Effects
on
some
immunological
traits
after
crossing
three
rabbit
breeds
in
Egypt
Mahmoud
S.
El
‐
Tarabany
&
Khairy
M.
El
‐
Bayoumi
Summary
The authors evaluate the effect of crossing three purebred rabbit breeds in a complete 3 × 3 diallel crossbreeding experiment (New Zealand White, V‐line and Gabali) on certain immunological traits, including IgG titre. Sheep red blood cells (SRBCs) were administered at 8 weeks of age and boosted at 10 weeks of age. V‐line/Gabali crossbreeds gave the highest significant (p<0.05) level of serum total protein at 8 and 12 weeks of age (4.71 and 7.70 g/dl, respectively). Its reciprocal crossbreed Gabali/V‐line resulted in the highest estimate of serum globulin at 8 weeks of age (2.07 g/dl). V‐line/Gabali crossbreeds showed the highest positive heterosis percentage (H%) of total protein, albumin and globulin at 8 weeks of age (27.98, 30.28 and 25.00, respectively). However, its reciprocal cross‐ breeds, Gabali/V‐line, revealed negative estimates for total protein and albumin at the same age (–4.07 and –29.80). V‐line/Gabali crossbreeds gave the highest titre (3.167) and positive H% (0.69) of IgG to the second dose of SRBCs. On the contrary, the highest negative estimate to the second dose was recorded in Gabali/New Zealand White crossbreeds (–2.49). Using the Gabali breed as the female line gave superior results for most immunological parameters.
Keywords
Breed, Breeding, Crossbreed, Egypt, Heterosis, IgG, Immunity, Rabbit.
Gli
effetti
di
alcuni
tratti
immunologici
a
seguito
di
incrocio
di
tre
razze
di
coniglio
in
Egitto
Riassunto
Gli autori studiano l’effetto dell’incrocio di tre razze pure di conigli in un esperimento di incrocio 3 × 3 (New Zealand White, V‐line e Gabali) su alcuni tratti immunologici, compresi il titolo IgG. Sono state somministrate le cellule di globuli rossi di una pecora (SRBCs), a 8 settimane e ripetute a 10 settimane di età. L’incrocio delle razze V‐line/ Gabali ha dato il massimo risultato (p<0.05) nei livelli sierici di proteine totali sia a 8 che a 12 settimane di età rispettivamente (4.71 e 7.70 gr/dl). Il reciproco incrocio delle razze Gabali/V‐line ha portato al più alto livello di globulina di siero a 8 settimane di età (2.07 /dl). L’incrocio V‐line/ Gabali ha mostrato una maggiore concentrazione di eterosi (% H) delle proteine totali, albumina e globulina a 8 settimane di età (27.98, 30.28 e 25.00, rispettivamente). Tuttavia, il reciproco incrocio V‐line/Gabali, ha mostrato le titolazioni negative per proteine totali e albumina alla stessa età (–4.07 e –29.80). L’incrocio Gabali/V‐line ha mostratol il titolo più alto (3.167) ed eterosi positiva % (0.69) ad IgG nei confronti della seconda dose di SRBCs. Al contrario, la titolazione negativa più alta sulla seconda dose è stata registrata nell’ incrocio Gabali/New Zealand White (–2.49). Utilizzando la razza Gabali come la linea femminile, si dimostrano
risultati superiori rispetto ai parametri
immunologici.
Parole chiave
Allevamento, Coniglio, Egitto, Eterosi, IgG, Immunogenicità, Incrocio di razze, Razza.
Introduction
In recent years, domestic rabbits have been recommended as a good alternative source of dietary protein for the increasing human populations of developing countries (10). Genetic variations of antibody response to sheep erythrocytes have been demonstrated in several species, including mice (3), chickens (20) and rabbits (5).
The immune response to a natural, non‐ specific, non‐pathogenic, multi‐determinant and T‐cell‐dependent antigen, such as sheep red blood cells (SRBCs), provides an indication of the status of natural immunity (13). A study of antibody response in various age categories of rabbits showed that even day‐old rabbits are able to form specific antibodies of isotype IgG (8). Different reactivity in the haemagglutin‐ ation (HA) test and formation of antigen variant was detected in a study designed to analyse selected parameters of non‐specific humoral immunity in rabbits (11).
The objective of our work was to investigate the effects of purebred rabbits in comparison to crossbreeds and their reciprocal crosses on antibody response (IgG) to sheep red blood cells SRBCs and some blood parameters including total protein, albumin, globulin, albumin globulin ratio, as well as estimation of heterosis of such traits.
Materials
and
methods
This study was conducted at the rabbit farm of the Department of Animal Wealth Development at the Faculty of Veterinary Medicine of Zagazig University, Egypt.
Experimental
colony
The three pure rabbit breeds were used in this study were as follows:
New Zealand White
V‐line
El‐Gabali.
Two bucks and five does of 6 months of age from each breed were used in the experiment. Bucks and does were apparently healthy and vaccinated against pasteurellosis and viral haemorrhagic disease of rabbits (17) at 6 and 10 weeks of age, respectively.
Colony
management
and
mating
plan
Rabbits were housed in an open‐sided hutch. Breeding animals were kept individually in triangular galvanised wire cages (40 × 60 × 50 cm) provided with a nipple system for watering and a manual feeder. A metal nest box was attached to the doe’s cage.
Litters were weaned, ear tagged and separated in cages at 4 weeks of age. They were raised in identical management and nutritional con‐ ditions. Individual records were established for each breeding animal. Rabbits of all ages were fed commercial pelleted ration containing crude protein (not less than 18.5%), crude fibre (11.5%) and lipids (2.5%). Clean fresh water was available at all times to all rabbits. The environmental temperature was maintained at approximately 27°C. Mature bucks and does were mated in a full 3 × 3 diallel design. Two bucks and five does from each of the above three breeds were used.
Traits
studied
Blood parameters
Blood samples were collected from randomly assigned animals (purebred, crossbred and reciprocal crossbred) for serological examination. Blood samples were collected from the ear vein by puncture. Serum samples were taken in clean, sterilised and labelled tubes and were frozen at –20°C until assay (16). Specific diagnostic kits for total protein and albumin were used for colorimetric evaluation and the globulin concentration (g/d) was obtained as a total protein‐albumin concentration (4, 12). The source of the diagnostic kits was BK8145C (CinnaGen Inc. Shahrak Ekbatan, Teheran).
Immunological traits
100 ) P ‐ X P ‐ X ( 1/2 ) P ‐ X P ‐ X ( 1/2 ‐ F ‐ X % H 2 1 2 1 1
samples from each genotype were collected one week after each injection; the serum was separated and frozen at –20°C until assay. The enzyme‐linked immunosorbent assay (ELISA) was used to detect antibodies (IgG) as a response to SRBCs (Bethyl rabbit IgG ELISA quantitative kit: E120‐111, Bethyl Laboratories, Montgomery, Texas).
Data handling and statistical analysis
All available data were analysed using the SPSS Statistical Analysis System package (14). Individual titres were transformed to log10 and the log geometric mean (log GM) was calculated for each genotype in accordance with the following formula:
i X Log n 1 GM Log (22)
Least squares means (LSM) ± standard errors (SE) were calculated and tested for significance using the ‘T’ test (15).
The data were analysed using different statistical models as following:
eijk Cj Bi μ
Yijk
Yijk = any observed value μ = overall mean
Bi = effect of breed (i = 1, 2, and 3, i.e. New Zealand White, V‐line and El‐Gabali)
Cj= effect of crossing (j = 1, 2, 3… 6) ijk
e
= random deviation due to unexplainedsource.
Heterosis (hybrid vigor)
Heterosis or hybrid vigor is estimated for the different crossbreds and reciprocal crossbreds based on the following formula (9):
where: 1 F ‐
X = actual heterozygote mean of the first
generation
1
P ‐ X , P2
‐
X = means of the parents breed.
Results
LSM ± SE, as well as heterosis percentage (H%) of some blood parameters (serum total protein, albumin, globulin and albumin/globulin [A/G]
ratio) due to crossing New Zealand White, V‐ line and Gabali rabbits at 8 and 12 weeks of age are summarised in Tables I and II.
The log GM ± standard deviations (SD) of antibody titre (IgG) to SRBCs as well as the H% due to crossing of New Zealand White, V‐ line and Gabali rabbits at 8 and 12 weeks of age are presented in Tables II and III.
Discussion
Blood
parameters
There were non‐significant differences among different pure lines for most parameters at either 8 or 12 weeks of age. Similar results were recorded in other similar studies (2). The V‐line genotype had significantly (p<0.05) higher total protein and globulin (4.10 and 1.99 g/dl) at 8 weeks of age. The overall mean of crossbred animals was significantly higher than that of purebreds for serum albumin at 8 weeks of age (2.35 vs 2.06 g/dl).
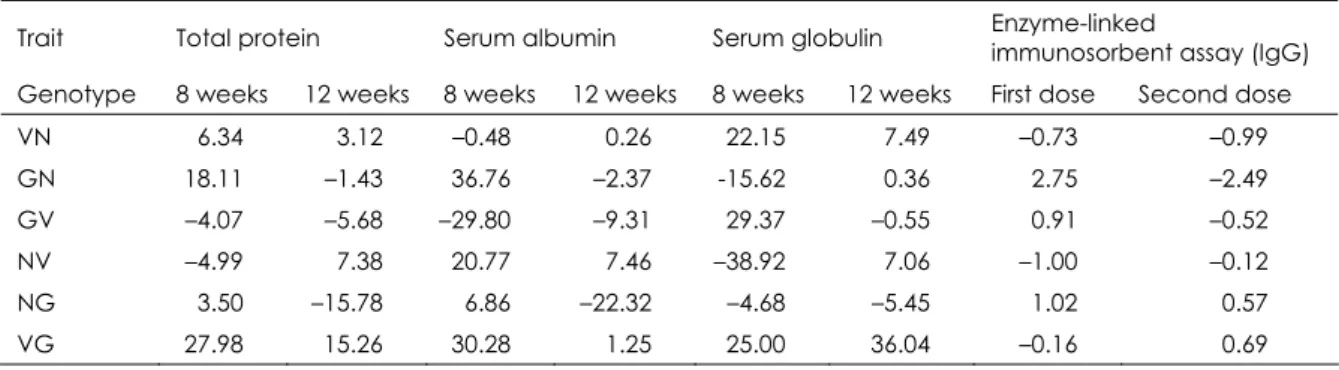
The V‐line/Gabali crossbreed recorded the highest significant (p<0.05) level of serum total protein at either 8 or 12 weeks of age (4.71 and 7.70 g/dl). In addition, Gabali/New Zealand White genotype gave the highest level of serum albumin at 8 and 12 weeks of age (2.79 and 4.11 g/dl). Meanwhile, V‐line/Gabali genotype had the highest estimate of serum globulin at 8 weeks of age (2.07 g/dl). Significant differences among crossbreeds at 12 weeks were reported (6). The author found that Flander × New Zealand White crossbred demonstrated high and significant levels of serum total protein at 12 weeks of age.
Table I
Least square means ± standard errors of some immunological traits at different age intervals due to crossing of New Zealand White, V-line and Gabali breeds
Trait Total protein Serum albumin Serum globulin Albumin/globulin
ratio
Genotype No. 8 weeks 12 weeks 8 weeks 12 weeks 8 weeks 12 weeks 8 weeks 12 weeks
NN 20 3.31(b)±0.
21 6.38(a) ±0.30 2.03(a) ±0.15 3.99(a) ±0.40
1.35(a, b)
±0.22 2.38(a) ±0.09 1.89(a) ±0.45 1.69(a) ±0.24
VV 20 4.10
(a) ±
0.20
5.80(a) ±
0.38
2.11(a) ±
0.20
3.51(a) ±
0.41
1.99(a) ±
0.32
2.29(a) ±
0.08
1.26(a) ±
0.32
1.54(a) ±
0.21
GG 17 3.26(b) ±
0.15
7.56(a) ±
0.73
2.05a ±
0.17
4.43(a) ±
1.07
1.21(b) ±
0.10
3.12(a) ±
0.42
1.77(a) ±
0.27
1.55(a) ±
0.49
Mean 57 3.56(A) ±
0.14
6.58(A) ±
0.35
2.06(B) ±
0.09
3.98(A) ±
0.37
1.49(A) ±
0.16
2.59(A) ±
0.18
1.64(A) ±
0.20
1.59(A) ±
0.17
VN 21 3.94(a, b) ±
0.25
6.28(a, b) ±
0.27
2.06ab ±
0.17
3.76(a) ±
0.16
2.04(a) ±
0.23
2.51(b) ±
0.20
1.08(a) ±
0.19
1.51(a) ±
0.14
GN 16 3.88(a, b) ±
0.19
6.87(a, b) ±
0.09
2.79(a) ±
0.42
4.11(a) ±
0.48
1.08ab ±
0.29
2.76(b) ±
0.42
2.08(a) ±
0.61
1.60(a) ±
0.39
GV 17 3.53(b) ±
0.59
6.30(a, b) ±
0.67
1.46(b) ±
0.18
3.60(a) ±
0.49
2.07(a) ±
0.43
2.69(b) ±
0.28
1.73(a) ±
0.09
1.35(a) ±
0.18
NV 21 3.52(b) ±
0.13
6.54(a, b) ±
0.40
2.50(a) ±
0.10
4.03(a) ±
0.28
1.02(b) ±
0.16
2.50(b) ±
0.13
2.68(a) ±
0.40
1.60(a) ±
0.06
NG 16 3.40(b) ±
0.29
5.87(b) ±
0.58
2.18(a, b)
± 0.31
3.27(a) ±
0.57
1.22ab ±
0.29
2.60(b) ±
0.03
2.09(a) ±
0.75
1.25(a) ±
0.20
VG 17 4.71(a) ±
0.32
7.70(a) ±
0.46
2.71(a) ±
0.25
4.02(a) ±
0.56
2.00(a) ±
0.32
3.68(a) ±
0.12
1.52(a) ±
0.28
1.10(a) ±
0.19
Mean 108 3.89(A) ± 0.13
6.59(A) ±
0.13
2.35(A) ±
0.13
3.80(A) ±
0.17
1.53(A) ±
0.14
2.79(A) ±
0.12
2.39(A) ±
0.56
1.40(A) ±
0.08
Means of purebreds, and crossbreds and its reciprocal within the same category having different superscripts (lower case) are significantly different (p<0.05)
Means of purebreds versus crossbreds and its reciprocal within the same column having different superscripts (upper case) are significantly different (p<0.05)
N New Zealand White V V-line
G Gabali
Table II
Heterosis percentages of some immunological traits for crossbreds and reciprocal crossbreds due to crossing of New Zealand White, V- Line and Gabali rabbits
Trait Total protein Serum albumin Serum globulin Enzyme-linked
immunosorbent assay (IgG) Genotype 8 weeks 12 weeks 8 weeks 12 weeks 8 weeks 12 weeks First dose Second dose
VN 6.34 3.12 –0.48 0.26 22.15 7.49 –0.73 –0.99
GN 18.11 –1.43 36.76 –2.37 -15.62 0.36 2.75 –2.49
GV –4.07 –5.68 –29.80 –9.31 29.37 –0.55 0.91 –0.52
NV –4.99 7.38 20.77 7.46 –38.92 7.06 –1.00 –0.12
NG 3.50 –15.78 6.86 –22.32 –4.68 –5.45 1.02 0.57
VG 27.98 15.26 30.28 1.25 25.00 36.04 –0.16 0.69
N New Zealand White V V-line
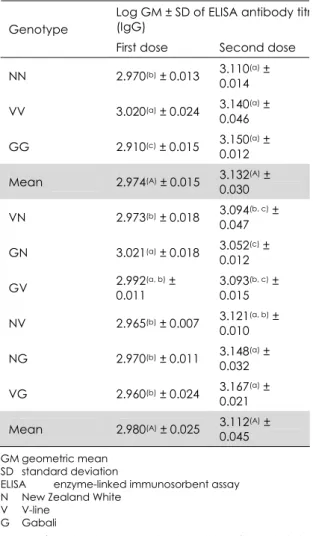
Table III
Log geometric mean ± standard deviation of enzyme-linked immunosorbent assay antibody titre (IgG) to sheep red blood cells due to crossing of New Zealand White, V-line and Gabali breeds
Log GM ± SD of ELISA antibody titr (IgG)
Genotype
First dose Second dose
NN 2.970(b) ± 0.013 3.110
(a) ±
0.014
VV 3.020(a) ± 0.024 3.140
(a) ±
0.046
GG 2.910(c) ± 0.015 3.150(a) ±
0.012
Mean 2.974(A) ± 0.015 3.132
(A) ±
0.030
VN 2.973(b) ± 0.018 3.094(b, c) ±
0.047
GN 3.021(a) ± 0.018 3.052
(c) ±
0.012
GV 2.992(a, b) ±
0.011
3.093(b, c) ±
0.015
NV 2.965(b) ± 0.007 3.121
(a, b) ±
0.010
NG 2.970(b) ± 0.011 3.148
(a) ±
0.032
VG 2.960(b) ± 0.024 3.167(a) ±
0.021
Mean 2.980(A) ± 0.025 3.112
(A) ±
0.045
GM geometric mean SD standard deviation
ELISA enzyme-linked immunosorbent assay N New Zealand White
V V-line G Gabali
Means of purebreds, and crossbreds and its reciprocal within the same category having different superscripts (lower case) are significantly different (p<0.05)
Means of purebreds versus crossbreds and its reciprocal within the same column having different superscripts (upper case) are significantly different (p<0.05)
New Zealand White crossbreds (–16.00) at three months of age (7).
Antibody
titre
(IgG)
against
sheep
red
blood
cells
The log GM ± SD of antibody titre (IgG) to SRBCs as well as H% due to crossing of New Zealand White, V‐line and Gabali rabbits at 8 and 12 weeks of age are given in Tables II and III.
Purebred New Zealand White rabbits had the highest significant IgG titre to the first dose of SRBCs (3.02). Non‐significant differences were detected among purebred rabbits to the second dose of SRBCs. These results confirmed those obtained in a study to evaluate antibody response among temperate rabbit breeds by Thomas and Nandakumar (18) who found that the effect of breed was not significant on the antibody response at 0, 7 and 14 days post immunisation.
Non‐significant differences were observed among overall purebreds and crossbreds at either first or second dose against SRBCs. The highest titre to the first dose was recorded for the Gabali/New Zealand White crossbreed (3.02), while the same crossbreed demon‐ strated the lowest estimate after the second dose (3.052). V‐line/Gabali crossbreeds gave the highest IgG titre (3.167) to the second dose of SRBCs. Significant variations among crossbreeds were recorded by Hassan (6) who found that Flander × New Zealand White crossbred showed the highest antibody titre to the first dose of SRBCs.
were studied. The IgG titre increased sharply up to 7‐14 days post immunisation (19).
Conclusion
From the above results, the Gabali breed is used as the female line at crossing, demonstrating the superiority of most immunological parameters, especially the serum globulin and IgG titre, to SRBCs.
References
1. Abdel-Hamid T.M. 2007. Genetic studies on some productive, reproductive and immunological traits
in rabbits. MSc thesis, Faculty of Veterinary Medicine, Zagazig University, Zagazig, 234 pp.
2. Basha H.I. 2004. Biochemical genetic variations due to crossing of rabbit. MSc thesis, Faculty of
Veterinary Medicine, Zagazig University, Zagazig, 229 pp.
3. Biozzi G., Stiffel C., Mouton D., Bouthillier Y. & Decreusefond C. 1970. Serum concentration and
allotypes of immunoglobulins in two lines of mice genetically selected for ‘high’ or ‘low’ antibody synthesis. J Exp Me d, 132, 752-764.
4. Coles E.H. 1974. Veterinary clinical pathology. W.B. Saunders Company, Philadelphia, 211-213.
5. El-Bayomi K.H.M., Mandour M.A., Sharaf M.M., El-Fiky S.A. & Hemeda S.H.A. 1992. Estimation of
heterosis and combining ability of humoral and cell- mediated immune response in rabbit crosses.
In Proc. Second Congress of the Faculty of Veterinary Medicine, Cairo University, 2-4 March, Cairo.
Faculty of Veterinary Medicine, Cairo University, Cairo, 127-136.
6. Hassan R.A. 2005. Effect of crossing on productive performance of rabbits. MSc thesis, Faculty of
Veterinary Medicine, Suez Canal University, Ismailia, 134 pp.
7. Hemeda Sh.A. 1991 Genetic variation in immune response of rabbit to some antigens. PhD thesis,
Faculty of Veterinary Medicine, Alexandria University, Alexandria, 208 pp.
8. Jeklova E., Leva L., Kudlackova H., & Faldyna M. 2007. Functional development of immune response
in rabbits. Ve t Immuno l Immuno p a tho l, 118, 221-228.
9. Legates J.E & Warwick E.J. 1990. Breeding and Improvement of farm animals, 8th Ed. McGraw Hill
Publications in the Agricultural Sciences, New York, 342 pp.
10. Lukefahr S.D. & Cheeke P.R. 1991. Rabbit project development strategies in subsistence farming
systems. 1. Practical considerations. Wo rld Anim Re v, 68, 60-70.
11. Niedźwiedzka-Rystwej P. & Deptuła W. 2009. Non-specific humoral immunity in rabbits infected with
the selected German strains of the RHD (rabbit haemorrhagic disease) virus. J Immuno l, 34 (4),
218-221.
12. Peterson R.G., Nash T.E., & Shelford J.A. 1982. Heritabilities and genetic correlation for serum and
production traits of lactating Holsteins. J Da iry Sc i, 95, 1556-1561.
13. Pinard M.H., Van Arendonk J.AM., Nieuwland M.G.B. & Van der Zijpp A.J. 1992. Divergent selection
for immune responsiveness in chickens: estimation of realized heritability with an animal model.
J Anim Sc i, 70, 2986-2993.
14. Statistical Package for Social Science (SPSS) 2001. SPSS/PC+ (2001), for the PC/XT. SPSS INC, an IBM
Company, Chicago, Illinois.
15. Steel R.G.D. & Torrie J.H. 1960. Principles and procedures of statistics McGraw Hill Book Comp. Inc.,
New York, 672 pp.
16. Stoot G.H. & Fellah A. 1983. Cholesterol immunoglobulin absorption nearly related to concentration
for calves. J Da iry Sc i, 6, 1315-1328.
17. Swift S.T. 1999. Diseases of domestic rabbits, Second Ed. Canadian Council on Animal Care (CCAC),
Ottawa, 67-68.
18. Thomas M. & Nandakumar P. 2001. Genetic influence of antibody response to chicken RBC among
19. Tripathi T., Shahid M., Raza A., Khan H.M., Siddiqui M., Malik A. & Ali Khan R. 2010. In vivo
comparative immunotoxic study of histamine receptors (H1R, H2R, H3R and H4R)-agonist. Ea ste rn J
Me d, 15, 48-56 (www.easternjmed.org/PDF/2010_2/48.pdf accessed on 15 August 2010).
20. Van der Zijpp A.J., Frankena K., Boneschanscher J. & Nieuwland M.G.B. 1983. Genetic analysis of
primary and secondary immune responses in the chicken. Po ult Sc i, 62, 565-572.
21. Van der Zijpp A.J., Scott T.R. & Glick B. 1986. The effect of different routes of antigen administration on the humoral immune response of the chick. Po ult Sc i, 65, 809-811.