revbrashematolhemoter.2015;37(6):414–416
w w w . r b h h . o r g
Revista
Brasileira
de
Hematologia
e
Hemoterapia
Brazilian
Journal
of
Hematology
and
Hemotherapy
Case
Report
Spurious
laboratory
alterations
in
pernicious
anemia
Adriana
Isabel
Dias
Roque
∗,
Cláudia
Nadine
da
Silva,
Joana
Maria
dos
Reis
Mata
Albuquerque
Martins,
Maria
Amélia
Fonseca
Pereira
HospitalDistritaldaFigueiradaFoz,FigueiradaFoz,Portugal
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received11November2014 Accepted15July2015
Availableonline17August2015
Introduction
Theauthorsreporta caseofperniciousanemia presenting withnormocytosis, “nonthyroidal illness syndrome” (NTIS) andelevatedcancerantigen15.3(CA15.3).
MegaloblasticanemiaiscausedbydefectsinDNA synthe-sisandtypicallypresentswithmacrocytosis.Oneofitscauses islowserumvitaminB12(cobalamin)levels,causedin20–50% ofthecasesbyadecreaseinintrinsicfactorduetoatrophyof thegastricmucosaandconsequentlylossofparietalcells− anautoimmuneconditionknownasperniciousanemia.1,2
CA15.3, aserum tumor marker, isa mucinous antigen of the Mucin 1 (MUC-1) glycoprotein that is expressed by epithelialcells(mainlybreastepithelialcells)anderythroid precursors (it isabsent inmatureerythrocytes).3 Its
eleva-tion ismainly associatedwithbreast adenocarcinoma, but it can alsobe foundin other cancersand insomebenign diseases.3–5
NTISisaconditioninwhichthereare abnormalthyroid functiontestsinapatientwithoutthyroiddiseaseduetoan
∗ Correspondingauthorat:HospitalDistritaldaFigueiradaFoz,Gala,3094-001FigueiradaFoz,Portugal. E-mailaddress:[email protected](A.I.D.Roque).
adaptiveresponseanadaptiveresponseinordertominimize tominimizethecatabolismrateduringhigh-stressperiods. Itusuallypresentswithdecreasedfreetriiodothyronine(FT3) andthyroid-stimulatinghormone(TSH)andnormalfree thy-roxine(FT4).6,7
Case
report
Herein,thecaseofa44-year-oldfemalepatientadmittedto the EmergencyRoomwithcomplaintsofastheniaand dys-pepsia forthree months is reported. There were no other associated symptoms, such as fever, weight loss or hem-orrhage. The patient denied alcohol, tobacco or substance abuse, regular medications,dietaryrestrictions, priorblood transfusionsoranyotherrelevantmedicalorsurgicalevent. Epidemiologicalandfamilydatawereirrelevant.
Onadmission,shewashemodynamicallystable,withpale mucosa and icteric sclera; the remaining physical exami-nation, includingcardiac and pulmonaryauscultation, and abdominalandcervicalpalpation,wasunremarkable.
http://dx.doi.org/10.1016/j.bjhh.2015.07.008
revbrashematolhemoter.2015;37(6):414–416
415
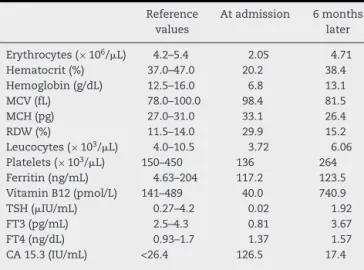
Table1–Evolutionofanalyticalparameters.
Reference values
Atadmission 6months later
Erythrocytes(×106/L) 4.2–5.4 2.05 4.71 Hematocrit(%) 37.0–47.0 20.2 38.4 Hemoglobin(g/dL) 12.5–16.0 6.8 13.1
MCV(fL) 78.0–100.0 98.4 81.5
MCH(pg) 27.0–31.0 33.1 26.4
RDW(%) 11.5–14.0 29.9 15.2
Leucocytes(×103/L) 4.0–10.5 3.72 6.06
Platelets(×103/L) 150–450 136 264 Ferritin(ng/mL) 4.63–204 117.2 123.5 VitaminB12(pmol/L) 141–489 40.0 740.9 TSH(IU/mL) 0.27–4.2 0.02 1.92
FT3(pg/mL) 2.5–4.3 0.81 3.67
FT4(ng/dL) 0.93–1.7 1.37 1.57
CA15.3(IU/mL) <26.4 126.5 17.4
MCV: mean corpuscular volume; MCH: mean corpuscular hemoglobin; RDW: red blood cell distribution width; TSH: thyroid-stimulating hormone; FT3: free triiodothyronine; FT4: freethyroxine;CA15.3:cancerantigen15.3.
The blood count revealed a normocytic normochromic anemia [hemoglobin (Hb): 6.8g/dL; mean corpuscular vol-ume (MCV):98.4fL], with reduced leukocyte (3.72×103/L), platelet (136×103/L) and reticulocyte (26.2×103/L;index 0.63%)counts.Theperipheralbloodsmearshowed anisopoik-ilocytosis,hypersegmentedneutrophilsandnoblasts.
The serum level of total vitamin B12 was markedly decreased (40.0pmol/L), with normal folic acid, and iron stores(ferritin:117.2ng/mL;erythrocytesedimentationrate: 10mm/h).
The patient had unconjugated hyperbilirubinemia, ele-vated lactate dehydrogenase (LDH) (4788IU/L; normal: 240–480), and low haptoglobin (0.3mg/dL; normal: 30–200). No changes in liver profile enzymes, kidney function or electrolyte balance were detected. The direct Coombs test wasnegative(Table1).
A bone marrow aspirate smear revealed hypercellular-ity, with hyperplasia and immaturity of erythroid lineage, megaloblastosis,giantmetamyelocytesandhypersegmented neutrophils,findingsconsistentwithmegaloblasticanemia.
FT3andTSHweredecreased,whileFT4and thyrotropin-releasing hormone (TRH) were normal. The investigations for infections by cytomegalovirus, Epstein Barr virus and parvovirusB19werenegative.Nuclear,anti-dsDNA,and anti-thyroidautoantibodieswerealsonegative.
Available tumor markers for the most probable tumors for gender and age (carcinoembryonic antigen, CA-125 and CA 15.3)were assessed, identifyingan increase in CA 15.3 (126.5U/mL; normal: <26.4). In order to exclude an occultmalignancy,low-radiation radiological studies(chest radiography,mammographyand thyroid,abdominal, pelvic and breast ultrasounds) were performed, that revealed no anomalies.
Gastric parietal cell antibodies were positive and anti-intrinsicfactorantibodieswerenegative.Gastricendoscopy and biopsies showed an atrophic pangastritis, without
Helicobacterpyloriinfection,corroboratingthediagnosisof per-niciousanemia.
Thepatient’spreviousbloodcountshadrevealedpersistent microcytosis(MCV80fL),Hb(11.5–12.5g/dL),andnormal fer-ritin.High-performanceliquidchromatography(HPLC)ofHb wasnormal(HbA2:2.9%;HbF:0.3%)excluding-thalassemia. WedidnotperformotherHbstudies,namely␣-thalassemia. Thepatientreceived2000gofintramuscular cyanocobal-aminplustwounitsofpackedredbloodcells(duetomarked complaints of tiredness) and was referred to the Internal MedicineOutpatientClinic,withmonthlyadministrationsof 1000gofcobalamin.
Atthecontrolappointment,sixmonthsafterthe begin-ningofthetreatment,acompleteresolutionofsymptomshad occurred,andlaboratoryabnormalitieshadnormalized. How-ever,MCVandmeancorpuscularhemoglobindecreasedtothe lowerlimit,whichsuggestsaconcomitantcauseof microcy-tosis(Table1).
Discussion
Although the diagnosis ofmegaloblastic anemia isusually easytosuspectafterreviewingthecompletebloodcountand peripheralbloodsmear,theabsenceofmacrocytosis,usually due to a concomitant cause ofmicrocytosis (such as tha-lassemiaorirondeficiency),coulddelaythediagnosis.1,8,9
In this case, ineffective erythropoiesis was suspected duetothe unconjugatedhyperbilirubinemia,elevated LDH, anddecreasedhaptoglobin;thenegativedirectCoombstest excludedanautoimmuneetiologyforthishemolyticanemia. LowvitaminB12levels,positivityforgastricparietalcell anti-bodiesandstomachbiopsyresults(whichweonlyassessed later) corroborated the hypothesis of perniciousanemia.1,2
Becauseofaconstitutionalhistoryofmicrocytosiswith nor-malironstores,anHbdisorder(␣-thalassemiaorHbvariants not assessed by HPLC after excluding -thalassemia) was highlysuspect.Meanwhile,becausethesetestsarenot avail-able in ourinstitution and as they would notsignificantly affectthetherapeuticapproach,theywerenotperformed.In many casesofhypoproliferativeanemiaand pancytopenia, it isalsoessential toexcludehematopoieticdiseases,bone marrowinfiltrationandinflammatorycauses.1
Perniciousanemiaismostfrequentinpeoplewithother autoimmune diseases, mainly thyroid, with anti-thyroid antibody positivityin about half ofpatients. So, the onset of autoimmune diseases should be monitored during the follow-up.1,2
Inthiscase,theinitialstudyofthyroidfunctionrevealed changes inthe thyroid hormone profile.However, asthere wereonlyslightdecreasesinTSHandFT3,theFT4andTRH werenormal,anti-thyroidantibodieswerenegativeandthere werenochangesinthyroidultrasound,adiagnosisofNTIS wasestablished.6,7Althoughthisentityisusuallyassociated
416
revbrashematolhemoter.2015;37(6):414–416thiscase,theTSHlevelnormalizedaftertheacutestress,being concordantwithanadaptiveresponse,highlightingthelack ofusefulnessofassessingthyroidhormonesinstress.
CA15.3isanimmunodominantepitopeinthe extracellu-larportionofthemembraneboundmucin.Themucinprotein, encodedbytheMUC1gene,isacellsurfacetransmembrane glycoprotein,expressedattheapicalsurfaceofmostepithelia (suchasthemammarygland)innormaltissue,andseemsto beinvolvedinerythroiddifferentiationduringerythropoiesis.5
In tumors, the mucin proteinundergoes proteolytic cleav-age,andreleasestheextracellularsegment,whichhasbeen identifiedasCA15.3,intothebloodstream.5Likewise,when
erythroblastsbecomeapoptotic,theCA15.3levelsincrease duetothereleaseofthemucinfragmentandLewisantigen, whichisalsorecognizedbytheantibodythatusuallybindsCA 15.3.Therefore,CA15.3levelsincreasewithoutanunderlying tumor.3,5,10StudieshaveshownthatserumCA15.3levelsare
associatedwiththedegreeofperipheralhemolysisand inef-fectiveerythropoiesisinvitaminB12deficiencyandinother causesofhemolysis,suchasthalassemiaorsicklecell ane-mia.CA15.3isnotrelatedwithanincreaseintheincidenceof breastcancerorotherneoplasmsovertime,onthecontrary,it isoneofthetumormarkersmorefrequentlyfoundincreased innon-neoplasticdiseases.3,10
Presentdatadonotrecommendusingtumormarkersfor screeninganddiagnosisduetolowsensitivityandspecificity; thefindingofanincreasedmarkerlevel mandatesimaging studiesinordertoexcludecancer,whichcanleadto inappro-priateandunnecessaryinvestigations.3,4
The treatment of pernicious anemia is based on 4–6 weekly administrations of intramuscular vitamin B12 at a dose of 1000g (cyanocobalamin quarterly; hydroxocobal-amin monthly) after a loading dose. Oral replacement therapy has not yet been validated in pernicious anemia andthetransfusionofpackedredbloodcellsisusuallynot indicated.6,7
Besides the replacement of vitamin B12 stores, it is important to be vigilant for the co-occurrence of gastric adenocarcinoma,neuroendocrinetumorsandmarginal non-Hodgkinlymphomabyendoscopicexamination,becausethey aremorefrequentinpatientswithperniciousanemia.1-3
Conclusion
This case illustratesspurious laboratory findings of mega-loblasticanemia. Moreover,anextensivelaboratory investi-gationsometimescanbemoreconfoundingthanclarifying, andlaboratoryabnormalitiesfoundintheacutephaseofthe diseasemayleadtounnecessaryexams.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
r
e
f
e
r
e
n
c
e
s
1.BizzaroN,AnticoA.Diagnosisandclassificationofpernicious
anemia.AutoimmunRev.2014;13(4–5):565–8.
2.AndresE,SerrajK.Optimalmanagementofpernicious
anemia.JBloodMed.2012;3:97–103.
3.SymeonidisA,Kouraklis-SymeonidisA,ApostolopoulosD,
ArvanitopoulouE,GiannakoulasN,VassilakosP,etal.
IncreasedserumCA15.3levelsinpatientswithmegaloblastic
anemiaduetovitaminB12deficiency.Oncology.
2004;67(5–6):359–67.
4.DuffyMJ.Serumtumormarkersinbreastcancer:aretheyof
clinicalvalue?ClinChem.2006;52(3):345–51.
5.RughettiA,BiffoniM,PierelliL,RahimiH,BonannoG,
BarachiniS,etal.RegulatedexpressionofMUC1epithelial
antigeninerythropoiesis.BrJHaematol.2003;120(2):344–52.
6.FarwellAP.Nonthyroidalillnesssyndrome.CurrOpin
EndocrinolDiabetesObes.2013;20(5):478–84.
7.PantaloneKM,NasrC.ApproachtoalowTSHlevel:patience
isavirtue.CleveClinJMed.2010;77(11):803–11.
8.GreenR.AnemiasbeyondB12andirondeficiency:thebuzz
aboutotherB’s,elementary,andnonelementaryproblems.
HematologyAmSocHematolEducProgram.2012;2012:492–8.
9.AsliniaF,MazzaJJ,YaleSH.Megaloblasticanemiaandother
causesofmacrocytosis.ClinMedRes.2006;4(3):236–41.
10.SymeonidisA,Kouraklis-SymeonidisA,ConstantinidouI,
SolomouE,KougelouS,VassilakosP,etal.IncreasedCA15.3
levelsintheserumofpatientswithhomozygous
beta-thalassaemiaandsicklecell/beta-thalassaemia.BrJ