Soldag. insp. vol.19 número2
Texto
Imagem



Documentos relacionados
A Constituição Federal de 1988 formaliza a participação nas políticas públicas e as leis orgânicas das diversas políticas (saúde, assistência social, criança
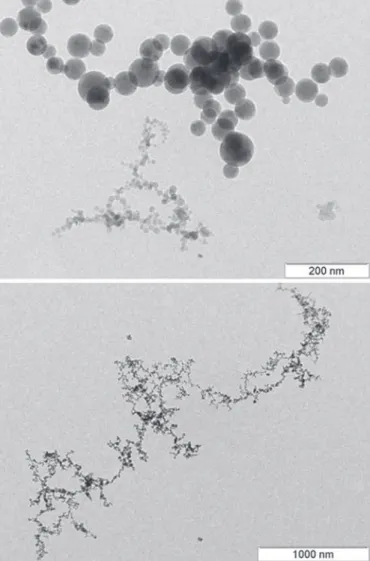
O complexo descrito como AgBNH foi caracterizado por análises elementar C,H,N vide Tabela 3 e térmica TG/DTA, espectroscopias IV, Raman e de Ressonância Magnética Nuclear de 1H e de
Envolver o sector privado é cada vez mais fulcral no sucesso da habitação, quer seja ao nível do arrendamento, da aquisição, da reabilitação ou da aquisição de imóveis. Assim,
The debate about the spatial dispersion of the population has become recurrent in recent ye- ars, due in part to the high explanatory power and the consequences inherent to
The aim of the present study was to evaluate the effects of different aeration frequencies on biomass temperature and gas emissions during composting of pig and poultry carcasses
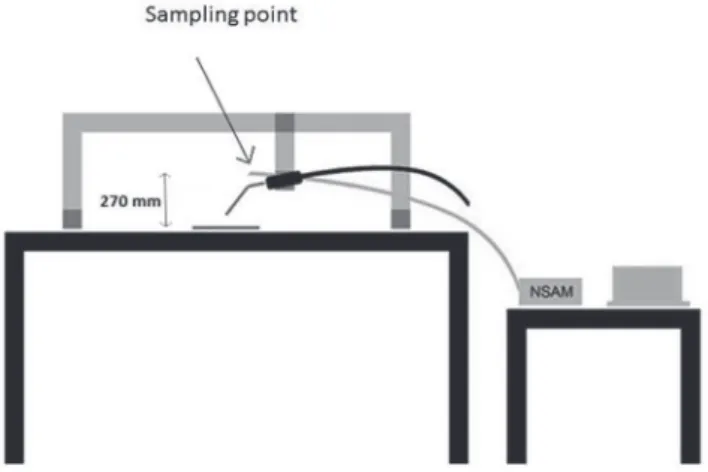
To control the gas pressure inside the gap a cryopump was designed and coupled to the cold block (Section 4.4 ) through a stainless steel capillary,
Na presente empreitada, temos como exemplo a utilização de micro-estacas na estrutura das antecâmaras de ligação ao aeroporto e em coberturas metálicas de
Como a presença de alelos nulos nos locos pode causar uma superes- timação nas estimativas de divergência genética (CHAPUIS; ESTOUP, 2007), recalculou-se o G’ ST com apenas