Impacts of zinc layer and pouring method
on interface performance for Al-22Si/ZL104
bi-metal
*Xiao-guang Yuan
Male, born in 1963, Ph.D and Professor. His research focus is mainly on cast alloys and their casting technologies.
E-mail: [email protected]
Received: 2016-07-20; Accepted: 2016-12-10 Jun-feng Zhao1,2
, Fu-yang Cao2
, Jian-fei Sun2
, *Xiao-guang Yuan1
, Hong-xian Shen2
, and Xin-yi Zhao2 . 1. School of Materials Science and Engineering, Shenyang University of Technology, Shenyang 110178, China;
2. School of Materials Science and Engineering, Harbin Institute of Technology, Harbin 150001, China
M
any technological improvements have beendeveloped by automotive manufacturers for reducing harmful greenhouse gas emissions and improving
fuel eiciency [1]. In recent years, there has been a trend
towards the increased use of light-weight materials [2]. To improve the durability of engine cylinders, there is a need for developing new materials that exhibit improved abrasion resistance and mechanical strengths [3]. The
spray-formed hypereutectic Al-Si alloy possesses excellent abrasion resistance and a high strength-to-weight ratio, making this material an attractive choice for use as cylinder liners [4]. However, the application of spray-formed
hypereutectic Al-Si alloy has been limited due to relatively high production costs and complicated processing. Based on cost and the compatibility with the thermal physical properties, the use of Al22Si-aluminum bi-metal materials would be the most economical method to make local reinforcement materials [5]. However, few studies have
Abstract:
Bi-metal material consisting of spray-formed Al-22Si and ZL104 is a suitable candidate for applications in internal combustion engines. This research investigated the effects of surface treatment and appropriate gating system on the microstructures and mechanical properties in evaluating the optimal strategy for producing high quality bi-metal materials. The bi-metal materials were prepared using ZL104 gravity casting by different pouring types around the spray-formed Al-22Si with varied surface treatments. The wettability between Al-22Si and ZL104 was significantly improved when Zn coating was used to remove the natural oxide layer. This research also obtained the improved interfacial microstructures and interfacial bonding strength for materials when applying theappropriate pouring method. The hardness proiles of Al-22Si/ZL104 bi-metal were consistent with the observed
microstructures. The average tensile strength of the bi-metal material with zinc coating is ~42.3 MPa, which is
much higher than that with oxide ilm at ~10 MPa. The process presented is a promising and effective approach for
developing materials in the automotive industry.
Key words:
bi-metal; Al alloy; interface bonding; zinc coatingCLC numbers: TG146.21 Document code: A Article ID: 1672-6421(2017)01-039-07
been done to evaluate the properties of bi-metal materials used as cylinders in practice.
Bi-metal materials are designed as two metals put into contact with each other and then bonded together [6].
Metallurgical bonding as a chemical bonding method occurs between a substrate and a coating material located in close contact to the substrate and diffused evenly into the substrate. For Al-based bi-metals, a thin oxide layer with extremely high stability on the surface inhibits the metallurgical bonding formation during casting [7]. Therefore, many studies have been performed to remove this natural aluminium oxide layer to promote the metallic bonding [8,9].
The spray-formed Al-22Si alloy exhibits excellent abrasion resistance, pressure tightness, and has a low
coeicient of thermal expansion [10]. The ZL104 alloy
Fig. 1: (a) Schematic illustration of compound casting process and (b) after processing of cylinder liner
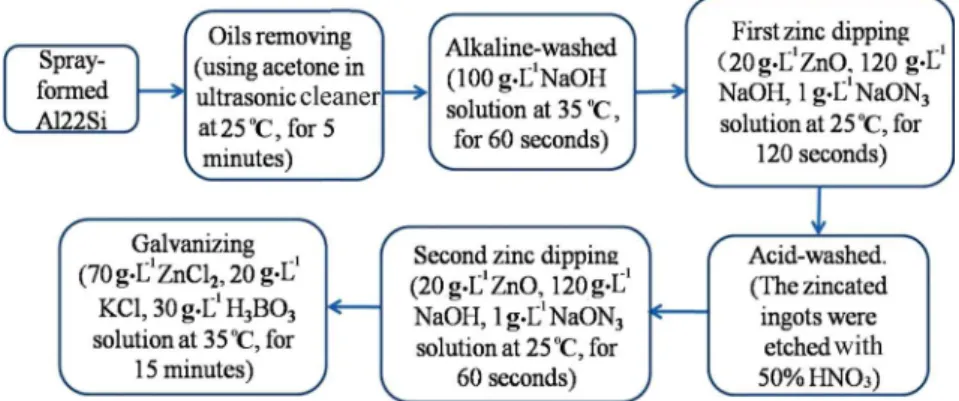
Fig. 2: Flow chart of Zn-coating preparation
Table 1: Chemical compositions of spray formed Al-22Si and commercial ZL104 alloys
Materials Si Fe Ni Cu Mn Mg V Al
Al22Si 21.51 2.17 2.12 2.96 0.70 0.65 0.12 Bal
ZL104 8.0-10.5 0-0.9 0.2-0.5 0.17-0.3 Bal
suitable galvanization and casting parameters for improving the interface performance of bi-metallic parts.
1 Experimental procedures
1.1 Raw materials
Al-22Si (wt.%) alloy was manufactured by spray deposition and hot extrusion, and the detailed process can be found in Ref. [11].
ZL104 used in the experiment was obtained from commercial used materials. Chemical compositions of the spray formed Al-22Si and ZL104 alloys are listed in Table 1.
1.2 Casting
The experimental schematic illustration is shown in Fig. 1(a). An Al-22Si cylinder with 105 mm diameter, 8 mm thickness, and 50 mm height was used as substrate. To observe the
matrix wettability with diferent surface treatments, half of the
matrix surface was subject to lathe processing of a 1 mm deep rectangular groove, as shown in Fig. 1(b). The spray-formed Al-22Si cylinder was heated at 350 °C in a furnace and held for 20 min. The ZL104 alloy was re-melted in an electrical furnace. The temperature of the ZL104 was kept at 900±10 °C for 20 min, then the melt was poured into a waiting mould. In
order to explore the efect of appropriate gating systems on the
interface, pouring systems with two ingates and four ingates were designed as shown in Fig. 4(c) and (d), respectively.
In this experiment, oxide film was removed through the zincate electroplating process. Figure 2 shows the Zn-coating preparation procedure [7]. Between all treatment steps, the ingots
were rinsed with deionized water to avoid contamination of chemicals. The quality of the electrodeposition layer depends on the galvanization time and current density. The optimum current density selected was 4 Adm-2 for 900 s, which allowed optimal
Fig. 3: SEM of the different process steps with zincate treatment of Al-22Si alloy: (a) removing of aluminum oxide, (b) after double zinc dipping, (c) after electric galvanization, (d) enlargement of zincate morphologies in (c)
1.3 Interface properties
The microstructure of the bi-metal interface was investigated by scanning electron microscopy (SEM) using a Gemini 1530 microscope equipped with an energy dispersive spectroscopy (EDS) detector.
The Vickers microhardness measurements were done with a HV-1000Z-type digital microhardness tester with 4.9 N load for 15s. The values reported thereafter are the average value of at least three measurements.
The interfacial bonding of the bimetal was evaluated according to tensile strength. The tensile specimens were tested using the tensile machine Instron 5569 at ambient temperature. The tensile strengths presented are the average of three measurements.
2 Results and discussions
2.1 Microstructures formed on the surface of
spray-formed Al-22Si at different steps of
zincate process
Figure 3 shows the typical microstructures formed on the surface
of spray-formed Al-22Si at diferent steps of the zincate process.
In Fig. 3(a), the aluminium oxide was cleared and after the pre-treatment of double zinc dip process, just a few small uncoated sections are visible, as indicated in Fig. 3(b). The zinc dipping
alone showed insuicient coverage of the aluminium alloy, thus
some areas remained uncovered. This zinc dipping followed by electrochemical deposition allowed more complete coverage of the surface with the zinc coating, as shown in Fig. 3(c). Fig. 3(d) shows an enlarged version of the zincate morphologies in Fig. 3(c). The zinc layer thickness shows minor variations due to the rough surface of the Al-22Si substrate. The plating thickness ensured the aluminium alloy is not oxidized before casting and improved the wettability of the substrate alloy.
2.2 Distribution of temperature ield and
stress ield in the casting process
Procast software was used to gain information about the temperature field and stress field of the aluminium melt.
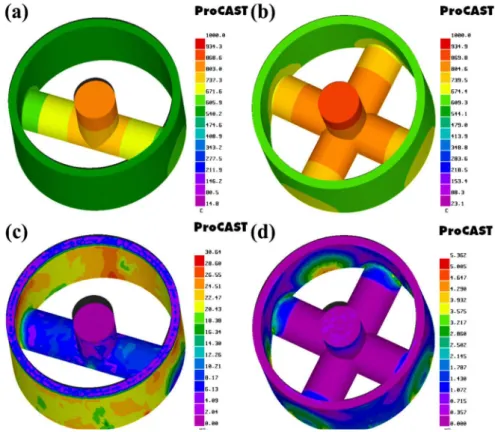
According to the temperature ield distribution diagram shown
in Fig. 4(a), when there are two ingates, the relatively large
temperature diferences appeared among diferent parts, resulting
pouring gates, which also means the higher temperature of metal liquid contacts the cylinder. On the other hand, the casting heat became enhanced and the cylinder temperature increased with
the increased number of ingates, so at the end, the solidiication
rate of the cast becomes slower after completion of mold
illing. This prolongs the reaction time between the casting and
cylinder and provides a more favourable temperature condition for elements diffusion during the bi-metallic combination. The gating system with four ingates shows a more uniform
solidiication temperature distribution than that with two ingates based on the temperature ield during the solidiication process.
Figures 4(c) and (d) show stress field distributions at the end
of solidiication with diferent gating systems. After the basic
solidification of the casting with two ingates, the ingates still
show liquid-solid two-phase when the casting almost solidiied
in the gating system with two ingates, which resulted in the relatively larger difference of solidifying contraction between
the casting and the ingate. The casting is under solid contraction while the ingate is still under solidification contraction, and as the solid contraction rate is lower than the solidification contraction rate, it leads to the tensile stress appearance at the joint between the ingate and the casting, and then the casting tends to elliptical deformation. At last, the break-off or loose coupling will appear at the position far from the ingate. The solidification state is homogeneous in the gating system with four ingates. So, the overall casting shows uniform contraction and smaller internal stress. Meanwhile, the inside of the casting shows compressive stress and the outside of the casting shows tensile stress, which is conducive to tight coupling and increases contact pressure between the casting and the cylinder. Therefore, from the perspective of the temperature field and stress field, gating systems with four ingates are more conducive to the best combination for the Al22Si/ZL104 bi-metal.
Fig. 4: Characteristic temperature ield during solidiication (a. two ingates, b. four ingates) and stress ield distribution at the end of solidiication (c. two ingates, d. four ingates)
2.3 Macro-characteristics
Figures 5(a)-5(c) show the macro characteristics of the bi-metal castings obtained using different methods. Figure 5(a) shows the interface between Al-22Si and ZL104 without galvanization: almost all the rectangular slots have observable gaps in the interfacial macro-structure, showing poor integrity of the bi-metal casting. Figure 5(b) shows the interfacial macro-morphology of the bi-metal casting after zinc plating and with two ingates. There are a few gaps in the rectangular slots, and these gaps are very small. Figure 5(c) shows the macro characteristics of the bi-metal material with four ingates and zinc coating, there are no macroscopic cracks visible at the compound interface.
In the studies of C. Koerner, and W. Jiang, et al., compound casting was used to achieve a metallurgical bonding interface between the two materials [7,12]. Without zinc coating and with
two ingates, the rectangular slots showed gaps due to the
formation of oxide ilm at the surface, as shown in Fig. 5(a). The gaps form because this thin oxide ilm inhibits metallic bonding
during the casting process. After electro galvanizing, the wettability between the Al-22Si and ZL104 is improved by the coating layer [13], thus increasing the integrity of the interface.
Fig. 5: Marco-characteristics of Al-22Si /ZL104 bi-metal castings obtained using different treatment methods: (a) without zinc plating, (b) two ingates after being galvanized, (c) four ingates after being galvanized
Fig. 6: SEM micrograph of interfacial microstructures of Al-22Si/ZL104 bi-metal using different treatment methods: (a) without zinc plating and with two ingates, (b) two ingates after galvanizing, (c) four ingates after galvanizing of molten metals. When the casting system was added to four
ingates, the gaps disappeared, which indicates the bonding improved between the Al-22Si and ZL104 at the interface by the ameliorative casting system. Therefore, for achieving excellent metallic bonding at interface, the spray-formed Al-22Si alloy cylinder should be coated with a Zn layer, and the means of improved pouring type should also be carefully controlled for conditioning the temperature during the forming process.
2.4 Interfacial microstructures
The SEM micrographs of Al-22Si/ZL104 bi-metal material
using diferent treatment methods are shown in Fig. 6, which
shows that different surface treatments resulted in different interfacial microstructures. There was a clear gap observed between the two materials for the samples without zinc coating and two ingates, as shown in Fig. 6(a). After electro galvanizing, the gap at the interface decreased, and the integrity of the interface was improved, as shown in Fig. 6(b). However, some voids were found in the region within the interlayer. Through the addition of up to four ingates, metallurgical bonds occurred during solidification and showed good bonding along the interface, as shown in Fig. 6(c).
In Fig. 6(a), the gaps exist between Al-22Si and ZL104 due to the absence of the zinc coating. The presence of this gap and
the high melting point of oxide particles will afect metallurgic
bonding and the performance of the bi-metal parts. In Fig. 6(b), the zinc coating improved the wettability between the
spray-formed Al-22Si and ZL104 melt, thus the number of gaps at the interface was decreased, but a small number of voids remained. The presence of voids at the interface results from the shrinkage
of the alloy during solidiication [5]. Our indings show that the
zinc coating provides limited improvement of bonding. M. Joseph Fernandus et al [14] showed that difusion depends mainly
on temperature and would increase with time. The rate of
difusion slowed over time according to Fick’s Second Law [15].
Therefore, if the temperature is not appropriate, the formation of an ideal interface will be inhibited. The experiment shown in Fig. 6(c) exhibited better bonding compared with other treatment methods because the temperature decreased more slowly due to the design of four ingates. The liquidus melting temperature of Zn and Al-22Si is ~420 °C and ~722 °C, respectively, and the thin coating will be melted at the pouring temperature. The pouring temperature of the ZL104 was ~900 °C, therefore
when the heat lux ills into the composite region, it will melt
part of the Al-22Si, thus allowing liquid-liquid interaction to occur. G. Mahendran [16] showed that liquid-liquid interaction of two metals was the optimal condition for formation of a sound interface during bi-metal casting.
2.5 Interfacial properties
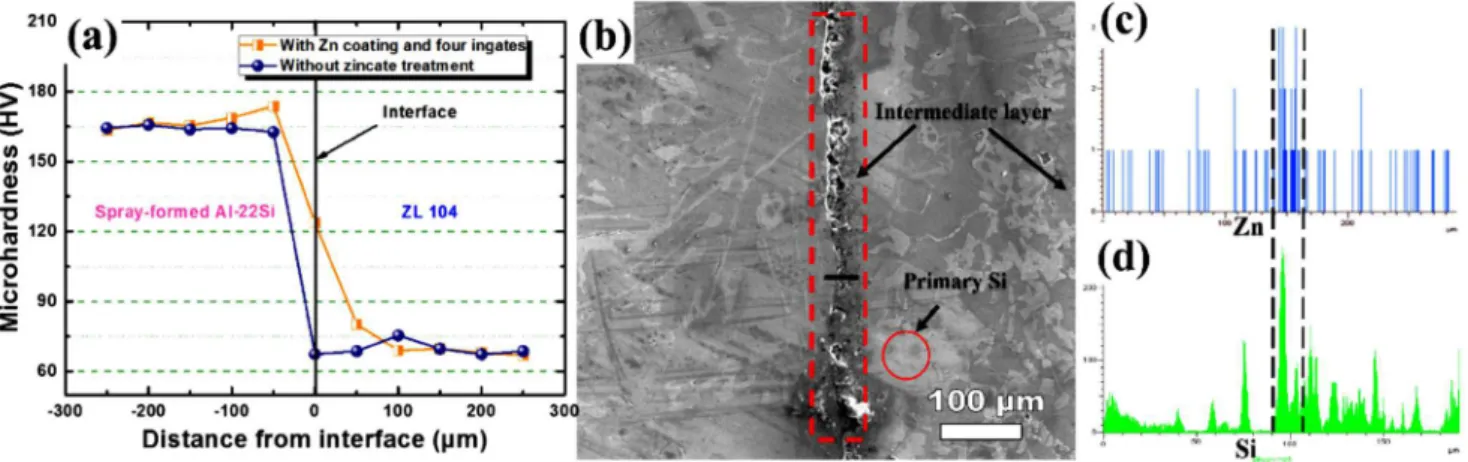
To understand the performances of the interface, Vickers microhardness was measured for the bi-metal material prepared
with diferent surface treatment methods. As shown in Fig. 7, the
alloy ranged from 160-175 and from 65-80, respectively. The hardness changed gradually across the interface for the sample with Zn coating and four ingates. In contrast, the sample without zincate treatment changed sharply and exhibited poor performance due to the presence of the oxide layer. The average values of hardness remained unchanged within the Al-22Si side, but decreased slowly near the vicinity of the interface as well as
within the ZL104 side. This is due to the difusion of Si atoms
from Al-22Si into ZL104 during casting processes [17].
During the solidiication of the bi-metal casting, Si difusion occurred from Al-22Si to the ZL104 resulting in the difusion bonding at the interface (Fig. 7d). At the start of difusion, the concentration diference of Si near the interface was relatively
large. Previous reports [7,18] suggested that difusion of alloying elements, with the help of high temperature, is required to produce an interface bonding. The Si phase grows from Al-22Si to the interface with ZL104. Additional elements of the cast alloy like iron or copper are neglected in the plots as their concentration is too low compared with silicon. Due to
Fig. 8: Interfacial bonding strength under different preparing conditions: 1- without zinc plating; 2- two ingates after galvanizing; 3- four ingates after galvanizing
the zinc content at interface being too low compared to the
silicon, its efect on the hardness efects have not been observed
in mechanical investigations. The zinc coating complete dissolution into the bulk compound and its primary functions on the Al-22Si are wetting properties.
The interfacial bonding strength of the bi-metals prepared
with diferent conditions are shown in Fig. 8. For the bi-metal
material made without galvanizing treatment, the Al-22Si and ZL104 alloy were barely bonded and the interfacial bonding strength was very low. The bi-metal of the Al-22Si alloy with electro-plated zinc layer showed increased interfacial bonding strength. With optimal pouring methods and with Zn coating, a better interface was formed with better strength.
The interface between Al-22Si and ZL104 showed reduced interfacial bonding strength due to the presence of voids [19].
The formation of the voids occurred due to the oxidation of the intermediate layer, as shown in Fig. 6(a) and resulting in a lack of bonding. Bonding strength depends on both the quality of the interface and composition [14, 20]. Without Zn coating, the
poor interface explained the poor strength of the joint. With Zn coating, there was a considerable improvement in strength. Carolin Koerner et al. [7] demonstrated that bonding strength
is not dominated by Zn dissolution and difusion but from the
extent of binding faults. Using Zn coating and an optimal casting method for decreasing the cooling rate allowed the formation of a better interface with improved strength.
3 Conclusions
(1) The very thin Zn coating on the spray-formed Al-22Si substrate improved wettability with molten ZL104 greatly, which provides enhanced interfacial bonding in the bi-metal material.
(2) A sound interface was obtained by using zinc coating on the surface of solid Al-22Si and carefully controlling the casting procedure for decreasing the cooling rate.
(3) The hardness profiles of the bi-metal material changed Fig. 7: (a) Variations in Vickers microhardness values from Al-22Si (left side) to ZL104 (right side) for different surface
gradually when with Zn coating and four ingates, but changed sharply if with oxide layer and two ingates, which agreed with the observed microstructures.
(4) The average tensile strength of bi-metals with zinc coating is ~42.3 MPa, which is much higher than that without zinc coating of ~10 MPa .
References
[1] McClure R J, Adriazolasteil C, Mulvihill C, et al. Simulating the dynamic effect of land use and transport policies on the health of populations. American Journal of Public Health, 2015, 105: S223-S229.
[2] Lee W, Lai Y, Chen P. A study on energy saving and CO2 emission reduction on signal countdown extension by vehicular Ad Hoc networks. IEEE Transactions on Vehicular Technology, 2015, 64(3): 890-900.
[3] Li J, Guoji C, Xiong S, et al. The fracture behaviors of A390 aluminum alloys cylinder liner under static loading. Special Casting and Nonferrous Alloys, 2012, 32(12): 1079-1085. (In Chinese)
[4] Hou L G, Cui H, Cai Y H. et al. Effect of (Mn+Cr) addition on the microstructure and thermal stability of spray-formed hypereutectic Al-Si alloys. Materials Science and Engineering: A, 2009, 527(1-2): 85-92.
[5] Sacerdote-Peronnet M, Guiot E, Bosselet F, et al. Local reinforcement of magnesium base castings with mild steel inserts. Materials Science and Engineering: A, 2007, 445-446: 296-301.
[6] Şimşir M, Kumruoğlu L C, Özer A. An investigation into stainless-steel/structural-alloy-steel bimetal produced by shell
mould casting. Materials & Design, 2009, 30(2): 264-270.
[7] Koerner C, Schwankl M, Himmler D. Aluminum-aluminum
compound castings by electroless deposited zinc layers. Journal of Materials Processing Technology, 2014, 214(5): 1094-1101.
[8] Rübner M, Günzl M, Körner C, et al. Aluminium–aluminium
compound fabrication by high pressure die casting. Materials Science and Engineering: A, 2011, 528(22-23): 7024-7029. [9] Liu Liming, Tan Jinhong, Liu Xujing. Reactive brazing of Al alloy
to Mg alloy using zinc-based brazing alloy. Materials Letters,
2007, 61(11-12): 2373-2377.
[10] Taghiabadi R, Ghasemi H M, Shabestari S G. Effect of iron-rich intermetallics on the sliding wear behavior of Al-Si alloys. Materials Science and Engineering: A, 2008, 490(1-2): 162-170.
[11] Jia Y D, Cao F Y, Ning Z L, et al. Hot deformation behavior of spray formed Al-22Si-5Fe-3Cu-1Mg alloy. Transactions of
Nonferrous Metals Society of China, 2011, 21(21): s299–s303. [12] Jiang W, Fan Z, Li C. Improved steel/aluminum bonding in
bimetallic castings by a compound casting process. Journal of Materials Processing Technology, 2015, 226: 25-31.
[13] Xu G, Luo A A, Chen Y, et al. Interfacial phenomena in
magnesium/aluminum bi-metallic castings. Materials Science
and Engineering: A, 2014, 595: 154-158.
[14] Fernandus M J, Senthilkumar T, Balasubramanian V. Developing Temperature-Time and Pressure-Time diagrams for diffusion bonding AZ80 magnesium and AA6061 aluminium alloys. Materials & Design, 2011, 32(3): 1651-1656.
[15] Dai J, Jiang B, Li X, et al. The formation of intermetallic
compounds during interdiffusion of Mg-Al/Mg-Ce diffusion
couples. Journal of Alloys and Compounds, 2015, 619(9): 411-416.
[16] Mahendran G, Balasubramanian V, Senthilvelan T. Developing
diffusion bonding windows for joining AZ31B magnesium–
AA2024 aluminium alloys. Materials & Design, 2009, 30(4): 1240-1244.
[17] Liu N, Jie J, Lu Y, et al. Characteristics of clad aluminum hollow billet prepared by horizontal continuous casting. Journal of Materials Processing Technology, 2014, 214(1): 60-66.
[18] Papis K J M, Löfler J F, Uggowitzer P J. Interface formation
between liquid and solid Mg alloys — An approach to continuously metallurgic joining of magnesium parts. Materials Science and Engineering: A, 2010, 527(9): 2274-2279. [19] Shirzadi A A, Assadi H, Wallach E R. Interface evolution and
bond strength when diffusion bonding materials with stable
oxide ilms. Surface and interface analysis, 2001, 31(7): 609 -618.
[20] Nerl C, Wimmer M, Hoffmann H, et al. Development of a continuous composite casting process for the production of bilayer aluminium strips. Journal of Materials Processing Technology, 2014, 214(7): 1445-1455.