Brazilian
Journal
of
OTORHINOLARYNGOLOGY
www.bjorl.org
ORIGINAL
ARTICLE
Correlation
between
vascular
endothelial
growth
factor
expression
and
presence
of
lymph
node
metastasis
in
advanced
squamous
cell
carcinoma
of
the
larynx
夽
,
夽夽
Rodrigo
Gonzalez
Bonhin
∗,
Vanessa
Brito
Campoy
Rocha,
Guilherme
Machado
de
Carvalho,
Alexandre
Caixeta
Guimarães,
Agrício
Nubiato
Crespo,
Carlos
Takahiro
Chone,
Eliane
M.I.
Amstalden
UniversidadeEstadualdeCampinas(UNICAMP),Campinas,SP,Brazil
Received15June2013;accepted24August2014 Availableonline20November2014
KEYWORDS Squamouscell carcinomaoflarynx; VEGF;
Immunohistochemistry; Cervicallymphatic metastases
Abstract
Introduction:Squamouscellcarcinomaisthemostcommonneoplasmofthelarynx,andits evolutiondependsontumorstaging.Vascularendothelialgrowthfactorisamarkerof angio-genesis,anditsexpressionmayberelatedtoincreasedtumoraggressiveness,asevidencedby thepresenceofcervicallymphaticmetastases.
Objectives:To evaluatethe expressionofthevascular endothelialgrowth factor markerin non-glotticadvancedsquamouscellcarcinomaofthelarynx(T3/T4)andcorrelateitwiththe presenceofcervicallymphnodemetastases.
Methods:Retrospectiveclinicalstudyandimmunohistochemicalanalysisofvascular endothe-lialgrowthfactor throughthe Germanscaleofimmunoreactivityinproducts ofnon-glottic squamouscellcarcinomas.
Results:Thisstudyanalyzed15casesofadvancednon-glotticlaryngealtumors(T3/T4),four ofwhichexhibitedcervicallymphaticmetastases.Therewasnocorrelationbetweenvascular endothelialgrowthfactorexpressionandthepresenceofcervicalmetastases.
Conclusion:Althoughvascularendothelialgrowthfactorwasexpressedinafewcases,there wasnocorrelationwiththespreadofcervicallymphmetastases.
© 2014Associac¸ãoBrasileira de Otorrinolaringologiae CirurgiaCérvico-Facial. Publishedby ElsevierEditoraLtda.Allrightsreserved.
夽 Pleasecitethisarticleas:BonhinRG,RochaVB,deCarvalhoGM,GuimaraesAC,Crespo AN,ChoneCT,etal.Correlationbetween vascularendothelialgrowthfactorexpressionandpresenceoflymphnodemetastasisinadvancedsquamouscellcarcinomaofthelarynx. BrazJOtorhinolaryngol.2015;81:58---62.
夽夽Institution:DepartmentofOtorhinolaryngology,HeadandNeck,UniversidadeEstadualdeCampinas(UNICAMP),Campinas,SP,Brazil.
∗Correspondingauthor.
E-mail:[email protected](R.G.Bonhin).
http://dx.doi.org/10.1016/j.bjorl.2014.08.016
1808-8694/©2014Associac¸ãoBrasileiradeOtorrinolaringologia eCirurgiaCérvico-Facial. PublishedbyElsevierEditoraLtda.All rights
PALAVRAS-CHAVE Carcinoma
epidermoidede laringe; VEGF;
Imunohistoquímica; Metástase
Correlac¸ãoentreexpressãodofatordecrescimentoendotelialepresenc¸ade metástaselinfáticanoscarcinomasepidermoidesavanc¸adosdelaringe
Resumo
Introduc¸ão: Ocarcinomadecélulasescamosaséaneoplasiamaisfrequentedalaringeeseu prognóstico depende do estadiamento. A progressão da doenc¸a está relacionada a fatores intrínsecoscelularesdocâncer,nãoconhecidos.OVEGF(vascularendothelialgrowthfactor)é ummarcadordeangiogêneseesuaexpressãopodeestarrelacionadaaumamaioragressividade tumoral,evidenciadapelapresenc¸ademetástaseslinfáticascervicais.
Objetivos: AvaliaraexpressãodomarcadorVEGFemcarcinomadecélulasescamosasdalaringe avanc¸ados(T3/T4),não glóticosecorrelacionarquantoàpresenc¸ade metástaseslinfáticas cervicais.
Método: EstudoclínicoretrospectivodeanáliseimunohistoquimicadoVEGFatravésdaescala Germânicadeimunorreatividadeemprodutosdecarcinomasepidermóidesnãoglóticos.
Resultados: Analisados15casosdetumoresavanc¸adosdelaringe(T3/T4)nãoglóticos,sendo setecompresenc¸ademetástaseslinfáticascervicais.Nãohouvecorrelac¸ãoentreaexpressão doVEGFeapresenc¸ademetástasescervicais.
Conclusão:OVEGFfoipoucoexpressadonoscasosestudadosenãofoiobservadasuacorrelac¸ão comadisseminac¸ãodemetástaselinfáticacervical.
©2014Associac¸ãoBrasileiradeOtorrinolaringologiaeCirurgiaCérvico-Facial.Publicado por ElsevierEditoraLtda.Todososdireitosreservados.
Introduction
Angiogenesis is characterized by the formation of new capillary blood vessels that originate from a pre-existing vasculature.This processisessentialtoprovidenutrients, oxygen,andgrowthfactorsthat supportcellfunction and survival.Therefore,itisassociatedwithtumorgrowthand metastasis.1,2
Acomplexinteractionbetweenendothelialcells, extra-cellularproteinmatrix,andsolublefactorsinplasmaoccurs inangiogenesis.Endothelialcellsleavetheirquiescentstate whenstimulatedbyVEGF(vascularendothelialgrowth fac-tor)andinitiatethefollowingsteps:dissolutionofthebasal membrane;migrationandproliferationofendothelialcells; capillary tube formation; and maturation and survival of newlyformedvessels.Thisensurestherequiredblood sup-plyforneoplasticevolution.1,2
Squamouscellcarcinomaisthemostcommonneoplasm ofthe larynxanditsprognosisdependsonthesizeof the lesion,theleveloflocalinvasion,cervicallymphaticspread, andpresenceofdistantmetastases.1,3
Histologically,squamouscellcarcinomaofthelarynxand hypopharynx aresimilar to that of other regions and are classified into different degrees of differentiation by the neoplastic proliferation of squamouscells and the nature of tumor infiltration. Depending on the degree of kera-tinizationand cellular atypia, theyare classified aswell, moderately,orpoorlydifferentiated.3
Lymphatic drainageof the glotticregionis scarce; this decreasesthemetastaticpotentialoftumorsthatdevelopin thisregion.Thesupraglotticandsubglotticregionshaverich lymphatic drainage, with the supraglottic region draining mainlyintotheupperjugulocarotidchainlymphnodesand toalesserdegreeintothemiddlejugulocarotidchainlymph
nodeswhereasthesubglotticregiontendstodrain ipsilat-erallytothemiddleandlowerjugulocarotidlymphnodes, thatmaydraintotheanterosuperiormediastinalchain.3For
thisreason,wedecidedtoexcludeglottaltumorsfromthe study.
Theliteraturediscussingtreatmentofthesetumorshas increasedandstudiesofnewchemotherapeuticagentsand targeted therapies have gained prominence. Examples of thisincludethepromising resultsreportedforcetuximab, amonoclonalantibody thatblocksendothelialgrowth fac-torreceptor(EGFR)usedforthetreatmentofheadandneck cancer,andbevacizumabandotherrecombinantanti-VEGF monoclonalantibodies.4,5
TheseantibodiesbindtoVEGFisoformswithhighaffinity andpreventthecytokinefrombindingwiththeendothelial cellreceptorandtriggeringthe angiogenesisprocess.The useofangiogenesis-inhibitingmonoclonalantibodiesforthe treatmentofcolorectal,kidney,andlungcancerhasalready beenapprovedbytheFDA.Forthetreatmentoflarynx/head andneckcancer,theyarestillbeingassessed.4,5
ConsideringthatVEGFplaysanimportantroleintumor angiogenesisandthatthisfactor is associatedwithtumor formation,progression,andmetastasis,itsexpressioncould berelatedtoincreasedtumoraggressivenessevidencedby cervicalmetastaticspread.2
The present studyaimedtoevaluate theexpression of VEGF markerin advanced, non-glottic squamouscell car-cinoma of the larynx (T3/T4) and correlate it with the presenceorabsenceofcervicallymphnodemetastasis.
Methods
cohort,usingmaterials (surgicalspecimens)obtainedfrom theresectionofnon-glotticsquamouscellcarcinomaofthe larynxfromsurgeriesinatertiaryuniversityhospitalduring aspecified periodoftenyears.Theproceduresperformed withthepatientswerehemi-laryngectomyortotal laryngec-tomy,withrespectivelymphadenectomy.
Patients were divided by the presence or absence of metastases and by surgical-clinical staging. Cases of non-glottic squamous cell carcinoma of the larynx and hypopharynx were obtained from the tumor bank of the PathologicalAnatomyDepartmentofatertiaryhospital.Of these,15casesofadvancednon-glotticlarynxtumorswere selected,aswellasthreecasesoftumorsofthebaseofthe tongue,tencasesofsupraglottictumors,andtwocasesof hypopharynx/pyriformsinustumors.
Inclusionandexclusioncriteria
Tumorswhose paraffin blockswerelocatedand contained enough material for new sections were included in the survey. We required adequate tissue and data for stag-ing each lesion by its tumor location, and determining lesion size,level of invasion into adjacent tissues, lymph nodeinvolvement,anddistantmetastasis.Clinicaldataand tumorstagingwereobtainedfrompatients’records.
Allcases that werenot in accordancewiththe above-mentionedcriteriawereexcluded.
Immunohistochemicalanalysis
Immunohistochemical analysis was carried out using the avidin-biotin-peroxidase method and the VEGF antibody (type IgG1, 1:100 dilution, Santa Cruz, pretreated with citratebuffer).
Positiveandnegativemarkercontrolswerepreparedin ordertocompare withthe studiedcases.Lymphoid tissue wasusedaspositivecontrol(aspermanufacturer’s instruc-tions).
Slidereadingwascarriedoutrandomly,byaninvestigator whoneitherhadknowledgeofthepreviousdiagnosisgiven bythe pathologist norknowledge of thepatient’s clinical status,bycomparingthe slideswithpositiveandnegative controls.Scannedphotographsathighmagnification(400×) were made of highly reactive areas (hot spots), using a NikonCOOLPIX Camera 995. Afterthat, the imageswere senttothecomputer forhistologicalanalysis,which used theImagelab2000software.
Allparameterswereevaluatedinablindedmanner. Finally, thehistologicaland immunohistochemical find-ingswere correlatedwiththe TNMstage (TNM-UICC-AJCC Classification[2010]).6
ThedegreeofVEGFexpressionwasbasedontheGerman scaleof immunoreactivity.This consistsinmultiplyingthe coefficientoftheimmunomarkerexpressionintensitybythe percentagecoefficientofpositivecells.
The coefficientofthe immunomarkerexpression inten-sity was graded from 0 to 3, thus characterized: 0, no staining; 1, stained weakly; 2, stained moderately; 3, stainedstrongly.
Toobtainthepercentagecoefficientofpositivecells,the following sequence wasused: coefficient, represented by
Table 1 Distribution of 15 patients according to TNM staging.
T3 T4
N0 4 7
N1/N2/N3 2 2
thenumberofpositive(stained)andnegative(notstained) cells,wascountedonasamplespaceofatleast500cells, and stratified asfollows: coefficient of 0 ifthere wasno staining;coefficientof1iftherewas1---10%positivity; coef-ficientof2iftherewas11---50%positivity;coefficient of3 iftherewas51---80%positivity;andcoefficientof4ifthere was81---100%positivity.
The final value of the German scale of immunore-activity (coefficient of the immunomarker expression intensity×coefficient of positive cell percentage) ranged
from 0 to12, stratified as: 0 (negative), 1---4 (poor), 5---8 (moderate), and 9---12 (strong). To facilitate the analysis, the negative/weakcases were considered asgroup 1 and themoderate/strongcasesasgroup2.
Statisticalanalysis
Afterpoolingthedata,statisticalanalyseswereperformed using the IBM software SPSS Statistics®, using the chi-squaredtestandFisher’sexacttest.
To simplify and better analyze the data, they were groupedasfollows:
• GermanScaleofImmunoreactivity:
Negativeandweak---Group1 Moderateandstrong---Group2
• Cellproliferationindex:
Lowandslightcellproliferation---Group1 Moderateandhighcellproliferation---Group2
• TNM(TNM-UICC-AJCCClassification---2010)6
T1andT2---Group1/T3andT4---Group2 N0---Group0/N1,N2,andN3---Group1
Ethicalaspects
The study was approved by the Ethics Committee of the institution(485/11).
Results
Atotalof15caseswereevaluated,includingfourwith cer-vicalmetastases.
Thestagingofthesetumors,definedthroughTNM(2010), isdescribedinTable1.Itcanbeobservedthatonlyfourof the15caseshadcervicalmetastases.Table1furtherdetails thedistribution ofthefour caseswithcervicalmetastasis accordingtotheTclassification.
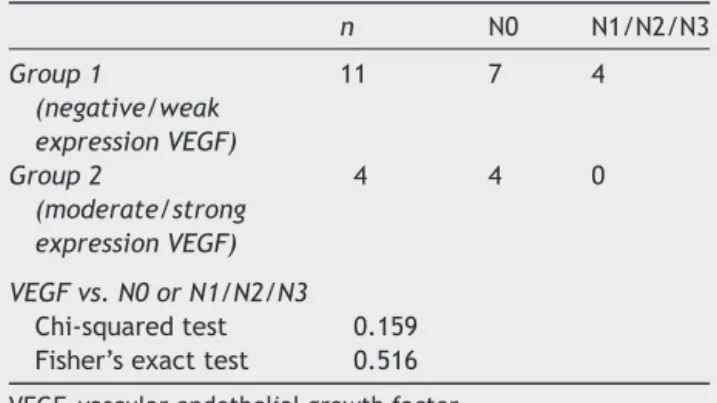
Table2 Distributionofgroups1and2(VEGFexpression) in15cases,theircorrelationwiththepresenceofcervical metastases,andstatisticalanalysis.
n N0 N1/N2/N3
Group1
(negative/weak expressionVEGF)
11 7 4
Group2
(moderate/strong expressionVEGF)
4 4 0
VEGFvs.N0orN1/N2/N3
Chi-squaredtest 0.159 Fisher’sexacttest 0.516
VEGF,vascularendothelialgrowthfactor.
inepithelialcellsofsquamouscarcinomaswasobservedin only26.6%ofthecasesstudied.
Table2alsoshowsthecorrelationofVEGFexpressionin groups 1 and 2, according tothe presence or absenceof cervicallymphnodemetastasis,withp=0.363.
There was no correlation between the overexpression of this marker with the presence of cervical lymph node metastasis(p=0.159).
Discussion
Intheliterature,studiesevaluatingheadandnecktumors, which include not only squamous cell carcinomas of the larynx (glottic and non-glottic), but also oral cavity and pharynx tumors, have reported an increase in VEGF correlatedwithincreasedangiogenesis,withneoplastic pro-gression,andwithprognosis;theanalysestookintoaccount thepositivityofthemarker,regardlessofitsintensity.1,2,7---9
Thereis only onerecent studythat specificallyaddressed laryngealcanceranditsassociationwithVEGF.Inthiscase, thepositivityof thismarkerwashigherthan72%,10 unlike
theresultsfoundhere,perhapsduetothesmallnumberof casesinthepresentsample.
Thereisdivergenceamongauthorsregardingthe corre-lationbetweenVEGFexpressionandthepresenceoflymph nodeanddistantmetastases.Someauthorsfound positive resultsbetweenVEGFexpressionandthepresenceof cervi-callymphnodemetastasesanddistantmetastases.2,8,9,11
The meta-analysisbyPanayiotisetal.foundthatVEGF correlates with poor survival in patients with head and neck squamous cell carcinoma; however, the correlation of VEGF overexpression withthe presence of lymphnode metastasis/distant metastasis is yet to be established.12
Boonkitticharoen also found no correlation between this markerandmetastases.13
A recent Asian study reported a close association between VEGF-C/VEGFR-3 expression and lymph node metastasesinsquamouscellcarcinomasofthelarynx.The expressionofthesemarkerswaspositiveonlyinthe analy-sisofcervicalmetastases,anddidnotcorrelatewithage, gender,Tstage,orprimarylocationofthelaryngeallesion.14
The antibody againstepithelial growthfactor receptor (EGFR) has already been approved by the FDA (Food and DrugsAdministration)forafewyearsforthetreatmentof
headandneckcarcinoma.However,itsuseinpatientswith tumorrecurrenceand/ormetastaticdiseasehasshownlittle efficacy.VEGFhasbeenimplicatedasapotentialmechanism ofresistancetoanti-EGFRtherapy.Therefore,recent clini-caltrialsseektheuseofanti-EGFRdrugsincombinationwith anti-VEGFantibodies(bevacizumab),acombinationthathas beenwelltoleratedbypatients(PhaseI/IIstudies).15---17
Currently, several phase II clinical trials have stud-ied the role of bevacizumab in tumors of the head and neck,includingitscombinationwithcetuximab,cisplatin, and radiotherapy for non-metastatic stage III/IV cancer; witherlotinibin locoregionaladvanced and/or metastatic disease; and with cetuximab, docetaxel, and cisplatin (induction) followed by cetuximab, cisplatin, and radio-therapy in previously untreated locoregional advanced disease.18
Theemergenceofthispromisingtreatmentraises impor-tantissues.Withbroad indications,theindiscriminateuse of bevacizumab can develop resistance. Therefore, stud-ies have sought to define and validate biomarkers of its efficiency. Until then, specific VEGF genotypes and arte-rialhypertension(inducedoraggravatedbythetreatment) appeartoberelatedtoabetterresponse.19Othermarkers
suchasCD105, CD34,andCD31,relatedtotheprocessof angiogenesis,maybeusefulintheidentificationofpatients whoarecandidatesformoreaggressiveanti-neoplastic ther-apy,includingtheuseofmonoclonalantibodies.20
SomeauthorsbelievethattheregulationofVEGF expres-sion can be modulated by the microRNA-206, and this pathwaycanbeusedin thefuturefor anticancertherapy relatedtothistumormarker.21
AlthoughthisstudydidnotobserveacorrelationofVEGF withcervicallymphnodemetastasis,thetargetedtherapy hasshownpromisingresultsforthetreatmentofheadand neckcancer.22
Conclusion
VEGFwasscarcelyexpressed,andnocorrelationwith cer-vicallymphmetastasisspreadwasobservedinthissample of15cases.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
References
1.NieD,HonnKV.Eicosanoidregulationofangiogenesisintumors. SeminThrombHemost.2004;30:119---25.
2.Sappayatosok K, Maneerat Y, Swasdison S, Viriyavejakul P, DhanuthaiK, ZwangJ, etal.Expression ofpro-inflammatory protein, iNOS, VEGF and COX-2 in oral squamous cell car-cinoma (OSCC), relationship with angiogenesis and their clinico-pathologicalcorrelation.MedOralPatolOralCirBucal. 2009;14:E319---24.
3.Altemani AMAM, AmstaldenEMI. Bogliolo.Patologia.8th ed. GuanabaraKoogan;2009.Diagnostichistopathologyoftumors. 3rded.Elsevier;2007.p.158---65.
4.Mohamed A, El-Rayes B, Khuri FR, Saba NF. Targeted
past decade. Crit Rev Oncol Hematol. 2014;91:186---96,
http://dx.doi.org/10.1016/j.critrevonc.2014.01.010.
5.DorseyK,AgulnikM.Promisingnewmoleculartargeted thera-piesinheadandneckcancer.Drugs.2013;73:315---25.
6.EdgeSB, ByrdDR, ComptonCC,Fritz AG,GreeneFL, Trotti A. Cancer stagingmanual. In:American JointCommittee on Cancer(AJCC).7thed.NewYork:Springer;2010.
7.GalloO,FranchiA,MagnelliL,SardI,VannacciA,BoddiV,etal. Cyclooxigenase-2pathwaycorrelateswithVEGFexpressionin headandneckcancer.Implicationfortumorangiogenesisand metastasis.Neoplasia.2001;3:53---61.
8.GalloO,MasiniE,BianchiB,BruschiniL,PaglieraniM,FranchiA. Prognosticsignificanceofcyclooxygenase-2pathwayand angio-genesisinheadandnecksquamouscellcarcinoma.HumPathol. 2002;33:708---14.
9.LimSC,ParkSY,DoNY.Correlationofcyclooxygenase-2pathway andVEGFexpressioninheadandnecksquamouscellcarcinoma. OncolRep.2003;10:1073---9.
10.SulluY,GunS,AtmacaS,KaragozF,KandemirB.Poorprognostic clinicopathologicfeaturescorrelatewithVEGFexpressionbut notwithPTENexpressioninsquamouscell carcinomaofthe larynx.DiagnPathol.2010;5:35.
11.Kyzas PA,Stefanou D, Agnantis NJ. COX-2expression corre-lates with VEGF-C and lymph node metastases in patients with head and neck squamous cell carcinoma. Mod Pathol. 2005;18:153---60.
12.KyzasPA,CunhaIW,IoannidisJPA.Factorimmunohistochemical expressioninheadandneckprognosticsignificanceofvascular endothelialgrowthsquamouscellcarcinoma:ameta-analysis. ClinCancerRes.2005;11:1434---40.
13.BoonkitticharoenV,KulapaditharomB,LeopairutJ,Kraiphibul P, Larbcharoensub N, Cheewaruangroj W, et al. Vascular endothelialgrowth factorA and proliferationmarker in pre-diction of lymph node metastasis in oral and pharyngeal squamous cell carcinoma.Arch OtolaryngolHead Neck Surg. 2008;134:1305---11.
14.WangZ,ChenY,LiX,XuL,MaW,ChangL,etal.Expressionof VEGF-C/VEGFR-3inhumanlaryngealsquamouscellcarcinomas anditssignificanceforlymphaticmetastasis.AsianPacJCancer Prev.2012;13:27---31.
15.CohenEE,DavisDW,KarrisonTG,SeiwertTY,WongSJ,Nattan S,etal.Erlotinibandbevacizumabinpatientswithrecurrent ormetastaticsquamous-cellcarcinomaoftheheadandneck: aphaseI/IIstudy.LancetOncol.2009;10:247---57.
16.ArgirisA,KotsakisAP,HoangT,WordenFP,SavvidesP,GibsonMK, etal.Cetuximabandbevacizumab:preclinicaldataandphase IItrialinrecurrentormetastaticsquamouscellcarcinomaof theheadandneck.AnnOncol.2013;24:220---5.
17.Yoo DS, KirkpatrickJP, CraciunescuO, Broadwater G, Peter-son BL, Carroll MD, et al. Prospective trial of synchronous bevacizumab, erlotinib and concurrent chemoradiation in locally advanced head and neck cancer. Clin Cancer Res. 2012;18:1404---14.
18.Cohen RB. Current challenges and clinical investigations of epidermal growth factor receptor (EGFR)- and ErbB family-targetedagentsinthetreatmentofheadandnecksquamous cellcarcinoma(HNSCC).CancerTreatRev.2014;40:567---77.
19.JubbAM, Harris AL. Biomarkers to predict the clinical effi-cacy of bevacizumab in cancer. Lancet Oncol. 2010;11: 1172---83.
20.LionelloM,StaffieriA,MarioniG.Potentialprognosticand ther-apeuticroleforangiogenesismarkersinlaryngealcarcinoma. ActaOtolaryngol.2012;132:574---82.
21.ZhangT,LiuM,WangC,LinC,SunY,JinD.Down-regulation of MiR-206 promotes proliferation and invasion of laryn-geal cancer by regulating VEGF expression. Anticancer Res. 2011;31:3859---63.