w w w . j c o l . o r g . b r
Journal
of
Coloproctology
Original
Article
Influence
of
the
application
of
enemas
with
oily
extract
of
curcumin
(
Curcuma
longa
)
on
the
tissue
content
of
neutral
and
acidic
mucins
in
the
colonic
mucosa
without
fecal
stream
夽
Carlos
Augusto
Real
Martinez
a,b,∗,
Vitor
Piquera
de
Oliveira
a,
Fabio
Guilherme
Campos
c,
Danilo
Toshio
Kanno
a,
Oscar
Orlando
Araya
Fernandez
a,
Caled
Jaoudat
Kadri
a,
José
Aires
Pereira
daUniversidadeSãoFrancisco(USF),ProgramadePós-Graduac¸ãoemCiênciasdaSaúde,Braganc¸aPaulista,SP,Brazil
bUniversidadeEstadualdeCampinas(UNICAMP),DepartamentodeCirurgia,Campinas,SP,Brazil
cUniversidadedeSãoPaulo(USP),FaculdadedeMedicina,SãoPaulo,SP,Brazil
dUniversidadeSãoFrancisco(USF),DisciplinadePatologia,Braganc¸aPaulista,SP,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received29August2017 Accepted30August2017
Availableonline22September2017
Keywords:
Colitis Mucins
Fattyacids,volatile Imageprocessing, computer-assisted
a
b
s
t
r
a
c
t
Purpose:Toverifyiftheapplicationofenemascontainingoilyextractsofcurcuminpreserves
thetissuecontentofmucinsintheglandsofthecolonicmucosawithoutfecalstream.
Method:Thirty-sixWistarratsweresubmittedtodiversionofthefecalstreambyproximal
colostomyanddistalmucousfistula.Theanimalsweresubdividedintothreegroups,and accordinglyreceivedenemaswithsalineandoilyextractofcurcuminatconcentrationsof 50mg/kg/dayor200mg/kg/day.Aftertwoorfourweeksofintervention,theirrigatedcolic segmentswereremoved.NeutralandacidicmucinswereidentifiedbyPeriodic-acidSchiff andAlcian-Bluetechniques,respectively.Thecontentofbothmucinsubtypeswas mea-suredbycomputerizedmorphometry.Mann–Whitneytestwasusedtoanalyzetheresults, adoptingasignificancelevelof5%(p≤0.05).
Results:Therewasanincreaseinthetissuecontentofneutralmucinsinanimalstreated
withcurcuminataconcentrationof50mg/kg/dayforfourweeks,whereasinthegroup treatedwith200mg/kg/daytherewasanincreaseindependentofthetimeofintervention. Thecontentofacidicmucinsincreasedinanimalstreatedwith200mg/kg/dayregardlessof theinterventiontime,whereasinthosetreatedwith50mg/kg/dayanincreasewasobserved onlyafterfourweeks.
夽StudyconductedatUniversidadeSãoFrancisco(USF),ProgramadePós-Graduac¸ãoemCiênciasdaSaúde,LaboratóriodeInvestigac¸ão Médica(LIM),Braganc¸aPaulista,SP,Brazil.
∗
Correspondingauthor.
E-mail:[email protected](C.A.Martinez).
https://doi.org/10.1016/j.jcol.2017.08.002
Conclusion: Enemaswithcurcuminpreservethecontentofneutralandacidicmucinsinthe colonicepitheliumwithoutfecalstream.
©2017SociedadeBrasileiradeColoproctologia.PublishedbyElsevierEditoraLtda.Thisis anopenaccessarticleundertheCCBY-NC-NDlicense(http://creativecommons.org/ licenses/by-nc-nd/4.0/).
Influência
da
aplicac¸ão
de
clisteres
contendo
extrato
oleoso
de
curcumina
(
Curcuma
longa
)
no
conteúdo
tecidual
de
mucinas
neutras
e
ácidas
na
mucosa
cólica
sem
trânsito
intestinal
Palavras-chave:
Colite Mucinas
Ácidosgraxosdecadeiacurta, voláteis
Análisedeimagemassistidapor computador
r
e
s
u
m
o
Objetivo: Verificarseaaplicac¸ãodeclisterescomextratooleosodecurcuminapreservao
conteúdodemucinasnasglândulasdamucosacólicasemtrânsitointestinal.
Método: TrintaeseisratosWistarforamsubmetidosàderivac¸ãointestinalpor
colosto-mia proximale fístula mucosadistal. Osanimais foram subdivididosem trêsgrupos, segundo receberem clisteres com soro fisiológico 0,9%, extrato oleoso de curcumina nasconcentrac¸õesde50mg/kg/diaou200mg/kg/dia.Apósduasouquatro semanasde intervenc¸ãoforamremovidosossegmentoscólicosirrigados.Asmucinasneutraseácidas foramidentificadaspelastécnicasdoPASeAlcian-Blue,respectivamente.Oconteúdo tecid-ualdeambosossubtiposdemucinasfoimensuradopormorfometriacomputadorizada. Utilizou-setestedeMann-Whitneyparaanálisedosresultadosadotando-senívelde sig-nificânciade5%(p≤0,05).
Resultados: Houveaumentonoconteúdodemucinasneutrasnosanimaistratadoscom
curcuminanaconcentrac¸ãode50mg/kg/diaporquatrosemanas,enquantonostratados com200mg/kg/diahouveaumentoindependentedotempodeintervenc¸ão.Oconteúdode mucinasácidasaumentounosanimaistratadoscom200mg/kg/diaindependentedotempo deintervenc¸ão,enquantonostratadoscom50mg/kg/diaencontrou-seaumentoapenas apósquatrosemanas.
Conclusão: Clisterescomcurcuminapreservamoconteúdodemucinasneutraseácidasno
epitéliocólicosemtrânsitointestinal.
©2017SociedadeBrasileiradeColoproctologia.PublicadoporElsevierEditoraLtda.Este ´eumartigoOpenAccesssobumalicenc¸aCCBY-NC-ND(http://creativecommons.org/ licenses/by-nc-nd/4.0/).
Introduction
Themucuslayerthatcoversthecolicepitheliumrepresents thefirstlineofdefenseagainsttheaggressioncausedby anti-gens,virus,andbacteriaintheintestinallumen.1,2 Mucins,
the main constituents of the mucus layer, are glycopro-teinssynthesized bygobletcells.3,4 Themucin molecule is
formedby a glycidicfraction and byanother protein frac-tion.Dependingon thecomponentspresent inthe glycidic portion, they are subdivided into neutral mucins rich in glycogen,andintoacidicmucinsrichinsialicacid.1,4Acidic
mucins,ontheotherhand,aresubdividedintosulfomucines when there is a predominance ofthe sulfate anion in its molecule,orintosialomucinswhensialicacidisthe predom-inantcarbohydrate.1,4,5 Inadditiontoprovidingmechanical
protection, the mucins have a lubricatingaction, facilitat-ingthe progressionoffecalcontentsthroughoutthecolon. Mucinsslowthediffusionofhydrogenions,inhibitthe activa-tionofpepsinogen,andexertanantibacterialactionagainst pathogenicbacteriathatoccurinsidethecolon.Inthisway, mucinsareconsideredasthefirstlineofdefenseofthecolonic epithelium.6Shortchainfattyacids(SCFA)representthemain
energysubstrateforgobletcellstoproduceadequateamounts ofmucus,inordertomaintaintheintegrityofthisfirstline ofmechanicaldefense.7,8 Deficienciesintheregular supply
ofSCFAdecreasetheexpressionofthosegenesresponsible for the translation of the protein fractionof the glycopro-tein molecule.2,7 Important changes in the constitutionof
mucinsinbacterialcolitis,inflammatoryboweldisease(IBD), adenomatous polyps, colorectal cancerand, morerecently, in diversion colitis (DC) have been described.1,4,7–9 Studies
usingexperimentalmodelsofDChaveshownthatthereis areductioninthecontentofneutralmucins,acidicmucins, sulfomucines,andsialomucinsinepithelialglandsdevoidof thenormalsupplyofSCFA.1,4,5Itwasalsodemonstratedthat
inexperimentalDC,thereductioninthecontentofthe differ-enttypesofmucinsisdirectlyrelatedtothehigherlevelsof tissueoxidativestress,duetothegreaterproductionof reac-tiveoxygenspecies(ROS)fromthecolonicmucosalepithelial cellsdevoidofaregularsupplyofSCFA.4,10Consideringthese
DC confirmedthis possibility byshowingthat the applica-tion ofenemas withdifferent antioxidant substances was abletoreducethecolonicmucosal damagecausedbyROS, decreasethetissueinflammatoryinfiltrate,andincreasethe mucin content in the colic epithelium devoid ofintestinal transit.11–14Curcuministhemainactiveingredientobtained
from the Curcuma longa rhizome extract.15 This natural
flavonoidhasimportantantioxidant,anti-inflammatory,and anti-neoplasticactivity.15,16 These properties have resulted
inanincreasinguse ofthis substanceindifferentdiseases whose common denominator is the presence of oxidative stressandinflammation.16Ithasalreadybeendemonstrated
thattheadministrationofcurcuminbringspromisingresults asacomplementarytherapyinpatientswithIBD.17
Experi-mentalstudieshaveshownthatthepreventiveapplicationof curcumin-containingenemashasimprovedmucosal inflam-mationandpreservedsulfomucineandsialomucinecontents and alsothe mainconstituent proteinsofthe intercellular occlusionjunctionsinanexperimentalmodelofDC.18–20
Ulti-mately,thesefindingssuggestthattheantioxidantactionof thesubstanceisabletomaintaintheintegrityofthemucus barrierofthecolonicepitheliumagainstthedeleteriousaction ofROS.
Although the effects of curcumin have already been demonstratedinDCmodels,todate,nostudyhasevaluated thesubstance’sabilitytopreservethecontentandexpression patternofneutralandacidicmucinsinexperimentalmodels ofEC.Thus,theaimofthepresentstudywastoevaluatethe efficacyofcurcumin-containingenemasinthereductionof theinflammatoryprocessandinthemaintenanceofthe neu-tralandacidicmucincontentintheglandsofthecolononic mucosaexcludedfromintestinaltransit.
Materials
and
methods
ThisstudyisinaccordancewiththeguidelinesoftheColégio BrasileirodeExperimentac¸ãoAnimal(COBEA).TheResearch ProjectwasapprovedbytheComitêdeÉticanoUsode Ani-malemPesquisa(CEUA)oftheUniversidadeSãoFrancisco, Braganc¸aPaulista,SãoPaulo(No.002.04.10).
Experimentalanimal
Thirty-sixmaleWistarrats(Rattusnorvegicusalbinus)from Ani-lab,Animais de Experimentac¸ão Criac¸ão eComércio Ltda., Paulínia,SãoPaulo, Brazil,were used. Theanimals’ weight ranged from 300 to 320g and on average they aged four months.
Experimentalgroups
Threegroupswerestudiedwith12animalseach,dividedinto thedailyapplicationofenemaswith0.9%physiological solu-tion(PS)andofcurcuminoilyextract(SigmaAldrichdoBrasil Ltda.,SãoPaulo,Brazil)atconcentrationsof50mg/kg/dayand 200mg/kg/day,respectively.Sixanimalsfromeachgroupwere submittedtoeuthanasiaaftertwoweeksofintervention;the remaininganimalsweresacrificedafterfourweeks.
Surgicalprocedure
Onthedayoftheintervention,theanimalswereweighedand anesthetizedwith2%xylazinehydrochloride(Anasedan PET-CevaSaúdeAnimalLtda,Paulínia,SãoPaulo)andketamine hydrochloride(DopalenPET-CevaSaúdeAnimalLtda, Paulí-nia)atadoseof0.1mL/100gintramuscularly.Then,therats wereplacedonasurgicalboard,inahorizontaldorsal decu-bitus position for trichotomy followed by antisepsis. The laparotomy was performedbymeans ofamedian incision with3cmofextensionand,afteracavityinventory,thePeyer’s patchatthetransitionbetweentherectumandthesigmoid wasidentified.Withtheaidofapachymeter,theleftcolonwas sectioned6cmabovethepatch.Theproximalcolonwas exter-nalizedasaterminalcolostomyinthelefthypochondrium. Afterthepreparationoftheproximalstoma,thecaudal seg-ment ofthesectioned colonwascatheterizedandirrigated with40mLofsalineheatedat37◦Cuntiltheeffluentdrained
throughtheanusoftheanimalshowedabsenceofstool out-put.Aftercleansing,thecatheterwasremovedandthedistal colonexternalizedintheformofamucousfistulaintheleft iliacfossa.Theabdominalwallsynthesiswasmadebyplanes. Attheendoftheoperation,theanimalswerekeptwarm and,afterreturningtoawakingstate,werehousedin individ-ualcages,withwaterintakeandstandardizedration(Nuvilab
CR1®)adlibitum.Theratswereisolateduntileuthanasiatime
(twoor fourweeks),inthesame environmentalconditions ofhumidity,luminosityandtemperatureofthepreoperative period.Noadditionalcarewastakenwithrespecttooperative woundorstomata;theratsreceivedonlyoneanalgesic admin-isteredinthedrinkingwaterinthefirstthreedaysafterthe operation(DipironaPET,BiovetLtda,VargemGrandePaulista, SãoPaulo).
Interventionwithproposedsolutions
Theanimalsweresubmitteddailytotheapplicationof ene-maswiththeproposedsolutions,accordingtothegroupto whichtheybelonged.Theuseofthesubstanceswasalways performedusingapolyethylenecatheter14F(internal diam-eter)graduatedincentimeters.Thecatheterwasintroduced bytheanimal’sanusatastandarddepthof2cmfromtheanal border.Next,theenemacontainingtheinterventionsolution proposedforeachexperimentalgroupwasapplieduntilthe solutiondrainedthroughthedistalmucousfistula.The inter-ventionsolutionswereappliedmanuallyin30swiththeaid ofasyringe.
Collectionofmaterial
removal, the animals were sacrificed with an intracardiac infusionofalethaldoseofthiopental.
Histologicaltechniques
Theremovedcolicsegmentwasopenedthroughthe antime-senterial border, and washed with saline to remove fecal residues.Alongitudinalsegmentoftheexcludedcolonfrom eachanimal,measuring30mminlength,wasfixedonacork piecesothatthefragmentwasstretchedandthenimmersed in 10% buffered formaldehyde solution for72h. Once this periodhaselapsed,thespecimensweredehydratedin alco-holanddiaphanizedinxylene.Thematerialwasembedded inparaffinblocks.Ineachblockthree4-mthicklongitudinal sliceswereobtainedperpendicularlytothemucosalsurface forassemblyoftheslides.
Thefirstslidewasstainedbythehematoxylin–eosin(HE) techniquetoevaluatethehistologicalchangesfound (pres-enceofcolitis).ThesecondslidewasstainedbythePeriodic Acid-Schiff(PAS)histologictechnique,withtheaimof identi-fyingthe neutralmucins,whilethethirdslidewasstained with the Alcian Blue (AB) technique to identify the acidic mucins.Withthetechniquesused,theneutralmucinswere stainedinmagenta,whiletheacidicmucinsstainedblue.
All the slides were analyzed by the same pathologist withexperience in IBD who was unaware ofthe origin of thematerial. For the study oftheslides, anoptical micro-scope(EclipseDS-50i,NikonInc.,Osaka,Japan)wasused.The imagesobtainedateachselectedfocalfieldwerecapturedbya camera(DS-Fi-50;NikonInc,Osaka,Japan)previouslyattached tothemicroscopeandthendigitizedandstoredforlater com-puterizedmorphometricanalysis.
Thediagnosis ofDC was considered in the face ofthe followingfindings:lossofepithelialsurface;thepresenceof inflammatoryinfiltrates,andthepresenceofmucosaledema. Foreachvariableavalue(incrosses)wasassignedthatvaried fromzeroto3crosses:(0=absent,1=slight,2=moderate,and 3=severe).Foreachanimal,threedifferenthistologicalfields wereread.Themedianobtainedbythesumofthevariables foundineachanimalafterthereadingsofthethreedistinct fieldsresultedintheinflammatorygraduationscore.
Quantificationofmucincontentbycomputerassisted imageanalysis
Quantification of tissue content of mucin subtypes was doneusingacomputer-aidedimageprocessingsystem (NIS-Elements, version 3.0, Nikon Inc., Japan). The program analyzedtheimagespreviouslystoredinthesystem.Ineach image,afterselectingthehistologicalfieldtobeanalyzed,the programdeterminedtheentirespectrumofthechosencolor bycolorhistogramswiththeuseofaRGB(red,green,blue) system.Afterquantifyingthecontentoftheacidic mucins, alwaysstained inablue shade, theprogramautomatically transformedeveryspectrumofthebluecolorinapercentage ofpixels,withthecalculationofthenumberofpixelsineach histologicalfieldanalyzed.Likewise,todeterminethecontent ofthe neutralmucins stainedinmagentabythePAS tech-nique,theprogramidentifiedtheentirespectrumofcolors containingthe magenta shade.The evaluationofthe
con-tentsofeachsubtypeofmucinwasalwayscarriedout ina focalfieldinwhichitwaspossibletoidentifythree contigu-ousandintactcolonicglands.Thefinalvalueconsideredfor thecontentofneutralandacidicmucinsineachanimalwas the averageobtainedafterreading threedifferent histolog-icalfields.Thefinalcontentwasalways theaverageofthe threereadings,beingexpressedinpercentagevaluesperfield (%/field).
Statisticalanalysis
Theresultsobtainedwereanalyzedbytheapplicationofthe BiostatProgram,version5.0.Thevaluesfortheinflammatory scoreswere expressedas amedian.For the comparisonof the inflammatory scores found in the three experimental groups, themedian testwasapplied.Significant valuesfor theinflammatoryscorewerecompared,withtheassignment ofonetriangle()whenp<0.05%,or oftwotriangles() whenp<0.01%.Thevaluesfoundinthecontent ofneutral or acidic mucins in each experimentalgroup were always expressedbymeanvalues,withtherespectivestandarderror. Thecomparisonoftheneutraland acidicmucins contents among the three experimental groups was also done in a paired form (i.e., by comparing two groups at a time) by the use of the Mann–Withney test. The significance level of5%(p≤0.05)wasadopted.Significant valuesforthe con-tentofbothmucinsubtypeswerefoundinthecomparison between the animals submitted to the intervention with curcumin (independent of the concentration used) versus
animals submitted tointerventionwith saline.The signifi-cantvaluesfoundforthecontentsofbothmucinsubtypes, when comparedtothe animalssubmitted tothe interven-tion with both concentrations of curcumin (50mg/kg/day and 200mg/kg/day) were always identified with a cross(†).
Results
Fig.1Ashowsthemucosal surfaceobtainedfromthecolon treated with PS for two weeks, while Fig. 1B shows the excludedcolonsubmittedtotheinterventionwithcurcumin oilyextractataconcentrationof200mg/kg/dayforthesame periodoftime.
Fig.2Ashowsthedistributionoftheneutralmucinsinthe animalssubmittedtosalineinterventionfortwoweeks,while
Fig.2Bshowsthedistributionoftheneutralmucinsafterthe interventionwithcurcuminoilyextractataconcentrationof 200mg/kg/dayforthesameperiodoftime.
Fig.3Ashowsthedistributionpatternofacidicmucinsin animals submittedtoPSinterventionfortwoweeks,while
Fig.3Bshowsthedistributionofacidicmucinsinthecolonic mucosa submitted to the intervention with curcumin oily extractattheconcentrationof200mg/kg/day.
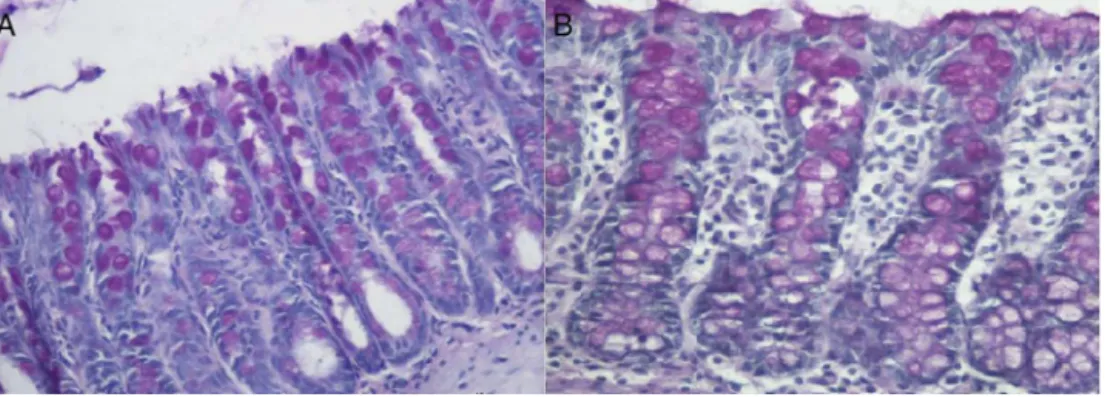
Fig.1–(A)ColonicmucosadevoidoffecalstreamofanimalsubmittedtotheinterventionwithPSforfourweeks.Notethe irregularityofthemucosalsurface,tortuosityofcrypts,inflammatoryinfiltrate,andedema;(B)colonicmucosadevoidof fecalstreaminananimalsubmittedtointerventionwithcurcuminataconcentrationof200mg/kg/dayforfourweeks.The epithelialsurfaceisshowntointegratethealignedcoloniccrypts,withlessedemabetweenthecolicglands(HE:200×).
Fig.2–(A)Tissueexpressionofneutralmucinsinthecolonicmucosadevoidoffecalstreamofananimalsubmittedtothe salineinterventionfortwoweeks.Notetheatrophyofcryptsandthereductionoftheneutralmucincontentingobletcells; (B)tissueexpressionofneutralmucinsinthecolonicmucosadevoidoffecalstreaminananimalsubmittedtointervention withcurcuminataconcentrationof50mg/kg/dayfortwoweeks.Wecanobserveahighercontentofneutralmucinsinthe colicglands(PAS:200×).
Table1–Inflammatoryscoreandtissuecontentofneutralandacidicmucinsinanimalssubmittedtointerventionwith PSandcurcuminatdosagesof50mg/kg/dayand200mg/kg/dayaftertwoandfourweeks.
Physiologicalsolution Curcumin50mg/kg/day Curcumin200mg/kg/day
2w/o 4w/o 2w/o 4w/o 2w/o 4w/o
Score 2a 3a 2 1 1 0
Neutral 7.3±0.3 4.3±0.2 5.7±0.1 5.7±0.2b 7.9±0.3b,c 8.5±0.4b,c
Acidic 9.1±0.3 6.1±0.2 10.2±0.6b 12.0±0.3b 11.8±0.2b,c 13.7±0.4b,c
a Physiologicalsolution>curcumin50mg/kg/dayor200mg/kg/day.
b Curcumin50mg/kg/dayorcurcumin200mg/kg/day>physiologicalsolution.
c Curcumin200mg/kg/day>curcumin50mg/kg/day.
Discussion
Thecolicepithelium constitutesthe mostperfect morpho-functionalbarrierinlivingbeings.Consistingofasinglelayer ofpolarizedcellsandfirmlyadheredtoeachother,thecolonic epitheliumseparatestheintestinalcontent,richinbacteria andantigens,fromthesterileinternalenvironment.Thiskey mechanismisformedbyseverallinesofdefense,represented mainlybythegelatinouslayerofmucusthatcoversthe intesti-nalepithelium,bytheintricatemechanismsofintercellular junction,andbythebasalmembrane.11Itisalsoworth
men-tioningthepresenceofasophisticatedimmunesystem by differentdefensecells andbyantibodies.Themaintenance ofthiswholedefensesystemdependsontheadequate sup-plyofanenergeticsubstrate tothespecialized cellsofthe intestinalepithelium,sothattheycansynthesizetheproteins constitutingthedifferentstructuresthatmakeupthemucosal barrier.20,19
SCFAconstitutethemostimportantenergysubstratefor thecellsofthecolicepithelium.Thesesubstances are pro-ducedfromthefermentationofundigestedcarbohydratesand polysaccharidesfrom thedietbyanaerobicmicroorganisms thatarecomponentsofthemicrobiotapresentinthecolon.21
Thankstotheirhighvolatility,thesesubstancesarerapidly absorbedbytheintestinalepithelium;itisestimatedthatof thetotalproduced,lessthan10%arepresentinthefeces.22
Acetic,propionic and butyric acids are the main subtypes formedandareestimatedtocorrespondtomorethan95% ofallSCFApresentinthegastrointestinaltract.23
Theincreasing interest inSCFA isrelated to aincrease in scientific evidence regarding its beneficial physiological effectson the organism.24 Amongtheirvarious properties,
it has been demonstrated that SCFA, in particular butyric acid,representthemainsubstrateusedbycellsofthecolonic epithelium toproduce all the energynecessary to the dif-ferent stages of cellular metabolism dependent on energy expenditure.Amongthesestages,thegenetranscriptionand synthesisofthedifferentproteinspresentincellsstandout.25
Inthecolon,-oxidationofbutyricacidisresponsibleforthe consumptionofabout70%ofallavailableoxygen;ithasbeen shownthatthisisthemainfuelusedbythecellsofthecolonic mucosa,forexampleforproteinsynthesisofmucins.1
Stud-ieshavealsoshownthatSCFAreducethelevelsofoxidative stressinthecolicepitheliumbecause,besidesreducingROS production,thesesubstancesareableofstimulatingthe activ-ityoftheantioxidantsystemspresentinthetissues.26Onthe
otherhand,thelackofaSCFAsupply,asoccursintheexcluded colon,increasesROSproduction,causingdamagetothe dif-ferentdefensesystemsthatformthecolicepithelialbarrier. Oxidativestresshasbeenconsideredaconvincingmolecular explanationfortheonsetofDC.10,27
Anumberofexperimentalstudieshaveshownthat impor-tantmodificationsoccurinthecontentandexpressionpattern of the glicidic fractions of mucins in goblet cells present in colonic mucosal glands devoid ofthe regular supply of SCFA.1,4,5 Initially, theseauthors demonstrated that, in the
colonicmucosadevoidoffecalstream,thecontentofneutral andacidicmucinswasdepleted,incontrasttowhatoccurred inthemucosawithapreservedtransit.1Anditwasshowed
thatthismucindepletionwasalsomaintainedwiththe pro-gressionoftheexclusiontime.Theauthorshypothesizedthat thedeficiencyinSCFAsupply,bymodifyingthemitochondrial energeticmetabolismtoobtainenergy,wouldincreaseROS production,andthattheresultingoxidativestresswouldbe themainresponsibleforreducingthecontentoftheglicidic fractionsofthemucinmolecules.1Thesamegroupconfirmed
thepreviouslydescribedresults,showingthatthereductionin thetotalcontentoftheacidicmucinsoccurredmainlybythe reductionofthesialomucins,whichpracticallydisappeared after18weeksofintestinalexclusion.5Inordertoverifyifthe
oxidativestresscouldberelatedtothereductionofthe con-tentofthesemolecules,inasubsequentstudyinwhichthe sameanimalswereusedtheauthorsdemonstratedthe occur-renceofaprogressiveincreaseofthelevelsofROSwiththe courseofthetimeofexclusion.IncreasedROSproductionwas relatednotonlytoanincreasedepithelialdamageinthose segmentsdevoidoffecaltransit,butalsotothedepletionof thecontentofallsubtypesofmucin.10
TheimportanceofaregularsupplyofSCFAinthesynthesis ofmucinswasconfirmedbystudiesthatanalyzedthetissue expressionofseveralgenesresponsibleforthetranscription oftheproteinfractionsofmucin.8 Theresultsshowedthat
butyrate stimulatesthe expressionofMUC1, MUC2, MUC3,
MUC4,andMUC5B,increasinginsixtimestheexpressionof
MUC2,themaingenerelatedtomucin synthesisincolonic epithelium.8Thesefindingsconfirmtherelevanceofan
ade-quatesupplyofSCFA,inparticularbutyrate,foranadequate synthesisofmucins.
cytotoxicradicalsareresponsible forthedestruction ofthe mucuslayerthatmakesuptheepithelialbarrierofthecolonic mucosa.Thus,theuseofsubstanceswithantioxidantactivity becomesaninterestingstrategytoreversethemucosallayer damagecausedbyROSincasesofDC.Anumberof experimen-talstudiesseemtoconfirmthispossibility.Substanceswith antioxidantaction,suchasn-acetylcysteineandmesalazine wereabletoreverseoxidativestressandimprovethe inflam-matoryprocessinexperimentalmodelsofDC.13,14 However,
theevaluationoftheefficacyoftheuseofantioxidantsinthe maintenanceofthemucincontentintheepitheliumexcluded fromfecaltransitwasonlystudiedlater,whentwostudies evaluatedtheeffectsofsucralfateapplicationinan experi-mentalmodelofDC.28,29Thefirststudyevaluatedtheeffects
oftheapplicationofenemaswithsucralfate,asubstancewith recognizedantioxidantactivityandwhichcanstimulatethe productionofmucusbytheepitheliumofthegastrointestinal tract.Theresultsshowedthatsucralfate,besidesimproving theinflammatoryprocess,wasabletoincreasethetissue con-tentofneutralandacidicmucinsintheepitheliumexcluded fromfecaltransit.28Soonafter,asecondstudymeasuredthe
tissue content of acidic mucin subtypes (sulfomucins and sialomucins)in thesesame animals.29 Theresultsshowed
thattheuse ofsucralfate-containingenemas increasedthe totaltissuecontentoftheacidicmucins,mainlybecausethis substance increasesthe content ofsialomucins –precisely thesubtypeofacidic mucin that presentsa greater reduc-tionofitscontentintheepitheliumdevoidoffecaltransit.4,5
Recently,inagreementwiththefindingsofthesetwo stud-ies,ithasbeendemonstratedthattheuseofsucralfatemay alsoableincreasethetissuecontentoftheMUC2proteinin colonicmucosadevoidofintestinaltransitinan experimen-talmodelofDC.30However,themechanismthatexplainshow
thishappensisstillwaitingforanexplanation.Itispossible thatsucralfatemayexertanantioxidanteffect,protectingthe mucosafromoxidativestress;however, onecannotdismiss thepossibilitythatthesubstancemaystimulatethe transcrip-tionoftheMUC2gene–therebyincreasingthecontentofthe correspondingproteinfraction.
Allthesefindingshaveshownthatthereductionof oxida-tivestress levelsinthe mucosaexcludedfrom fecaltransit throughtheuseofantioxidantsubstancesiseffectiveinthe treatmentofDC.Thus,theuseofcurcumin,apotentnatural antioxidantwiththerapeuticefficacyinthetreatmentofIBD –andespeciallywhenthissubstanceisassociatedwithother drugs–could becomeavalidstrategyforthe treatmentof DC.17Curcumin,inadditiontoitsantioxidanteffect,inhibits
theinflammatorypathwaysbyreducingtheactivityofIL-1 andCOXandmodulatingtheAKT/mTORpathwaythat con-trolscellenergymechanisms.18Onestudyprovedthattheuse
ofacombinationtherapywithcurcuminandmesalaminewas showntobesuperiorversustheisolateduseofmesalaminein thetreatmentofpatientswithIBD.18Althoughthereisclinical
evidencethatcurcumincanincreasetheproductionofmucins inthecolonicepithelium,asinglestudymeasuredthetissue contentofsulfomucinsandsialomucinsinthecolonexcluded from fecalstreaminan experimentalmodelofDC20. The
authorsobservedthattheapplicationofenemascontaining curcuminoilyextractnotonlyimprovedthemucosal inflam-matoryprocess,butalsoincreasedthecontentofbothacidic
mucinsubtypes.Thestudyalsoshowedthatthisincreasein sulfomucineandsialomucinecontentwasrelatedtothe con-centrationusedandtothetimeofintervention.20
Theresultsofthepresentstudyconfirmtheeffectiveness oftheapplicationofenemaswithcurcuminincecalsegments deprived ofintestinal transit. We observed the occurrence ofreductionoftheinflammatoryscoreintheanimals sub-mittedtotheinterventionwithcurcumin versusanimalsof the controlgroup, independent of the concentration used. Wealsoverifiedthattheneutralmucincontentinthecolon without transitofthose animalssubmittedtoPS interven-tiondecreasedwiththeprogressionoftheinterventiontime. On the other hand,inthe animals submitted tothe inter-ventionwithcurcumin,especiallyinthegroupstreatedwith the highestconcentration (200mg/kg/day)and fora longer periodoftime,thecontentofneutralmucinsincreased signif-icantly.Whenanalyzingthetissuecontentofacidicmucins, we also found a reduction of the content in the animals submittedtothe salineintervention,relatedtothetimeof exclusion.Conflictingwiththisfinding,theinterventionwith curcumin,regardlessoftheconcentrationused,increasedthe tissuecontentofacidicmucinsversusanimalssubmittedto salineintervention.Theincreaseinthetissuecontentofacidic mucins inthe animals submitted tothe interventionwith curcumin wasrelated to the use ofhigher concentrations, especiallywhenappliedforalongerinterventionalperiod.
Theresultsofthepresent studyconfirmthe findingsof previous studies, showing the benefits of the application ofenemas withcurcumin oily extractforthe treatmentof experimentalDC.Thereductionoftheinflammatoryprocess inthemucosadevoidoffecalstream,aswellastheincrease intheproductionofbothsubtypesofmucins,seemto con-firmtheefficacyofthissubstanceinthetreatmentofdifferent formsofcolitis.However,studiesinhumansarestillnecessary toconfirmtheviabilityofusingenemascontainingcurcumin forthetreatmentofthesediseases.
Funding
Fundac¸ão de Amparo à Pesquisa do Estado de São Paulo (FAPESP).CaseNumber2010/12492-7.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
r
e
f
e
r
e
n
c
e
s
1.NonoseR,SpadariAPP,PriolliDG,MáximoFR,PereiraJA, MartinezCAR.Tissuequantificationofneutralandacid mucinsinthemucosaofthecolonwithandwithoutfecal streaminrats.ActaCirBras.2009;24:267–75.
2.GaudierE,HoeblerC.Physiologicalroleofmucinsinthe colonicbarrierintegrity.GastroenterolClinBiol. 2006;30:965–74.
4. MartinezCAR,NonoseR,SpadariAPP,MáximoFR,PriolliDG, PereiraJA,etal.Quantificationbycomputerized
morphometryoftissuelevelsofsulfomucinsandsialomucins indiversioncolitisinrats.ActaCirBras.2010;25:231–40.
5. KeliE,BouchouchaM,DevroedeG,CarnotC,OhrantT, CugnencPH.Diversion-relatedexperimentalcolitisinrats. DisColonRectum.1997;40:222–8.
6. DeplanckeB,GaskinsHR.Microbialmodulationofinnate defense:goblet-cellsandtheintestinalmucuslayer.AmJClin Nutr.2001;73:1131S–41S.
7. WillemsenLE,KoetsierMA,VanDeventerSJ,VanTolEA.Short chainfattyacidsstimulateepithelialmucin2expression throughdifferentialeffectsonprostaglandinE1andE2 productionbyintestinalmyofibroblasts.Gut.2003;52:1442–7.
8. GaudierE,RivalM,BuisineMP,RobineauI,HoeblerC. ButyrateenemasupregulateMUCgenesexpressionbut decreaseadherentmucusthicknessinmicecolon.Physiol Res.2009;58:111–9.
9. JohanssonME,SjövallH,HanssonGC.Thegastrointestinal mucussysteminhealthanddisease.NatRevGastroenterol Hepatol.2013;10:352–61.
10.MartinezCAR,RibeiroML,GamberoA,MirandaDDC,Pereira JA,NadalSR.Theimportanceofoxygenfreeradicalsinthe etiopathogenesisofdiversioncolitisinrats.ActaCirBras. 2010;25:387–95.
11.PereiraJA,RodriguesMR,SatoDT,SilveiraJúniorPP,DiasAM, SilvaCG,etal.Evaluationofsucralfateenemainexperimental diversioncolitis.JColoproctol(RioJ).2013;33:182–90.
12.MartinezCAR,RodriguesMR,SatoDT,SilvaCMG,KannoDT, Mendonc¸aRLS,etal.Evaluationoftheanti-inflammatoryand antioxidanteffectsofthesucralfateindiversioncolitis.J Coloproctol.2015;35:90–9.
13.MartinezCA,deAlmeidaMG,daSilvaCM,RibeiroML,da CunhaFL,RodriguesMR,etal.Enemaswithn-acetylcysteine canreducethelevelofoxidativedamageincellsofthe colonicmucosadivertedfromthefaecalstream.DigDisSci. 2013;58:3452–9.
14.CaltabianoC,MáximoFR,SpadariAP,daConceic¸ãoMiranda DD,SerraMM,RibeiroML,etal.5-Aminosalicylicacid(5-ASA) canreducelevelsofoxidativeDNAdamageincellsofcolonic mucosawithandwithoutfecalstream.DigDisSci.
2011;56:1037–46.
15.AggarwalBB,YuanW,LiS,GuptaSC.Curcumin-freeturmeric exhibitsanti-inflammatoryandanticanceractivities: identificationofnovelcomponentsofturmeric.MolNutr FoodRes.2013;57:1529–42.
16.SamarghandianS,Azimi-NezhadM,FarkhondehT,SaminiF. Anti-oxidativeeffectsofcurcuminon
immobilization-inducedoxidativestressinratbrain,liver andkidney.BiomedPharmacother.2017;87:223–9.
17.LangA,SalomonN,WuJC,KopylovU,LahatA,Har-NoyO, etal.Curcuminincombinationwithmesalamineinduces
remissioninpatientswithmild-to-moderateulcerativecolitis inarandomizedcontrolledtrial.ClinGastroenterolHepatol. 2015;13:1444–9.
18.KadriCJ,PereiraJA,CamposFG,OrtegaMM,BragionCB, MartinezCA.Anti-inflammatoryeffectsofenemas containinganoilyextractofcurcumininanexperimental modelofdiversioncolitis.HistolHistopathol.2017;32:161–9.
19.MartinezCA,KadriCJ,KannoDT,AlvesJúniorAJJ,CoyCS, PereiraJA.Claudin-3andoccludincontentintheglandsof colonicmucosadevoidfromfecalstreamsubmittedtotopical interventionwithoilextractofCurcumalonga.ActaCirBras. 2017;32:65–73.
20.AlvesAJ,Júnior,PereiraJA,PansaniAH,MagroDO,CoyCS, etal.Tissuesulfomucinandsialomucincontentincolon mucosawithoutintestinaltransitsubjectedtointervention withCurcumalonga(curcumin).ActaCirBras.2017;32:182–93.
21.HenningssonAM,BjorckIM,NymanEM.Combinationsof indigestiblecarbohydratesaffectshort-chainfattyacid formationinthehindgutofrats.JNutr.2002;132:3098–104.
22.denBestenG,vanEunenK,GroenAK,VenemaK,Reijngoud DJ,BakkerBM.Theroleofshort-chainfattyacidsinthe interplaybetweendiet,gut,microbiota,andhostenergy metabolism.JLipidRes.2013;54:2325–40.
23.HuJ,LinS,ZhengB,CheungPC.Short-chainfattyacidsin controlofenergymetabolism.CritRevFoodSciNutr. 2016;27:1–7.
24.TanJ,McKenzieC,PotamitisM,ThorburnAN,MackayCR, MaciaL.Theroleofshort-chainfattyacidsinhealthand disease.AdvImmunol.2014;121:91–119.
25.AgarwalVP,SchimmelEM.Diversioncolitis:anutritional deficiencysyndrome?NutrRev.1989;47:257–61.
26.HamerHM,JonkersDM,BastA,VanhoutvinSA,FischerMA, KoddeA,etal.Butyratemodulatesoxidativestressinthe colonicmucosaofhealthyhumans.ClinNutr.2009;28:88–93.
27.MartinezCAR,FabrisFM,SilvaCMG,RodriguesMR,SatoDT, RibeiroML,etal.Oxidativestressandchangesinthecontent andpatternoftissueexpressionof-cateninproteinin diversioncolitis.JColoproctol.2012;32:343–58.
28.ChaimFM,SatoDT,RodriguesMR,DiasAM,SilveiraJúniorPP, PereiraJA,etal.Evaluationoftheapplicationofenemas containingsucralfateintissuecontentofneutralandacid mucinsinexperimentalmodelofdiversioncolitis.ActaCir Bras.2014;29:544–52.
29.BonassaCE,PereiraJA,CamposFG,RodriguesMR,SatoDT, ChaimFD,etal.Tissuecontentofsulfomucinsand sialomucinsinthecolonicmucosa,withoutfecalstream, undergoingdailyinterventionwithsucralfate.ActaCirBras. 2015;30:328–38.