMicrobiol. 2008;46:341–5.
11.DobbsT,KennedyS,PauC,McDougalJS,ParekhBS. Performancecharacteristicsoftheimmunoglobulin
G-captureBED-enzymeimmunoassay,anassaytodetect recenthumanimmunodeficiencyvirustype1
seroconversion.JClinMicrobiol.2004;42:2623–8.
12.McDougalJS,ParekhBS,PetersonML,BrasonBM,DobbsT, AckersM,GurwithM.ComparisonofHIVtype1incidence observedduringlongitudinalfollow-upwithincidence estimatedbycross-sectionalanalysisusingtheBEDcapture enzymeimmunoassay.AIDSResHumRetroviruses. 2006;22:945–52.
13.KassanjeeR,McWalterTA,BärnighausenT,WelteA.Anew generalbiomarker-basedincidenceestimator.Epidemiology. 2012;23:721–8.
14.WorldHealthOrganization(WHO).ProceedingoftheMeeting
ontheDevelopmentAssaystoEstimateHIVincidence2009.
Availablefrom:
http://www.who.int/diagnosticslaboratory/links/fhichapel hillhivincidenceassayproceedings0409.pdf[cited 21.02.13].
15.GuyR,GoldJ,CallejaJM,etal.Accuracyofserologicalassays fordetectionsofrecentinfectionwithHIVandestimationof populationincidence:asystematicreview.LancetInfectDis. 2009;9:747–59.
16.CavalcantiAMS,BritoAM,SalustianoDM,LimaKO,SilvaSP, LacerdaHR.RecentHIVinfectionsratesamongHIVpositive patientsseekingvoluntarycounselingandtestingcentersin themetropolitanregionofRecife–PE,Brazil.BrazJInfectDis. 2012;16:157–63.
17.MorgadoMG,BastosFI.EstimatesofHIV-1incidencebasedon serologicalmethods:abriefmethodologicalreview.Cad SaúdePública.2011;27:S7–18.
18.BelloG,Valesco-de-CastroCA,BongertzV,etal.Immune activationandantibodyresponsesinnon-progressingelite controllerindividualsinfectedwithHIV-1.JMedVirol. 2009;81:1681–90.
19.SchechterM,LagoRF,MeloMF,etal.Identificationofa high-riskheterosexualpopulationforHIVpreventiontrialsin RiodeJaneiro,Brazil.ProjetoPrac¸aOnzeStudyGroup.J AcquirImmuneDeficSyndr.2000;24:175–7.