ww w . r e u m a t o l o g i a . c o m . b r
REVISTA
BRASILEIRA
DE
REUMATOLOGIA
Original
article
Clinical
characteristics
and
frequency
of
TLR4
polymorphisms
in
Brazilian
patients
with
ankylosing
spondylitis
Natalia
Pereira
Machado
a,
Eliana
Nogueira
b,
Karen
Oseki
a,
Pâmela
Carolina
Cruz
Ebbing
a,
Clarice
Silvia
Taemi
Origassa
b,
Tatiane
Mohovic
b,
Niels
Olsen
Saraiva
Câmara
b,
Marcelo
de
Medeiros
Pinheiro
a,∗aDivisãodeReumatologia,DepartamentodeMedicina,UniversidadeFederaldeSãoPaulo(UNIFESP),SãoPaulo,SP,Brazil bDivisãodeNefrologia,DepartamentodeMedicina,UniversidadeFederaldeSãoPaulo(UNIFESP),SãoPaulo,SP,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received10February2016
Accepted1May2016
Availableonline5August2016
Keywords:
Ankylosingspondylitis
TLR-4polymorphisms
HLA-B27
a
b
s
t
r
a
c
t
Objectives:Innateimmunityisinvolvedinthephysiopathologyofankylosingspondylitis
(AS),withtheparticipationofGram-negative bacteria,modulationofhumanleukocyte
antigen(HLA)B27andtheinvolvementofpatternrecognitionreceptors,suchasToll-like
receptors(TLRs).Theaimofthisstudywastoinvestigatetheclinicalcharacteristicsand
fre-quencyofTLR4polymorphisms(Asp299GlyandThr399Ile)inacohortofBrazilianpatients
withAS.
Methods:Across-sectionalstudywascarriedoutinvolving200patientswithadiagnosis
ofASandahealthycontrolgroupof200individuals.Diseaseactivity,severityand
func-tionalcapacityweremeasured.ThestudyofTLR4polymorphismswasperformedusingthe
restrictionfragmentlengthpolymorphismmethod.HLA-B27wasanalyzedbyconventional
polymerasechainreaction.TheIBMSPSSStatistics20programwasusedforthestatistical
analysis,withp-valueslessthan0.05consideredsignificant.
Results:Meanageanddiseasedurationwere43.1±12.7and16.6±9.2years,respectively.
Thesamplewaspredominantlymale(71%)andnon-Caucasian(52%).A totalof66%of
thegroupofpatientswerepositiveforHLA-B27.Thesampleofpatientswascharacterized
bymoderatefunctionalimpairmentandahighdegreeofdiseaseactivity.Nosignificant
associationwasfoundbetweenthetwoTLR4polymorphismsandsusceptibilitytoAS.
Conclusions:TLR4polymorphisms399and299werenotmorefrequentinpatientswithAS
incomparisontothehealthcontrolsandnoneoftheclinicalvariableswereassociatedwith
thesepolymorphisms.
©2016ElsevierEditoraLtda.ThisisanopenaccessarticleundertheCCBY-NC-ND
license(http://creativecommons.org/licenses/by-nc-nd/4.0/).
∗ Correspondingauthor.
E-mail:[email protected](M.M.Pinheiro).
http://dx.doi.org/10.1016/j.rbre.2016.07.004
2255-5021/©2016ElsevierEditoraLtda.ThisisanopenaccessarticleundertheCCBY-NC-NDlicense(http://creativecommons.org/
Características
clínicas
e
frequência
de
polimorfismos
em
TLR4
em
pacientes
brasileiros
com
espondilite
anquilosante
Palavras-chave:
Espondiliteanquilosante
PolimorfismosemTLR-4
HLA-B27
r
e
s
u
m
o
Objetivos: Aimunidadeinataestáenvolvidanafisiopatologiadaespondiliteanquilosante
(EA),comaparticipac¸ãodebactériasgram-negativas,modulac¸ãodoantígenoleucocitário
humano(HLA)B27eoenvolvimentodereceptoresdereconhecimentodepadrões,comoos
receptoresToll-like(TLR).Oobjetivodesteestudofoiinvestigarascaracterísticasclínicasea
frequênciadepolimorfismosemTLR4(Asp299GlyeThr399Ile)emumacoortedepacientes
brasileiroscomEA.
Métodos: Fez-seum estudotransversalqueenvolveu200pacientescomdiagnósticode
EAeumgrupocontrolesaudávelde200indivíduos.Mediram-seaatividadedadoenc¸a,a
gravidadeeacapacidadefuncional.OestudodospolimorfismosemTLR4foifeitocomo
métododepolimorfismodefragmentosderestric¸ão.OHLA-B27foianalisadoporreac¸ão
emcadeiadapolimeraseconvencional.Usou-seoprogramaSPSSStatistics20daIBMpara
aanáliseestatísticaeforamconsideradossignificativosvaloresdepinferioresa0,05.
Resultados: Amédiadeidadeeadurac¸ãodadoenc¸aforamde43,1±12,7e16,6±9,2anos,
respectivamente.Aamostrafoipredominantementedosexomasculino(71%)edenão
bran-cos(52%).Dogrupodepacientes66%eramHLA-B27positivos.Aamostradepacientesfoi
caracterizadaporumaalterac¸ãofuncionalmoderadaeumelevadograudeatividadeda
doenc¸a.Nãofoiencontradaassociac¸ãoestatisticamentesignificativaentreospolimorfismos
emTLR4easusceptibilidadeàEA.
Conclusões: OspolimorfismosemTLR4399e299nãoforammaisfrequentesempacientes
comEAemcomparac¸ãocomcontrolessaudáveisenenhumadasvariáveisclínicasesteve
associadaaessespolimorfismos.
©2016ElsevierEditoraLtda.Este ´eumartigoOpenAccesssobumalicenc¸aCC
BY-NC-ND(http://creativecommons.org/licenses/by-nc-nd/4.0/).
Introduction
Pattern-recognition receptors (PRRs) are a set of receptors
involvedin recognitionofpathogens in multicellular
orga-nisms.Toll-like receptors(TLRs)1functionasPRRsandplay
anessentialroleintherecognitionofmicrobialcomponents
and endogenous ligandsinduced duringthe inflammatory
response.2–4
AmongthegenepolymorphismsofTLR,someofthemost
widelystudiedaretwoco-segregatedfunctionalmutationsin
theextracellulardomainofhumanTLR4,whicharelocatedon
chromosome4andareassociatedwithhyporesponsiveness
tobacteriallipopolysaccharides(LPS).5Basedonitsevidence
ofassociation withanincreasedriskofinfectionby
Gram-negativebacteria,5suchpolymorphismshavebeenevaluated
insomeinflammatorydiseasesinwhichtheparticipationof
thesemicroorganismshasbeenimplicatedinthe
etiopathol-ogy,suchasankylosingspondylitis(AS).
Basedonthepremiseofsubclinicalcolitisinpatientswith
ASandanimalmodelsthatdemonstratetheparticipationof
aninfectioustriggerbyGram-negativebacilli6,7modulatedby
thepresentationofthe antigentoHLAB27,8 theaimofthe
present study was to identifythe frequencyofTLR4
poly-morphisms (Asp299Glyand Thr399IIe) inBrazilianpatients
withASandinvestigatepossibleassociationsbetweenthese
polymorphismsand greater susceptibilitytothe disease as
wellasclinicalandlaboratoryaspectsofdiseaseactivityand
chronicity.
TheresultsoftheassociationbetweenTLRpolymorphisms
and AS have been controversial in some clinical trials,9
probably becauseof differentpopulation studied.This fact
motivates investigation ofthis association inmiscigenated
populations,likeBrazilianone.
Materials
and
methods
TwohundredpatientswithadiagnosisofASaccordingtothe
modifiedNewYorkcriteria10oraxialspondyloarthritis11were
recruitedfromthespondyloarthritisclinicofthe
Rheumato-logySectoroftheFederalUniversityofSaoPaulo(Brazil)and
200healthyindividualswereselectedfromamongvolunteers
givingbloodatthehospitalofthesameinstitutionbetween
May2011andOctober2013.Allparticipantsagreedto
partici-pateinthestudybysigningastatementofinformedconsent
(ethicscommitteenumber1804/10).
Demographic and clinical data were collected and
spe-cificassessmenttoolswereemployedforthecharacterization
ofdisease activity andseverity, such asthe Bath
Ankylos-ingSpondylitisDiseaseActivityIndex(BASDAI),12Ankylosing
SpondylitisDiseaseActivityScore(ASDAS),13 Bath
Ankylos-ing SpondylitisFunctionalIndex (BASFI),14 BathAnkylosing
Spondylitis Metrology Index (BASMI)15 and modified Stoke
AnkylosingSpondylitisSpineScore(mSASSS).16
TheanalysisofAsp299GlyandThr399Ilepolymorphisms
of TLR4 was performed using polymerase chain reaction
(NCOIfor299andHinfIfor399).17,18GenomicDNAextraction
was performed using whole blood collected in
ethylenedi-aminetetraacetate (EDTA) usinga DNA extractionkit (DNA
NucleoSpin®, Macherey-Nagel). The PCRamplification was
performedwith50ngoftheDNAtobestudiedinatotal
vol-umeof20Lcontaining0.8Lofpotassiumchloride50mM,
2L ofTris(pH 8.4),0.6mMof magnesium chloride,0.4L
ofeachprimer(10nM),0.4Lofdeoxyribonucleotidemixture
(dATP,dCTP,dGTPanddTTP)and0.06LunitsofTaqplatinum
DNApolymerase(0.015U/L).
Theautomaticthermalcycler(MJResearchPTC-200)was
programmedforamplification:initialdenaturation(95◦Cfor
4min),followedby35cyclesof95◦Cfor45s,55◦Cfor30sand
72◦Cfor1min30s,withfinalextensionat72◦Cfor10min.The
forwardandreverseprimerswererespectively5′-GATTAGCAT
ACTTAGACTACTACCTCCATG-3′and5′-GATCAACTTCTG
AAAAAGCATTCCCAC-3′forAsp299Glyand5′-GGTTGCTGT
TCTCAAAGTGATTTTGGGAGAAand5′-CCTGAAGACTGG
AGAGTGAGTTAAATGCT-3′forThr399Ile.Electrophoresisin
2%agarosegelwasperformedtoconfirmtheDNA
amplifica-tion.
Analiquotof5Lwiththeappropriaterestrictionenzyme
wasusedfordigestionofthePCRproductat37◦Cfor2h.
Elec-trophoresiswasperformedin4%agarosegel(Agarose1000
Invitrogen, Eugene,OR, USA)for identification ofthe TLR4
alleles.ThegelwasstainedwithSybrGold(Nucleicacidgel
stain,Invitrogen,Eugene,OR,USA)andvisualizedusingthe
Storm849system(MolecularDynamics,USA).
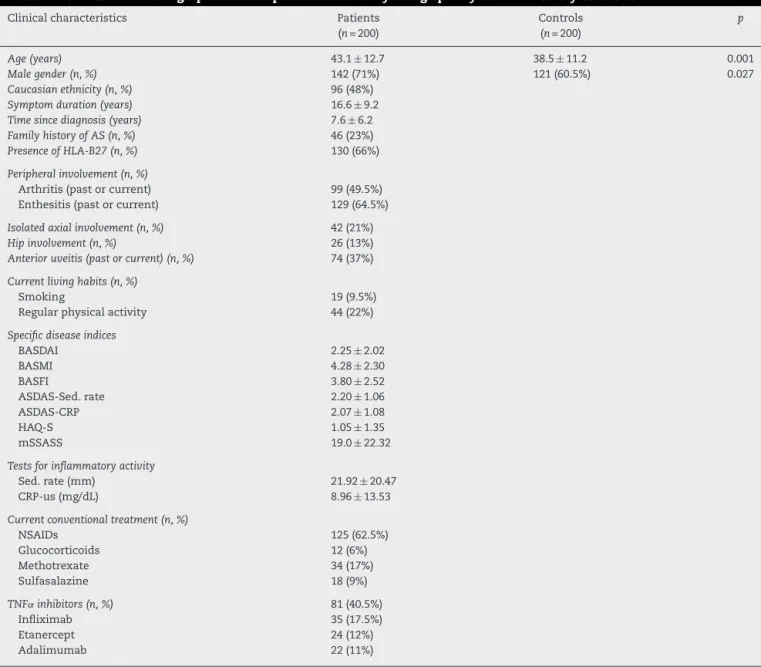
Table1–Clinicalanddemographicdataofpatientswithankylosingspondylitisandhealthycontrols.
Clinicalcharacteristics Patients
(n=200)
Controls (n=200)
p
Age(years) 43.1±12.7 38.5±11.2 0.001
Malegender(n,%) 142(71%) 121(60.5%) 0.027
Caucasianethnicity(n,%) 96(48%)
Symptomduration(years) 16.6±9.2
Timesincediagnosis(years) 7.6±6.2
FamilyhistoryofAS(n,%) 46(23%)
PresenceofHLA-B27(n,%) 130(66%)
Peripheralinvolvement(n,%)
Arthritis(pastorcurrent) 99(49.5%) Enthesitis(pastorcurrent) 129(64.5%)
Isolatedaxialinvolvement(n,%) 42(21%)
Hipinvolvement(n,%) 26(13%)
Anterioruveitis(pastorcurrent)(n,%) 74(37%)
Currentlivinghabits(n,%)
Smoking 19(9.5%)
Regularphysicalactivity 44(22%)
Specificdiseaseindices
BASDAI 2.25±2.02
BASMI 4.28±2.30
BASFI 3.80±2.52
ASDAS-Sed.rate 2.20±1.06
ASDAS-CRP 2.07±1.08
HAQ-S 1.05±1.35
mSSASS 19.0±22.32
Testsforinflammatoryactivity
Sed.rate(mm) 21.92±20.47
CRP-us(mg/dL) 8.96±13.53
Currentconventionaltreatment(n,%)
NSAIDs 125(62.5%)
Glucocorticoids 12(6%)
Methotrexate 34(17%)
Sulfasalazine 18(9%)
TNF˛inhibitors(n,%) 81(40.5%)
Infliximab 35(17.5%)
Etanercept 24(12%)
Adalimumab 22(11%)
AS,ankylosingspondylitis;NSAID,non-steroidalanti-inflammatorydrug;BASDAI,Bathankylosingspondylitisdiseaseactivityindex;BASMI, Bathankylosingspondylitismetrologyindex;BASFI,Bathankylosingspondylitisfunctionindex;ASDAS,Ankylosingspondylitisdiseaseactivity score;HAQ-S,Healthassessmentquestionnaire-Spondylitis;mSSASS,modifiedStokesankylosingspondylitisspinescore;Sed.rate,blood sedimentationrate;CRP-us,ultrasensitiveC-reactiveprotein.
TheanalysisofHLA-B27wasperformedusingconventional
PCRinanautomaticthermalcycler (MJResearchPTC-200):
70ngofDNAfromeachsample,0.9mol/Lofeachforwardand
reverseprimer,1.1mmol/Lofmagnesiumchloride,200mol/L
ofdeoxyribonucleotidemixture(dATP,dCTP,dGTPanddTTP)
and2UofTaqDNApolymeraseforatotalvolumeof25L.The
forwardandreverseprimersforthereactionwererespectively
E91S(5′-GGGTCTCACACCCTCCAGAAT-3′)and136AS(5′-CGG
CGGTCCAGGAGCT-3′).Thecyclingconditionswere100sat
94◦C,followedby30 cyclesof1minat94◦C,1minat64◦C
and2minat72◦C,withfinalextensionat72◦Cfor10minin
40consecutivecycles.
Toevaluatethereactionquality(internalcontrol),reactions
were performedusing primers for-globinfor all samples
underthesameconditionsusedforthe HLA-B27reactions.
TheprimersforthesereactionswerePCO4(5′-CAACTTCAT
CCACGTTCACC-3′)andGH20(5′-GAAGAGCCAAGGACAGGT
AC-3′).ThePCRproductswereanalyzedthrough
electrophore-sisin1%agarosegelrunforonehourat100V.
Numerical data were expressed as mean and standard
deviation.TheKolmogorov–Smirnovtestwasusedto
deter-mine the distribution ofthe data (normal or non-normal).
EithertheMann–WhitneytestorKruskal–Wallistestwasused
forcomparisonsamongthecategoricalandnumericaldata.
Thechi-squaretestwasusedtodeterminethedistributionof
theTLR4polymorphisms betweengroupsaswellasforthe
comparisonofthecategoricalvariables.Spearman’s
correla-tion coefficientswere calculated todeterminethe strength
ofcorrelationsamongthecontinuousvariables.Bivariateand
multivariatelogisticregressionmodelswereconstructedwith
variables thatexhibited significant associationsin the
pre-vious tests. The IBM SPSS 20 program was used for the
statisticalanalysis, withp-valuesless than 0.05considered
significant.
Results
Table 1 displays the clinical and demographic data of the
200 patients. As expected, the male gender was
predomi-nant.Approximatelyonequarterofthepatientshadafamily
historyofASand halfthesamplereportedcurrent orpast
peripheralinvolvement.Nearly40%ofthepatientshad
ante-rioruveitis.Theprevalenceratesofcurrentsmokingandthe
regularpracticeofphysicalexercisewerelow.
Meandiseaseactivitywashigh,withsignificantfunctional
andmobilityimpairmentreflectingthelongdurationofthe
disease.Morethan60%ofthepatientsmaderegularuseof
anon-steroidalanti-inflammatorydrugand35%useda
syn-theticdisease-modifyinganti-rheumaticdrug.Nearlyhalfthe
patientsusedTNF␣inhibitors,withequaldistributionamong
etanercept,infliximabandadalimumab.
The study of HLA-B27 was performed on 197 (98.5%)
patientswithASand60(30%)healthyindividuals,66%(n=130)
and1.6%(n=1)ofwhomtestedpositive,respectively.
Extra-articularmanifestationswerefoundin80patients(40%),the
mostfrequentofwhichwasacuteanterioruveitis(n=74;37%),
followedbycircinatebalanitis(n=3;1.5%),nonspecificcolitis
(n=2;1%)andsterileurethritis(n=1;0.5%).
Table2–FrequencyofTLR4polymorphisms(299and 399)inpatientsandcontrols.
TLR-4polymorphisms Patients n=200
Controls n=200
p
Asp299Gly
Wild 182(91%) 178(89%) 0.505
Heterozygote 17(8.5%) 22(11%)
Homozygote 1(0.5%) 0
Thr399IIe
Wild 187(93.5%) 186(93%) 0.50 Heterozygote 13(6.5%) 14(7%)
Homozygote 0 0
Chi-squaretest.
Nostatisticallysignificantdifferencesbetweenthepatients and healthy controls were found regardingAsp299Gly and Thr399Ile polymorphisms. Due to the very low number of homozygotes,heterozygousandhomozygouspatientswere included in the same group for the tests. The 299 and 399 polymorphisms were in Hardy–Weinberg(HW) equilib-riuminthepatients(HW-2=0.73,p=0.39andHW-2=0.22, p=0.63,respectively)andcontrols(HW-2=0.68,p=0.41and
HW-2=0.26,p=0.61,respectively),demonstratingthe
conser-vationofgenotypefrequenciesacrossgenerations(Table2).A
tendency wasfoundtowardagreaterfrequencyof
non-co-segregationoftheallelesinthecontrolgroupincomparison
tothepatientswithAS(Table3).
For a more detailed analysis, the patients were
sepa-ratedintosubgroupsbasedonclinicalcharacteristics.Female
patientshadahigherbodymassindex(BMI)aswellashigher
HAQ-SandASDAS-Sed.ratescores.Femalesalsohadshorter
diseaseduration,lesserseverityofsacroiliacinvolvementas
well aslower mSASSS and BASMI scoresincomparisonto
males (Table3).Aftercontrolling forconfoundingvariables
inthelogisticregressionmodel,onlyBMI(p=0.014)andthe
BASMIscore(p=0.02)remainedsignificantlyassociatedwith
gender.
InthelogisticregressionanalysisusingpositivityforB27as
the dependentvariable,significant associationswerefound
forperipheral arthritis(p=0.039),uveitis (p=0.033)and the
useofTNF␣inhibitors(p=0.003)(Table4).Amongthepatients
positiveforHLA-B27,atendencywasfoundtowarda
predom-inanceofCaucasians(p=0.058).Moreover,greaterprevalence
rateswerefoundinthisgroupregardinganterioruveitis,more
severesacroiliacinvolvement(GradeIV),longerdisease
dura-tion and a greater frequency of biological agents (p<0.05)
(Table5).
Patients with the adult formof the disease had higher
scoresontheBASFI,BASDAIandHAQ-S,wereolderandhad
longerdiseaseduration.Thosewiththejuvenileformofthe
diseasemorefrequentreportsofafamilyhistoryofAS,made
moreuseofsulfasalazineandreportedmoresideeffectsfrom
TNF␣inhibitors.However,noneofthesevariablesremained
statistically significant in the final multiple regression
model.
Non-CaucasianpatientshadahigherBMIandBASMIscore
incomparisontoCaucasianpatients(Table6).However,only
Table3–Characteristicsofpatientswithankylosing spondylitiesaccordingtogender.
Variables Male
(n=142)
Female (n=58)
p
Ethnicity
Caucasian 67(47.2%) 29(50%)
Non-Caucasian 75(52.8%) 29(50%) 0.717
Onset
Adult 118(83.1%) 50(86.2%)
Juvenile 24(16.9%) 8(13.8%) 0.586
HLA-B27+ 96(67.6%) 34(58.6%) 0.158
Arthritis 68(47.9%) 31(53.4%) 0.475
Entesitis 93(65.5%) 36(62%) 0.646
Anterioruveitis 53(37.3%) 21(36.2%) 0.882 Familyhistoryof
AS
36(25.3%) 10(17.2%) 0.216
Pureaxial involvement
29(20.4%) 13(22.4%) 0.754
BMI(kg/m2) 24.81±3.72 25.80±3.87 0.02
BASMI 4.63±2.22 3.43±2.27 0.001
mSASSS 22.54±23.34 10.64±17.2 <0.0001 ASDAS-Sed.rate 2.11±1.05 2.42±1.04 0.036
HAQ-S 1.03±0.61 1.11±2.33 0.03
Sed.rate(mm) 19.3±19.6 28.35±21.31 0.001 Diseaseduration
(years)
17.5±9.43 14.43±8.5 0.02
Radiographicsacroilitis
II 11(7.7%) 4(7.0%) 0.044
III 66(46.5%) 38(65.5%)
IV 65(44.0%) 16(27.6%)
AS, ankylosing spondylitis; BMI, body mass index; BASMI, Bath ankylosing spondylitismetrology index; ASDAS, Ankylos-ingspondylitisdiseaseactivityscore;HAQ-S,Healthassessment questionnaire-Spondylitis; mSASSS,modified Stokes ankylosing spondylitisspinescore;Sed.rate,bloodsedimentationrate; chi-squareandMann–Whitneytests.
Thepvalueinboldmeansstatisticallysignificant.
analysis(p=0.004).Themostfrequentdegreeofradiographic sacroiliitiswasGradeIII(n=103;51.8%),followedbyGradesIV (40.5%)andII(7.5%).
AmongthepatientsonTNF␣inhibitors,20(24.7%)needed tochangeagents:five(6.2%)duetoprimaryfailure(6.2%),six (7.4%)duetosecondaryfailureand 13(16%)duetoadverse events,especiallyinfusionreactionsandinfection.Moreover, four(2%)ofthesepatientschangedTNF␣inhibitorsformore thantwoagents.
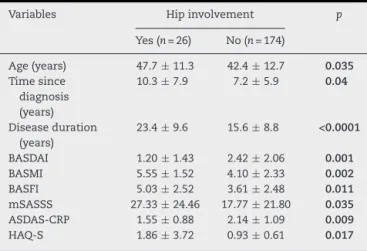
Anterioruveitiswasassociatedwithlongerdisease dura-tion(p=0.002),butlostitsstatisticalsignificanceinthefinal model.Hipinvolvementwasassociatedwithchronicityand lower disease activity scores(datanotshown). In thefinal logisticregressionmodel,longerdiseaseduration(p=0.039), a higher BASFI score (p=0.027) and a lower BASDAI score (p=0.024)remainedstatisticallysignificant.
PatientswithlongerdiseasedurationhadhighermSASSS andBASMIscoresaswellasalowerASDAS-Sed.ratescore. In the final model, only ASDAS-ESH (p=0.015) remained significant. Longer symptom duration was correlated with higherBASMI,BASFI,HAQ-SandmSSASS scoresaswell as alower BASDAIscore.Inthe finalmodel, BASMI(p<0.0001)
Table4–Characteristicsofpatientswithankylosing spondylitisaccordingtopositivityforHLA-B27.
Variables HLA-B27 positive (n=130)
HLA-B27 negative (n=67)
p
Ethnicity
Caucasian 69(53.1%) 26(39%) 0.058 Non-Caucasian 61(46.9%) 41(61.2%)
Onset
Adult 103(82.4%) 55(87.3%) 0.386 Juvenile 22(17.6%) 8(12.7%)
Arthritis 58(44.6%) 40(59.7%) 0.045
Enthesite 85(65.4%) 43(64.2%) 0.867
Uveitis 56(43.1%) 17(25.4%) 0.015
FamilyhistoryofAS 31(23.8%) 14(20.9%) 0.640
Radiographicsacroiliitis
II 9(6.9%) 6(8.9%)
III 59(45.4%) 44(65.7%)
IV 62(47.7%) 17(25.4%) 0.01
Pureaxial involvement
28(21.5%) 12(17.9%) 0.549
Diseaseduration (years)
8.28±6.57 6.39±5.07 0.039
TNF˛inhibitors 59(45.4%) 16(23.9%) 0.003
Chi-squareandMann–Whitneytests. AS,ankylosingspondylitis.
Thepvalueinboldmeansstatisticallysignificant.
andBASDAI(p=0.016)remainedsignificantafterthemultiple adjustments.
Whenthepatientswereclassifiedbyremission(<1.3), mod-erate(≥1.3and<2.1),high(≥2.1and<3.5)orveryhigh(≥3.5)
Table5–Characteristicsofpatientswithankylosing spondylitisaccordingtoethnicity.
Variables Caucasian (n=96)
Non-Caucasian
(n=104)
p
Onset
Adult 78(81.3%) 90(86.5%)
Juvenile 18(18.7%) 14(13.5%) 0.308
Arthritis 47(49%) 52(0.5%) 0.883
Enthesitis 61(63.5%) 68(65.4%) 0.786
Uveitis 34(35.4%) 40(38.5%) 0.656
FamilyhistoryofAS 23(24%) 23(22.1%) 0.757
Radiographicsacroilitis
II 6(6.2%) 9(8.6%)
III 59(61.4%) 45(43.3%)
IV 31(32.3%) 50(48.1%) 0.037
Pureaxial involvement
21(21.8%) 21(20.2%) 0.770
BMI(kg/m2) 24.81±3.72 25.8±3.87 0.048
BASMI 3.89±2.18 4.65±2.36 0.023
AS,ankylosing spondylitis;BMI,bodymass index;BASMI,Bath ankylosingspondylitismetrologyindex.
Chi-squareandMann–Whitneytests.
Table6–Characteristicsofpatientswithankylosing spondylitisaccordingtohipinvolvement.
Variables Hipinvolvement p
Yes(n=26) No(n=174)
Age(years) 47.7±11.3 42.4±12.7 0.035 Timesince
diagnosis (years)
10.3±7.9 7.2±5.9 0.04
Diseaseduration (years)
23.4±9.6 15.6±8.8 <0.0001
BASDAI 1.20±1.43 2.42±2.06 0.001 BASMI 5.55±1.52 4.10±2.33 0.002 BASFI 5.03±2.52 3.61±2.48 0.011 mSASSS 27.33±24.46 17.77±21.80 0.035 ASDAS-CRP 1.55±0.88 2.14±1.09 0.009 HAQ-S 1.86±3.72 0.93±0.61 0.017
BASDAI,Bathankylosingspondylitisdiseaseactivityindex;BASMI, Bathankylosingspondylitismetrologyindex;BASFI,Bath anky-losingspondylitisfunctionindex;ASDAS,Ankylosingspondylitis disease activity score; mSASSS, modified Stokes ankylosing spondylitisspinescore.
Chi-squareandMann–Whitneytests.
Thepvalueinboldmeansstatisticallysignificant.
diseaseactivitybasedontheASDASscore,thefollowing fre-quencieswerefound:14%(n=28),31.5%(n=63),37%(n=74) and17.5%(n=35),respectively.
TheBASDAI score waspositively correlated withBASFI, ASDAS-Sed. rate, ASDAS-CRP, Sed. rate, CRP and HAQ-S and negatively correlated with mSSASS. Following the lin-ear multivariate regression, BASDAI remained significantly correlated with BASFI (p=0.001), HAQ-S (p=0.047), ASDAS-Sed.rate,ASDAS-CRPandSed.rate(p<0.0001),butnotCRP (p=0.247).
Discussion
TLR4polymorphisms399and299werenotmorefrequentin BrazilianpatientswithASincomparisontothehealthcontrols becausetherewerenostatisticaldifferencesbetweenpatient and control groups. Theinfluence ofTLR4 polymorphisms in the etiopathogenesis of infections, especially by Gram-negativebacteria,is wellknown,19 assuchpolymorphisms
resultinaphenotypethatislittleresponsivetoendotoxins
stemmingfrom the infectious process that cause aberrant
transduction of the signal in the presence of
microorgan-isms.However,anumberofauthorshaverecentlyquestioned
theinfluenceofpolymorphismsofthisreceptoronthe
pro-gression,severityandoutcomeofinfections,suggestingthat
factorsrelatedtothehostaremoreimportantthan
polymor-phismsperse.20,21
Bacterial or intracellular components can initiate the
inflammatoryprocessandtriggersensitizationtoan
endoge-nousantigenthroughmolecularmimicry,persistently
acti-vating adaptive innate immunity and perpetuating the
inflammatory process. The trigger may not necessarily be
pathogenic,butrathermakesofpartofthenormalresident
microbiotaandcanculminateinthedevelopmentofdiseases,
suchasAS,ingeneticallysusceptibleindividuals.2
ThereissomeevidenceofanassociationbetweenTLR
poly-morphisms (299and399)and AS,6,22,23 butthis association
has notbeen confirmed in other studies.24–26 Thesame is
true forAsp896Gly.27 TheS180Lpolymorphism ofan
adap-torproteinofTLR2and4(TIRAP),whichhasdemonstrated
toplayaprotectiveroleagainsttheoccurrenceofsystemic
lupuserythematosus,hasalsodemonstratednoassociation
withAS.28
Kyoetal.foundthatmutantmice(C3H/Hej)forTLR4did
notdeveloparthritisaftertheintra-jointinjectionofLPSfrom
E.coli,unlikethewildgroup.29Thishasraisedthehypothesis
thatthemutationinTLR4maydiminishtheintensityofthe
innateimmuneresponse,playingaprotectiveroleratherthan
promotingautoimmunity.
Thepresentdatashowthatthe299and399polymorphisms
ofTLR4arenotgeneticfactorsofgreatersusceptibilitytoAS,
whichisinagreementwitharecentmeta-analysisinvolving
data compiledfrom nine studieson this topic.30 Moreover,
it isimportantto pointout thatone ofthelargest genetic
studies(TheAustralo-Anglo-AmericanSpondyloarthritis
Con-sortium), involving more than two thousand patients of
EuropeandescentwithAS,alsofoundnoassociationbetween
TLRs and susceptibility to the disease.9 According to the
authorscited,themainassociationsoccurredwithtwodesert
genes(2p15and21q22)aswellaswithIL-23R,IL-1R2,ANTXR2
andERAP-1.
TheexpressionofTLR4isrelatedtotheinterfacebetween
the immunesystemand the environmentaswell asacute
andchronicinflammatoryresponsesinpatientswithAS,31–33
but polymorphisms doesn’t seem to be associated with a
greater risk of developing the disease.30 Population-based
studies involving healthy individuals indicate
heterogene-ity in the geographic distributionof thesepolymorphisms.
Thefrequencyrangesfrom4to10%inCaucasians34inthe
presentcohort,whileafrequencyof16%isreportedamong
Africans.35 There are no reports of this polymorphism in
Asians.25
ConsideringthatBrazilianpopulationishighlymixedwith
multipleethnic groups, wecannotassure similar resultsif
patientsfromotherpartsofthecountry,particularlyno
Euro-peanbackground,hadbeenincluded.
Onecannotdiscardthepossibilityofaweakassociation
thatmayberepresentativeinalargersamplesizethanthat
employedinthepresentstudy.However,astudycarriedoutin
theUnitedKingdominvolvingmorethan500patientswithAS
alsofoundnoassociation.6Thesamehasbeentrueforother
ethnicities,suchasHungarian,Finnish,Korean,Canadianand
Dutchpopulations.Anotherpointtoconsideristhelow
preva-lenceofhomozygousgenotypesfortheTLR4polymorphisms
inthepresentcohort,whichissimilartofindingsdescribedin
otherpopulations.
IthasbeendemonstratedthattransgenicratsforHLA-B27
donotdevelopinflammationofthe enthesesor intestines,
althoughtheseanimalsdevelopgenitalandskinlesionsunder
sterileconditions,demonstratingtheroleofinfectiousagents
asaninflammatorytrigger.36Moreover,theparticipationof
the environmentisadeterminantintheemergence ofthe
disease,asonlytwotofivepercentofthepopulationpositive
forHLA-B27developsAS.Basedonthisfact,theinteraction
inthepresentsample,butwasnotdemonstrated,asno
signif-icantdifferencesinthepolymorphismswerefoundbetween
thepatientsandhealthycontrols.Moreover,thenumberof
patientswithpolymorphismswasrelativelysmall.
Regarding clinical aspects, males exhibited moresevere
axialradiographicdamageandlongerdiseasedurationthan
females, which is similar to findings reported in another
Braziliancohort37aswellasinother populations,although
with no significant gender difference regarding peripheral
involvement.38However,thesefindingsshouldbeinterpreted
withcaution,sincetheonlysignificantdifferencesafter
con-trollingforconfoundingvariables,especiallydiseaseduration
andage,wereBASMIscore,whichwashigheramongmales,
andBMI,whichwashigheramongfemales.
Miscegenationlikelycontributedtothetwopeculiarities
found inthe present sample. Thefirst was the lesser
fre-quency of positivity for HLA-B27 (66%) in comparison to
anotherBraziliancohort(78.2%)37andstudiesinvolving
Cau-casianpopulations (more than 90%).39 Fifty-two percentof
thepresentcohortwasnon-Caucasian,whichbetterreflects
the Brazilianpopulationincomparisonto previousstudies
(75.5%Caucasianpatients).37Thesecondpeculiaritywasthe
greaterperipheralormixedinvolvement,whichhasalsobeen
reported inother mixed-race populations.39,40 Amongonly
theCaucasianpatientsinthepresentstudy,thefrequencyof
positivityforHLA-B27was75%,whichisveryclosetothe
fre-quencyreportedinapreviousBraziliancohort39aswellasthat
reportedinarecentFrenchstudy.41
Thepatientswithhighchronicityindexscoresgenerally
hadlowerdiseaseactivity indexscores,suchasthe
associ-ationbetweenhipinvolvementandbothaworseBASFIand
lowerBASDAIscore.Likewise,diseasedurationwascorrelated
withalowerBASDAIandhigherBASMIscore.Interestingly,the
BASDAIscorewascorrelatedwiththeotheractivitymarkers
(ASDAS-Sed.rateandCRP)aswellasworsefunctionality,but
thecorrelationwithCRP,whichisoneofthemost
standard-izedparametersforstudyinginflammatoryactivityinpatients
withAS,42wasnon-significant.
Asexpected, the presenceofHLA-B27 was significantly
associatedwithextra-articularmanifestations(uveitis)43and
lesserperipheralinvolvement,butnoassociationswerefound
withtheradiographicscoreoranyspecificASassessmenttool,
includingactivity,functionandmobility.Thisfindingreflects
themorerecentknowledgethatthisgeneticaspectisnotan
associatedfactorofapoorerprognosisregardingthe
forma-tionofnewbone.44
The prevalence of the juvenile form (16%) was similar
to that found in Caucasians (8.6–21%) Turks (13.4%) and
other Brazilian cohort (16%)45 as well as lower than the
ratesreportedforMexicans(28–54%)andKoreans(41.3%).46
Theinclusionofthesepatientsdidnotexert animportant
impact on the clinical, laboratory and imaging outcomes
analyzed, whichis inagreement withthe notionthat this
subgroup is part of the same spectrum of the disease.In
contrast,someauthors reportlesseraxialinvolvementand
agreaterproportionofperipheralinvolvement,especiallythe
knees,incomparisontoonsetofthediseaseaftertheageof
16years.37,45,46
The present study has limitations that should be
addressed, such as the sample size for studies on gene
polymorphismsandthehigherproportionofCaucasian
indi-viduals in this cohort, what may not reflect Brazilian AS
population.OurdatasuggestthattheseTLR4variationsare
unlikelytoplayaroleintheetiopathogenesisofAS.This
find-ing,however,doesnotexcludethepossibilitythatfunctional
abnormalitiesoftheTLRsorothermoleculesclosely
associ-atedwiththeTLRssignalingareimportantinthepathogenesis
ofspondylarthropathies.
Funding
Supported byaresearch grant from theBrazilian fostering
agencyFundac¸ãodeAmparoàPesquisadoEstadodeSãoPaulo
(FAPESP2011/05517-6).
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
Acknowledgment
We thank Rheumatology and Nephrology Division, Federal
UniversityofSãoPaulo(UNIFESP/EPM).
r
e
f
e
r
e
n
c
e
s
1.MedzhitovR,Preston-HurlburtP,JanewayCAJr.Ahuman homologueoftheDrosophilaTollproteinsignalsactivationof adaptiveimmunity.Nature.1997;388(6640):394–7.
2.PollanenR,SillatT,PajarinenJ,LevonJ,KaivosojaE, KonttinenYT.MicrobialantigensmediateHLA-B27diseases viaTLRs.JAutoimmun.2009;32(3-4):172–7.
3.Pacheco-TenaC,ZhangX,StoneM,Burgos-VargasR,Inman RD.Innateimmunityinhost–microbialinteractions:beyond B27inthespondyloarthropathies.CurrOpinRheumatol. 2002;14(4):373–82.
4.TakedaK,KaishoT,AkiraS.Toll-likereceptors.AnnuRev Immunol.2003;21:335–76.
5.LeaverSK,FinneySJ,Burke-GaffneyA,EvansTW.Sepsissince thediscoveryofToll-likereceptors:diseaseconceptsand therapeuticopportunities.CritCareMed.2007;35(5):1404–10.
6.LamarqueD,NhieuJT,BrebanM,BernardeauC,
Martin-GarciaN,SzepesZ,etal.Lymphocyticinfiltrationand expressionofinduciblenitricoxidesynthaseinhuman duodenalandcolonicmucosaisacharacteristicfeatureof ankylosingspondylitis.JRheumatol.2003;30(11):2428–36.
7.DeRyckeL,KruithofE,VandoorenB,TakPP,BaetenD. Pathogenesisofspondyloarthritis:insightsfromsynovial membranestudies.CurrRheumatolRep.2006;8(4):275–82.
8.SparksJA,CostenbaderKH.Geneticsenvironment,and gene–environmentinteractionsinthedevelopmentof systemicrheumaticdiseases.RheumDisClinNorthAm. 2014;40(4):637–57.
9.ReveilleJD,SimsAM,DanoyP,EvansDM,LeoP,PointonJJ, etal.Genome-wideassociationstudyofankylosing
spondylitisidentifiesnon-MHCsusceptibilityloci.NatGenet. 2010;42(2):123–7.
11.RudwaleitM,vanderHeijdeD,LandewéR,ListingJ,AkkocN, BrandtJ,etal.ThedevelopmentofAssessmentof
SpondyloArthritisinternationalSocietyclassificationcriteria foraxialspondyloarthritis(partII):validationandfinal selection.AnnRheumDis.2009;68(6):777–83.
12.GarrettS,JenkinsonT,KennedyLG,WhitelockH,GaisfordP, CalinA.Anewapproachtodefiningdiseasestatusin ankylosingspondylitis:theBathAnkylosingSpondylitis DiseaseActivityIndex.JRheumatol.1994;21(12):2286–91.
13.MachadoP,LandeweR,LieE,KvienTK,BraunJ,BakerD,etal. AnkylosingSpondylitisDiseaseActivityScore(ASDAS): definingcut-offvaluesfordiseaseactivitystatesand improvementscores.AnnRheumDis.2011;70(1):47–53.
14.CalinA,GarrettS,WhitelockH,KennedyLG,O’HeaJ,Mallorie P,etal.Anewapproachtodefiningfunctionalabilityin ankylosingspondylitis:thedevelopmentoftheBath AnkylosingSpondylitisFunctionalIndex.JRheumatol. 1994;21(12):2281–5.
15.vanderHeijdeD,LandeweR,FeldtkellerE.Proposalofa lineardefinitionoftheBathAnkylosingSpondylitisMetrology Index(BASMI)andcomparisonwiththe2-stepand10-step definitions.AnnRheumDis.2008;67(4):489–93.
16.CreemersMC,FranssenMJ,van’tHofMA,GribnauFW,vande PutteLB,vanRielPL.Assessmentofoutcomeinankylosing spondylitis:anextendedradiographicscoringsystem.Ann RheumDis.2005;64(1):127–9.
17.JeffreysAJ,WilsonV,TheinSL.Individual-specific ‘fingerprints’ofhumanDNA.Nature.1985;316(6023):76–9.
18.LorenzE,FreesKL,SchwartzDA.DeterminationoftheTLR4 genotypeusingallele-specificPCR.Biotechniques.
2001;31(1):22–4.
19.MedzhitovR.Toll-likereceptorsandinnateimmunity.Nat RevImmunol.2001;1(2):135–45.
20.DraismaA,DorresteijnM,PickkersP,vanderHoevenH.The effectofsystemiciNOSinhibitionduringhuman
endotoxemiaonthedevelopmentoftolerancetodifferent TLR-stimuli.InnateImmun.2008;14(3):153–9.
21.JessenKM,LindboeSB,PetersenAL,Eugen-OlsenJ,BenfieldT. CommonT.N.F-alpha,IL-1beta,PAI-1,uPA,CD14andTLR4 polymorphismsarenotassociatedwithdiseaseseverityor outcomefromGramnegativesepsis.BMCInfectDis. 2007;7:108.
22.McCormackWJ,ParkerAE,O’NeillLA.Toll-likereceptorsand NOD-likereceptorsinrheumaticdiseases.ArthritisResTher. 2009;11(5):243.
23.SnelgroveT,LimS,GreenwoodC,PeddleL,HamiltonS, InmanR,etal.Associationoftoll-likereceptor4variantsand ankylosingspondylitis:acase-controlstudy.JRheumatol. 2007;34(2):368–70.
24.GergelyPJr,BlazsekA,WeiszharZ,PazarB,PoorG.Lackof geneticassociationoftheToll-likereceptor4(TLR4) Asp299GlyandThr399Ilepolymorphismswith spondylarthropathiesinaHungarianpopulation. Rheumatology(Oxford).2006;45(10):1194–6.
25.NaKS,KimTH,RahmanP,PeddleL,ChoiCB,InmanRD. AnalysisofsinglenucleotidepolymorphismsinToll-like receptor4showsnoassociationwithankylosingspondylitis inaKoreanpopulation.RheumatolInt.2008;28(7):627–30.
26.AdamR,SturrockRD,GracieJA.TLR4mutations(Asp299Gly andThr399Ile)arenotassociatedwithankylosing
spondylitis.AnnRheumDis.2006;65(8):1099–101.
27.vanderPaardtM,CrusiusJB,deKoningMH,MorreSA,vande StadtRJ,DijkmansBA,etal.Noevidenceforinvolvementof theToll-likereceptor4(TLR4)A896GandCD14-C260T polymorphismsinsusceptibilitytoankylosingspondylitis. AnnRheumDis.2005;64(2):235–8.
28.CantaertT,StoneMA,terBorgM,MoggR,DeVriesN,Wilson AG,etal.Afunctionalpolymorphismof
TIR-domain-containingadaptorproteinisnotassociated withaxialspondyloarthritis.AnnRheumDis.2008;67(5): 720–2.
29.KyoF,FutaniH,MatsuiK,TeradaM,AdachiK,NagataK,etal. Endogenousinterleukin-6,butnottumornecrosisfactor alpha,contributestothedevelopmentoftoll-likereceptor 4/myeloiddifferentiationfactor88-mediatedacutearthritis inmice.ArthritisRheum.2005;52(8):2530–40.
30.XuWD,LiuSS,PanHF,YeDQ.LackofassociationofTLR4 polymorphismswithsusceptibilitytorheumatoidarthritis andankylosingspondylitis:ameta-analysis.JointBoneSpine. 2012;79(6):566–9.
31.DeRyckeL,VandoorenB,KruithofE,DeKeyserF,VeysEM, BaetenD.Tumornecrosisfactoralphablockadetreatment down-modulatestheincreasedsystemicandlocalexpression ofToll-likereceptor2andToll-likereceptor4in
spondylarthropathy.ArthritisRheum.2005;52(7):2146–58.
32.AssassiS,ReveilleJD,ArnettFC,WeismanMH,WardMM, AgarwalSK,etal.Whole-bloodgeneexpressionprofilingin ankylosingspondylitisshowsupregulationoftoll-like receptor4and5.JRheumatol.2011;38(1):87–98.
33.YangZX,LiangY,ZhuY,LiC,ZhangLZ,ZengXM,etal. IncreasedexpressionofToll-likereceptor4inperipheral bloodleucocytesandserumlevelsofsomecytokinesin patientswithankylosingspondylitis.ClinExpImmunol. 2007;149(1):48–55.
34.CarvalhoA,MarquesA,MacielP,RodriguesF.Studyof disease-relevantpolymorphismsintheTLR4andTLR9genes: anovelmethodappliedtotheanalysisofthePortuguese population.MolCellProbe.2007;21(4):316–20.
35.MockenhauptFP,CramerJP,HamannL,StegemannMS, EckertJ,OhNR,etal.Toll-likereceptor(TLR)polymorphisms inAfricanchildren:commonTLR-4variantspredisposeto severemalaria.ProcNatlAcadSciUSA.2006;103(1): 177–82.
36.TaurogJD,RichardsonJA,CroftJT,SimmonsWA,ZhouM, Fernandez-SueiroJL,etal.Thegermfreestateprevents developmentofgutandjointinflammatorydiseasein HLA-B27transgenicrats.JExpMed.1994;180(6):2359–64.
37.Sampaio-BarrosPD,BertoloMB,KraemerMH,NetoJF,Samara AM.Primaryankylosingspondylitis:patternsofdiseaseina Brazilianpopulationof147patients.JRheumatol.
2001;28(3):560–5.
38.LeeW,ReveilleJD,DavisJCJr,LearchTJ,WardMM,Weisman MH.Aretheregenderdifferencesinseverityofankylosing spondylitis?ResultsfromthePSOAScohort.AnnRheumDis. 2007;66(5):633–8.
39.GallinaroAL,VenturaC,SampaioBarrosPD,GoncalvesCR. Spondyloarthritis:analysisofaBrazilianseriescompared withalargeIbero-Americanregistry(RESPONDIAgroup).Rev BrasReumatol.2010;50(5):581–9.
40.KohemCL,BortoluzzoAB,Gonc¸alvesCR,BragadaSilvaJA, XimenesAC,BértoloMB,etal.Profileoftheuseofdisease modifyingdrugsintheBrazilianRegistryof
Spondyloarthritides.RevBrasReumatol.2015;55(1):48–54.
41.CostantinoF,TalpinA,Said-NahalR,GoldbergM,HennyJ, ChiocchiaG,etal.Prevalenceofspondyloarthritisin referencetoHLA-B27intheFrenchpopulation:resultsofthe GAZELcohort.AnnRheumDis.2015;74:689–93.
42.MachadoP,LandeweR.Spondyloarthritis.Isittimetoreplace BasdaiwithAsdas?NatRevRheumatol.2013;9(7):388–90.
43.KhanMA.Update:thetwentysubtypesofHLA-B27.CurrOpin Rheumatol.2000;12(4):235–8.
45.DuarteAP,MarquesCDL,BortoluzzoAB,Gonc¸alvesCR,da SilvaJA,XimenesAC,etal.Epidemiologicprofileof
juvenile-onsetcomparedtoadultonsetspondyloarthitisina largeBraziliancohort.RevBrasReumatol.2014;54(6): 424–30.
46.BaekHJ,ShinKC,LeeYJ,KangSW,LeeEB,YooCD,etal.