ww w . r e u m a t o l o g i a . c o m . b r
REVISTA
BRASILEIRA
DE
REUMATOLOGIA
Review
article
Critical
revision
of
the
medical
treatment
of
gout
in
Brazil
Valderilio
Feijó
Azevedo
a,∗,
Maicon
Piana
Lopes
a,
Nathan
Marostica
Catholino
a,
Eduardo
dos
Santos
Paiva
a,
Vitor
Andrei
Araújo
a,
Geraldo
da
Rocha
Castelar
Pinheiro
baUniversidadeFederaldoParaná,HospitaldeClínicas,DepartamentodeClinicaMédica,Curitiba,PR,Brazil
bUniversidadedoEstadodoRiodeJaneiro,Servic¸odeReumatologia,RiodeJaneiro,RJ,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received20October2015 Accepted20June2016 Availableonline6April2017
Keywords:
Gout
Hyperuricaemia Treatment Goutyarthritis Medicines
a
b
s
t
r
a
c
t
Goutisconsideredthemostcommonformofinflammatoryarthritisinmenover40years. Theauthorspresentabriefreviewofthecurrenttreatmentofgoutanddiscusstheexisting pharmacologicallimitationsinBrazilforthetreatmentofthisdisease.Althoughallopurinol isstillthemaindrugadministeredfordecreasingserumlevelsofuricacidingoutpatients inthiscountry,theauthorsalsopresentdatathatshowagreatopportunityfortheBrazilian drugmarketforthetreatmentofhyperuricemiaandgoutandespeciallyforpatientsusing privateandpublic(SUS)healthcaresystems.
©2017ElsevierEditoraLtda.ThisisanopenaccessarticleundertheCCBY-NC-ND license(http://creativecommons.org/licenses/by-nc-nd/4.0/).
Revisão
crítica
do
tratamento
medicamentoso
da
gota
no
Brasil
Palavras-chave:
Gota
Hiperuricemia Tratamento Artritegotosa Medicamentos
r
e
s
u
m
o
Agotaéconsideradaaformamaiscomumdeartriteinflamatóriaemhomensacimade40 anos.Osautoresapresentamumabreverevisãosobreotratamentoatualdagotaediscutem aslimitac¸õesfarmacológicasexistentesnoBrasilparaotratamentodestaenfermidade. Apesardequeoalopurinolaindasejaaprincipalmedicac¸ãoparaareduc¸ãodosníveisde uricemiadepacientescomgotanopaís,osautorestambémapresentamdadosque apon-tamparaumagrandeoportunidadeparaomercadofarmacológicobrasileiroemrelac¸ãoao tratamentodahiperuricemiaedaartritegotosaeespecialmenteparapacientesusuários desistemasprivadosdesaúdeedoSUS(SistemaÚnicodeSaúde).
©2017ElsevierEditoraLtda.Este ´eumartigoOpenAccesssobumalicenc¸aCC BY-NC-ND(http://creativecommons.org/licenses/by-nc-nd/4.0/).
∗ Correspondingauthor.
E-mail:[email protected](V.F.Azevedo).
http://dx.doi.org/10.1016/j.rbre.2017.03.002
Introduction
Gout is a disease characterized by an accumulation of monosodiumurate(MSU)crystalsinthejoints,synovial tis-sue, bone,and skin, regardlessofthe presenceor absence ofclinicalmanifestations.1,2Thisaccumulationisaresultof
apersistenthyperuricemia.1TheMSUcrystalsarethesolid
formofuricacid,thefinalproductofpurinemetabolism;these crystalscanaccumulateinorganictissue.3 Thepurinesare
aresult ofthe breakdown ofmononucleotides, substances derivedfromthosenitrogenousbasesthatmakeupDNAand RNA.Inthebiologicalprocessofurateproduction,the com-poundsare,intheir laststages, metabolized intoxanthine which,inturn,isirreversiblyoxidizedtoproduceuricacid bytheactionoftheenzymexanthineoxidase,4,5which
com-prisesthearsenal ofperoxisomesinthe majorityofcells.3
Circulatinguricacid(UA)inthebloodmaintainits physiologi-callevelsatconcentrationsaround6.0mg/dL,6andtheexcess
iseliminatedbythekidneys.7Underphysiologicalconditions,
circulatingUAcantakepartinantioxidant,oxidant,and pro-inflammatoryreactions;suchparticipationsaremoreevident whencirculatingUAisfoundinhighserumconcentrations.5
Hyperuricemia is definedas the high urate serum con-centration,atapproximately6.8mg/dL,whichisthelimitof solubilityoftheurate.1Abovethislevelofsolubility,MSU
crys-talsmay accumulate inthe tissues, especiallyina caseof chronic,untreatedhyperuricemia.Inadditiontodisturbances inthegenerationorclearanceofuricacid,hyperuricemiacan beinitiatedoracceleratedinpatientswithkidney and car-diactransplant,astheseareusuallyassociatedwithchronic kidney disease and the use ofloop diuretics.8 UA may be
associated with different multifactorial disorders, whether dependentlyorindependently.Thereisadirectrelationship betweenUAlevelsanddevelopmentandprogressionof car-diovasculardisease.9–15 Thereisalsoevidenceofapositive
associationbetweenUAlevelsandhypertension,12,16 kidney
disease,16andtheriskofcoronaryevents.17Furthermore,
dis-ordersofpurinemetabolismorintheprocessofelimination ofuricacid,oranincreaseinproteinintake18maycontribute
totheelevationofUA.
Goutymanifestations may occur in threephases: acute crises(1),intercriticalperiod(2),whichoccursbetweencrises, iscompletelyasymptomatic,andhasavariableduration.At thebeginningofthediseasethisperiodcanlastforyears; how-ever,ittendstobecomegraduallyshorterwiththeprogression ofthedisease.Chronicarthropathy (3), themostadvanced stage,ischaracterizedbymultipleand/orpersistentcrises.8
Severalevents can triggeracuteattacks ofgout, includ-ing excessive alcohol intake, metabolicstress (such as the observedinacutemyocardialinfarctionorsurgery)or,more commonly, sudden changes in UA levels, as occurs after thebeginning uratereductiontherapy, leadingtoa resorp-tion of MSU crystals.8 Acute gout is recognized as one of
the most painful experiences, comparable to labor pains and visceral colic, such asnephritic colic.19 Aflare begins
whenmacrophagespresentinthesynovialfluidphagocyte thecrystalsandinitiatetheinflammatorycascade,releasing mediatorsandpromotingneutrophilchemotaxis.20
The classical clinical presentation of gout is an acute inflammatoryarthritis,whichusuallyisasingle-joint, recur-rent,intense,self-limitingprocess.8,19,21Inabout50%ofcases,
arthritisoccursinthefirstmetatarsophalangealjoint,being known aspodagra.12,22,23 Thejoints ofthe ankle and knee
arecommonsitesofarthritis.8Oligoarticularandpolyarticular
impairmentislesscommon,24butthiscanoccurinpatients
withlong-standing,untreatedgout,aswellasinpatientswith amarkedandsignificantreductionofuricacidlevels,arising from theurate-loweringtherapy. Someprodromes,suchas pain,discomfort,andlimitationofmovement,mayindicate theonsetofanacutegoutepisode.8
TophiaremacroscopiccollectionsvisibleMSUcrystalsby clinicalexamination,andusuallyindicatealong-standing dis-easewhichhasnotbeentreated.25,26Thepresenceoftophiis
relatedtoanincreaseofstructuraldamage27andlossofjoint
function,26anditsoccurrenceisdirectlyrelatedtoincreased
serum urate levels.25 Theurate-loweringtherapy is
associ-atedwithareductionoftophiformation,aswell astothe regression ofexistingformations.28Chronicgout alsoleads
toarestrictionofjointmobility,jointswelling,anda radiolo-gicallyapparentdeformity.8Well-defined‘punched-out’type
lesions,especiallywhenshowinganeggshelledge (overhang-ing bonyedges–Martelsignal)are characteristicradiologic findingsindicatingalongstanding,serious,untreatedchronic gout.29
Epidemiology
Despitethewidevariationofdatabetweendifferentcountries, itisbelievedthattheprevalenceofgouthasincreasedover thelastsixyears.16TheUSstudyNationalHealthInterview
foundanincreaseintheprevalenceofself-reportedgout,from 4.8/1000in196730to9.4/199631in1996.AccordingtotheJohns
HopkinsPrecursorsStudy,in1991theincidenceofgoutinthe UnitedStateswasestimatedat1.73per1000population.32In
Brazil,thereisalackofepidemiologicalstudiesinthisarea. The reasons for the apparent increasein the incidence of gout over the last few years have not been clarified, although several risk factors have already been described. Hyperuricemiaisdirectlylinkedtogout,since10%ofpatients withhyperuricemiadevelopgout,and 90%ofpatientswith gouthavehighconcentrationsofUA.33TheFraminghamstudy
indicatedandose-dependentincreaseintherelativeriskto goutdevelopmentwithincreasingUAlevels.Otherrisk fac-torsrelatedtogoutindicatedinthesamestudywerealcohol intake,bodymassindex,andbloodpressure.16,24,32,34–39Itis
known that both overweight and obesity can increase the endogenousproductionofuricacid.40Therelativeriskof
inci-dence of gout is 1.95 in menwith body mass index (BMI) between25and29.9kg/m2,comparedtotherelativeriskof
1.00whenBMIisbetween21and22.9kg/m2.Thesamestudy
notedanincrease intherelativerisk,to2.97,whenBMIis greater than35kg/m2.41Other studieshaveaddedthe
Diagnosis
Thegoldstandardinthediagnosisofgoutistheobservation ofmonosodiumuratecrystalsundercompensatedpolarized lightmicroscopy;forthisexamination,thesecrystalsexhibit negativebirefringence.Thesampleshouldbepreferably col-lectedfromnewlyaffectedjoints,aswellasfrompreviously affectedjoints.12
Nonetheless,inatypicalclinicalpresentation, the clini-caldiagnosisisreasonablyaccurate,beingacceptableinthe absence of microscopy or rheumatologists. This diagnosis, however,isnotdefinitive.12
Althoughacommonclinicalpractice,theuseof classifica-tioncriteriafordiagnosticpurposeshasevolvedoverthelast decade.46Nevertheless,recentlyadiagnosticcriterionforeasy
use,developedforprimarycare,showedgoodperformancein secondarycare,withagoodpositivepredictivevalueinthe diagnosisofgoutwhenthe analysisofsynovialfluidisnot available.47
AfterverificationofthepresenceofMSUcrystalsinthe jointenvironment,onemustquantifythisdeposition,aswell asitsextentandtheinducedstructuraldamage.Inaddition tohistory and physicalexamination, imagingstudies such asX-rays,ultrasound,anddualemissioncomputed tomogra-phycanbeusefultoassessthechronicphaseofthedisease and the damage done.48 The differential diagnoses should
alwaysbeconsidered,especiallyinoligoarticularor polyartic-ularpresentation,whichmaymimicseveraldiseases,suchas rheumatoidarthritisandspondyloarthropathies.Inthecase ofamonoarticularpresentation,septicarthritisshouldalways bediscarded.8
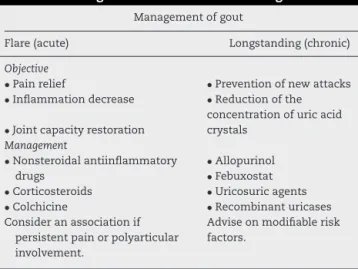
Treatment
Basically,thetreatmentofgoutcanbedividedintotwostages: acute flare management and long-term therapy (Table 1). Inthe first stage,it is critical torelievethe pain, reducing inflammation and joint impairment; for this purpose, the patient must be treatedwith anti-inflammatory agents; in thesecondstage,theaimistodecreaseUAconcentrations,
Table1–Managementofacuteandchronicgout.
Managementofgout
Flare(acute) Longstanding(chronic)
Objective
•Painrelief
•Inflammationdecrease
•Jointcapacityrestoration
•Preventionofnewattacks
•Reductionofthe
concentrationofuricacid crystals
Management
•Nonsteroidalantiinflammatory drugs
•Corticosteroids
•Colchicine
Consideranassociationif persistentpainorpolyarticular involvement.
•Allopurinol
•Febuxostat
•Uricosuricagents
•Recombinanturicases Adviseonmodifiablerisk factors.
aswellastopreventnewcrises.Anti-inflammatoriesareno longerappropriateinthisstage,wherebothpharmacological measures and non-pharmacological procedures should be adopted.28,49–53
Regarding drugprescription,bothfortheacute manage-mentandlong-termtherapy,somelvariablescaninfluence the choiceofmedication,suchasavailability,cost,efficacy, andclinicalindicationsduetopotentialcomorbiditiesofthe patient.Hypertension,cardiovasculardisease,chronickidney disease,anddiabetes areexamples ofcommonlyoccurring conditions associated with gout that should betaken into consideration beforethe implementationofanappropriate treatment.54
Management
of
acute
flares
For low-or moderate-intensityattacks (painindex≤6ona
scalefrom0to10)involvingonetothreesmalljointsorone totwolargejoints,monotherapyisrecommended.Itcanbe initiatedwithbothnonsteroidalanti-inflammatorydrugsand colchicineorcorticosteroids.55
The nonsteroidalanti-inflammatory drugs(NSAIDs) are the most widely used therapy for early treatment of acute gouty arthritis56 and, due to the intensity of crisis, these
agentscanbeadministeredinmaximumdosages.6Thisclass
includes drugs of different pharmacokinetic and pharma-codynamic properties,but withasimilaraction:theyexert an anti-inflammatory function by inhibiting the cyclooxy-genase (COX) enzymes, which catalyze the conversion of arachidonic acid derivedfrom membrane phospholipids to prostaglandins.54,57 NSAIDs can be non-selective (such as
ibuprofen,indomethacin,andnaproxen), withinhibitionof COX-1andCOX-2,orselective,withinhibitionofCOX-1(such asaspirin)orCOX-2(likecelecoxib andother “coxibs”).54,58
Among the NSAIDscommonly prescribed topatients with gout, indomethacinhasbeentraditionallyused.59However,
thereisevidencethatanyNSAIDhasasimilareffectin reduc-ing the acute inflammatory activity in gout.19 Since gouty
patientsoftenhavecomorbidconditionsandtheprescribed dosagesofanti-inflammatorydrugsare high,protonpump inhibitors can be used inorder to prevent gastrointestinal damage,suchasulceration,bleeding,andperforation.Inthis context,COX-2inhibitorsmayhavethesameeffectas COX-1 inhibitors, and may be a good option for patients with gastrointestinal disorders, such as gastroesophageal reflux diseaseandpepticulcer.60However,duetotheirrenaland
car-diovasculartoxicity,theuseofthesedrugsshouldbecarefully consideredandindividualized.
Colchicine, an alkaloid derived from meadow saffron extracts,54isalsorecommendedinfirst-linetreatment.6,54,55
Severaleffectsofthissubstancehavealreadybeenreported, being the inhibition ofcell division(by bindingto tubulin of microtubules and prevent spindle formation) the best-known effect.54 In gout, this function directly affects the
activity ofneutrophilsbypreventing diapedesis,Lysosomal mobilization, and degranulation, which releases not only pro-inflammatory substances, but also leukocytic chemo-tactic agents.61 Moreover,colchicine inhibitsthe formation
important for the processing and release of inflammatory cytokinesIL-1andIL-18),inducedbycrystalsofuricacid.57,61
Themetabolismofcolchicineisprocessedbythecytochrome P450 family and thus this drug may interact with other drugsmetabolizedbythispathway.54Moreover,therenaland
hepaticfunctionofpatientsmustbeassessedbeforetheuse ofcolchicine.19Regardingdosage,therecommendationsofthe
EuropeanLeagueAgainstRheumatism–EULAR6andthoseof
theAmericanCollegeofRheumatology–ACRaredifferent.62
Theformer organizationrecommendsa maximumdose of 0.5mg threetimes aday; conversely, the latterproposes a dose of1.2g, followed byanother administrationof0.6mg onehourlater,repeatedat12h.ACRguidelinesalsopropose thatthetreatmentwithcolchicinewillleadtobetterresultsif startedwithin24hoftheonsetofsymptoms,notexceeding 36handremaininguntiltheattackissuppressed.Intravenous administration of colchicine is extremely toxic and is not recommended.6
Anti-inflammatorysteroids,orsystemiccorticosteroids(SC), haveseveralknowneffectsinsuppressionofthe inflamma-toryresponse,suchassuppressionoftheimmuneresponse, inhibitionofprostaglandins andleukotrienes,direct inhibi-tionofpro-inflammatorytranscriptionfactors,andinhibition ofcytokinessuchasIL-1,IL-6,IL-8,andTNF-␣.54,63Inaddition,
SC upregulate genes for anti-inflammatory factors, involv-ing phospholipase A2 blockers.58 As to the administration
route,thereareseveraloptionsforcorticosteroids–oral, intra-venous, intramuscular or intra-articular.19 The oral use is
indicatedwhenthereisafailureintreatmentwithcolchicine and/orNSAIDs,orwhenthesedrugsarenotindicated.54
Evi-dence are lacking as to what is the ideal dosage for this treatment;Janssensetal.demonstratedthatadailydoseof prednisolone35mgisequivalentto500mgofnaproxentwice daily.64Intramuscularadministrationofsteroidsisanoption;
butitsuseislimitedtointra-hospitalenvironmentand, more-over,thereisalsonoconsensusonthedosetobeused.19An
intra-articularcorticosteroidinjectionafterjointaspirationis consideredtheidealtreatmentforacutegout,sincethepain reliefoccursquickly(duetoadecreaseintheintra-articular pressurecausedbytheinflammatoryprocess),andthe corti-costeroid,withitsminimalsystemicabsorption,promotesa largerlocaleffect.6
Corticotropin(ACTH)isahormonesecreted bythe pitu-itary gland,which directly influences steroidssecretion by the adrenal glands, stimulating the production ofcortisol, corticosterone,andandrogens.54,65 Forawhile,ACTH
repre-sentedanalternativetherapyinacutegoutyarthritiswhen administeredinintramuscularinjections.Themechanismof actionofthishormoneinpatientssufferinggoutisnot estab-lished;Itisknown thatthe ACTHmay inhibitperipherally goutyinflammation,byactivatingthemelanocortinreceptor type3,whichwouldbeasecondaryeffect.65,66Axelrodand
Prestonconcludedthat patientstreatedwithintramuscular ACTHexperiencedpainrelieffasterthanthosetreatedwith oralindomethacinwhencomparingtheeffectsofthesedrugs inpatientswithanacutegoutattack.67
AnotherpossibletherapytargetisIL-1,acytokineinvolved ingoutyinflammation.MSUcrystalsstimulateIL-1 produc-tionandsecretioninsynovialmonocytesandmononuclear cells,andactivatetheinflammasomeNLRP3,thusjustifying
thevalidityofinhibitingthisinterleukininthetreatment.68–70 IL-1inhibitorsareanakinra,canakinumab,andrilonacept,all of which are biological.20,70,71 These drugs should be
pre-scribed onlywhenall previouslyadministeredagentswere discardedbyfailureorinabilitytotreat.Nonetheless,thisis stillahigh-cost treatment,especiallywhencomparedwith NSAIDs,andthesethemedicationsarenotlicensedinmost countries.Finally,thereisevidencethaticepacksappliedto theinflamedsitecanbeanadjuvantprocedurefor pharma-cologicaltreatment.72
Long-termgouttherapyhastwogoals:toreducecirculating levels ofurate toalevel belowthe saturation point, keep-ingUA<6.0mg/dL,andtopreventtheformationofnewurate crystals.6Themosteffectivemethodforachievingthisgoalis
theurate-loweringtherapy(ULT)andseveralclassesofdrugs canbeusedincurrentclinicalpractice.However,ULTshould notberestrictedonlytodrugs,asthenon-pharmacological treatmentofgoutplaysasignificantroleinthepreventionof newcrises.73
Thefirststepaftertheresolutionofanacutegouty arthri-tis episode is to explain to the patient the nature of the attack, making him/her awareofthe possible etiology and the changes in lifestyle that can prevent new crisesfrom happening. Therefore,it isessentialtoobtainagood med-icalhistory,centeredonfamily history,clinicalhistory, and lifestylehabits. Therefore,itcanbesaidthatthetreatment ofgoutinitschronicformisanindividualizedtask.74
There-fore,patientsshouldbeinformedofallnon-modifiablerisk factorsthatcanaccelerategout, suchasage,ethnicity,and gender.Asformodifiableriskfactors–suchashyperuricemia, obesity,hypertension,dyslipidemia,ischemiccardiovascular disease,diabetesmellitus,chronickidneydisease,dietary fac-tors, and abrupt changes inthe levels ofurate – guidance on controlmeasures, suchas reducing bodymass, alcohol consumption,consumptionofpurine-richfoods,suchasred meat and seafood, and consumption of fructose-rich bev-erages and foods, such as apple and orange, is absolutely critical.18,19,75
Theaforementionedcomorbiditiesalsodeserveattention andshouldbecontrolled.Arelevanttopicistheattentionto themedicationsusedbythepatient.Diuretics,boththiazides andespeciallyloopdiuretics,increasetheriskofincidenceof gout, includingpatientswithstabledisease.76Furthermore,
Choi,H.etal.,inanobservationalcohortstudy,demonstrated thatbeta-blockers,angiotensinconvertingenzymeinhibitors, andangiotensinreceptorblockers(withtheexceptionof losar-tan)increasethelevelofUAbyreducingtherenalexcretion ofuricacid,whereaslosartanandcalciumchannelblockers increasetherenalexcretionofuricacid.77
The exact time to start ULTis not well established; by consensus,ULTshouldbestartedwhenthegoutiswell estab-lishedand inarelativelyserious level, i.e.recurrentattack (twoormoreper year),or aflareinapatientwithchronic kidneydiseasestage2orhigher;thepresenceofoneormore tophionclinicalorimagingexamination62;jointdamage,or
nephrolithiasis.Severalstudiesshowthatoncestarted,ULT shouldnotbediscontinuedbecauseoftheriskofrecurrence ofacutearthritis.78,79
xanthineoxidase(XO)catalyzesthetwolaststagesofpurine metabolism in humans: conversion from hypoxanthine to xanthineandfromxanthinetourate.4,80Therearetwodrugs
capableofinhibitingthisenzyme:allopurinolandfebuxostat.
Allopurinolis considered as afirst-line drugin reducing urate,i.e., the drug is effectivein up to90% ofpatients.73
Its use is well established and is administered only once daily;furthermore,itisinexpensiveandrelativelysafe.6Thus,
it is the most prescribed drug in ULT.81 Both allopurinol
as its main active metabolite, oxypurinol, are non-specific competitive inhibitors of hypoxanthine oxidase and xan-thineoxidase,respectively.Therefore,bothdrugsareableto reducetheproductionofuricacid,leadingtoanincreaseof xanthineand hypoxanthine concentrationsin extracellular fluid.73,81,82 Oxypurinol accounts for 90% of the
bioavail-ability of allopurinol and is preferably excreted via the kidneys.81
Theallopurinoldoseadjustmentshouldbeindividualized, starting with100mg and increasingin that same amount each month until urate levels are under control (target foruricemia≤6.0mg/dL,orideally 5.0mg/dL)with a
maxi-mumof900mg.55Thebestestablisheddailydoseis300mg;
however, inthis dosageapproximately half ofthepatients achieve disease control.83,84 Side effects appear in up to
20% of patients, with gastrointestinal intolerance, nausea, and cutaneous rash being the most common events.85 In
1984,hypersensitivitysyndrometoallopurinol(nowknownas DRESS–drugreactionorrashwitheosinophiliaandsystemic symptoms)85wasdescribed,involvingfever,cutaneousrash,
eosinophilia, hepatitis, progressive renal failure and death duetomultiple-organvasculitis.58Steven-Johnsonsyndrome
andtoxicepidermalnecrolysiscanjointhisclinicalpicture, or occur in isolation (known as SCAR – severe cutaneous adversereactions).85Thesereactionsoccurmorefrequentlyin
patientswithpre-existingrenalimpairment,ortakingdiuretic drugs.58Thereisalsoevidenceofincreasedriskfor
hypersen-sitivityreactionsinthosepatientswhostartedthistherapy recently.85
FebuxostatisahighlyspecificinhibitorofXO;thisdrugis derivedfrom tiazolocarboxycilicacid.Febuxostatselectively inhibitsboththeoxidizedandreducedformofXOby compet-itiveandnon-competitivemechanisms.Itsmetabolismoccurs mainlyintheliver,withalittleamountexcretedinurine34,41,86
(around10%).87 Due tothis feature, febuxostat wasshown
tobeapromisingalternativeforpatientswithchronic kid-neydisease.Moreover,comparedwithallopurinol,febuxostat doesnotrequiredoseadjustmentsandhasfewerdrug inter-actionsthat may limit efficacy or safety.34,41,86 Amongthe
identifiedsideeffects,therearechangesinliverfunctiontests, andinsomecases,apotentialadversecardiovasculareffect. Therefore,febuxostatisnotrecommendedinpatientswith animpairedheartfailure.19Severalstudiesconfirmthe
effec-tivenessoffebuxostatversusallopurinolatadoseof300mg, tothe pointseveralcountries–amongthemUnitedStates, Canada,and20Europeancountries–havealreadyallowedits useatdailydosesof40,80,or120mg.84,88Since2012,
febuxo-statisrecommendedbytheACRasafirst-lineurate-lowering therapy.89Duemainlytoitscost,itsuseismostsuitablein
caseoffailureorimpossibilityofusingallopurinol.90
Uricosuricagents(probenecid,benzbromarone,and sulfinpyra-zone)areweakorganicacidswhosemaineffectistoincrease therenalexcretionofuricacidbyinhibitinganiontransporters in theproximal convolutedtubule(URAT1), responsiblefor the resorption of urate. Probenecid is the only uricosuric agentcurrentlyavailableintheUnitedStates.Benzbromarone waswidelyavailableinEuropean,Asian,andSouth Ameri-cancountries,butitwasbannedin2003followingreportsof severelivertoxicityandbonemarrowsuppression.91
Urico-suricdrugsaresecond-lineagentsinthetreatmentofgout, becomingfirst-lineonlyinpatientswhocannottoleratethem, orwhenthereisnoindicationforanXOinhibitor.Inaddition, these drugsincreasesignificantlythe concentrationofuric acidinthecollectingducts,predisposingtostoneformation.62
The uricosuric properties of losartan and fenofibrate have alsobeendescribed,albeitdiscreet.92 Amildbutpersistent
uricosuriceffectwasobservedwiththeuseofvitaminCat dailydoseslowerthan500mg.93Lesinuradeisapromising
uri-cosuric agent, whichis currentlyata latestage ofclinical development,and isanon-nucleosidereversetranscriptase inhibitor.Originally,thisdrugwasdevelopedtotreatpatients withHIV.91
Non-primate mammals and some birds do not have urate asan endproduct ofpurinemetabolism, becausein another enzyme,uricase, catalyzesthe conversionofurate toallantoin,asolubleandreadilyexcretedproduct.94 Peglot-icase is a porcine recombinant uricase linked to various ethylene chainsto prolong their activity and reduce their immunogenicity.94 UA levelsdecrease tovery lowor
unde-tectablelevelsafewhoursafterintravenousadministration ofpegloticase8mg.52 Furthermore,repeated infusions
pro-mote the resorption of MSU crystal deposits, having an efficient and quick action in the reduction in tophi.95,96
Therearereportsofpatientsthatproduceantibodiesagainst pegloticase,decreasingitsactivityandincreasingtheriskof hypersensitivityreactions.97Currently,pegloticaseinfusionis
licensed inthe UnitedStates forpatients refractorytooral therapy.6,62
Purines
Hypoxanthine
Xanthine
Uric acid
Allantoin Uricase
Pegloticase
Hyperuricemia and ormation of urate crystals
Formation of uric acid calculi Renal excretion
(10% of the filtrate)
Deposition of urate crystals in joints, leading to an intense inflammatory
response (acute gout)
Colchicine NSAIDs Corticosteroids Canakinumab Uricosuric agents
Lesinurate Losartan Febuxostat
Allopurinol
Oxypurinol
URAT1
XO XO
XDH XDH
Febuxostat
Allopurinol
Oxypurinol
Fig.1–Pharmacologicagentsandtheirmechanismofactionforthetreatmentofgout.
(pegylateduricase)canperform thesame functionsofthis enzyme(Fig.1).
Options
and
limitations
of
the
Brazilian
drug
market
Goutisconsideredthemostcommonformofinflammatory arthritis in menaged over 40 years. Known sinceancient times,theso-called“diseaseofkings”hasattractedthe atten-tionofclinicalinvestigatorsoverseveralcenturies.However, its treatment has remained largely unchanged for almost a century, until mid-2008, when the FDA advisory board approvedthemarketingoffebuxostat,thefirstnewdrugin fourdecadesforgouttreatment.Sincethen,thereisstillno provisionfortheentry ofthisproductinBrazil,and many patientswithrenalfailurewhowouldbenefitfromfebuxostat needtoimportthemedicationwithamedicalprescription. Inthetreatmentofanacuteattack,colchicine,corticosteroids andawiderangeofnon-steroidalanti-inflammatorydrugsare availableintheBrazilianmarket,andthesedrugsareusedin accordancewiththeexperienceofexperts.Canakinumabis notacommontherapeuticoptionforacutecrisesduetoits highcostandalsotothegoodresponseoftheacutecrisesto traditionalmedicationsinmostcases.
ThereisnosetdateforthearrivalofPegloticase,which canbeusedinpatientswithrefractorychronictophaceous gout,intheBrazilianmarket.Rasburicase,whichismarketed inBrazil,isusedinthepreventionand treatmentoftumor
lysissyndrome,beingonlyapprovedforuseinadultpatients withleukemia,lymphomaorsolidtumors,whoaresubjected tocancertherapy,and whoare atriskofpresentingtumor lysissyndromeoranelevationofuricacid.Duetoitsoff-label indicationfortophaceousgoutandalsotothelackof conve-nienceofitsuse(sincethedrugisusedintravenouslyatdaily intervals),rasburicasehasnotbeenroutinelyusedinthe treat-mentofgout.Furthermore,aprecautionisrequiredinpatients with a history of atopy, asrasburicase can induceallergic responses. Clinical experience with this agent reveals that the patientshouldbeclosely monitoredforthe emergence ofadverseeventsofanallergicnature,especiallyskin reac-tions,andbronchospasm.Nonetheless,arecentsystematic reviewshowsthatingoutypatientswithrenalinsufficiency, rasburicase, febuxostat, benzbromarone, and allopurinol in associationwithbenzbromaroneappeartobeeffectiveand safe.98
Theuse ofbenzbromaroneisprohibitedintheUS mar-ket,butthetherapeuticexperienceinBrazilhasbeenpositive, bothinmonotherapyorassociatedtoallopurinolinrefractory cases.99 Althoughseveralnationalpharmaceuticals
compa-nies are producing this molecule, some patients complain of sporadic shortage ofthe product in the domestic mar-ketandoftenoptforapharmacologicalmanipulationofthe product.100
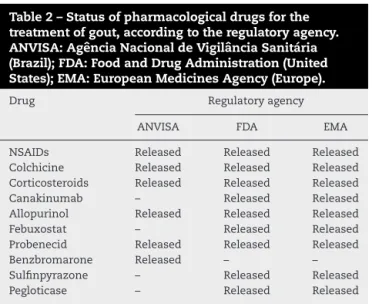
Table2–Statusofpharmacologicaldrugsforthe treatmentofgout,accordingtotheregulatoryagency. ANVISA:AgênciaNacionaldeVigilânciaSanitária (Brazil);FDA:FoodandDrugAdministration(United States);EMA:EuropeanMedicinesAgency(Europe).
Drug Regulatoryagency
ANVISA FDA EMA
NSAIDs Released Released Released
Colchicine Released Released Released
Corticosteroids Released Released Released
Canakinumab – Released Released
Allopurinol Released Released Released
Febuxostat – Released Released
Probenecid Released Released Released
Benzbromarone Released – –
Sulfinpyrazone – Released Released
Pegloticase – Released Released
necroepidermyoisis102 and death with the use of this
product.103
Unfortunately,itsactivemetabolite,oxypurinol,whichis availableontheEuropeanmarket,isalsonotavailableinthe Brazilianmarket.However,questionnaireappliedto rheuma-tologistsinthe1990sindicatedthatcolchicinewasthemost prescribedproductforthetreatmentofgoutinBrazil.104
Recentfindingsindicatingthattheelevationofuricacidis animportantcardiovascularriskfactorhasforcedachange ofsceneryinthetreatmentofpatientswithhyperuricemia, whichshouldimpactthetreatmentofgoutyarthritisinBrazil, ashasoccurredinothercountries.105Finally,forthenextfew
years,theauthors believethatthereisagreatopportunity fortheBraziliandrugmarketforthetreatmentofgoutand especiallyforpatientsusingprivateandpublic(SUS)health caresystems.Table2presentsacomparisonofthepresence ofthesemedicinesintheEuropean,US,andBrazilianmarkets.
Conflicts
of
interest
Dr.ValderilioFeijóAzevedo,Dr.EduardodosSantosPaivaand Dr.GeraldodaRochaCastelarPinheirodeclaretheir participa-tionintheadvisoryboardofAstraZeneca.Theotherauthors declarenoconflictsofinterestforthispublication.
r
e
f
e
r
e
n
c
e
s
1. BardinT,RichetteP.Definitionofhyperuricemiaandgouty conditions.CurrOpinRheumatol.2014;26:186–91.
2. Perez-RuizF,Herrero-BeitesAM.Evaluationandtreatment ofgoutasachronicdisease.AdvTher.2012;29:935–46.
3. GeorgeJ,StruthersAD.Roleofurate,xanthineoxidaseand theeffectsofallopurinolinvascularoxidativestress.Vasc HealthRiskManag.2009;5:265–72.
4. PacherP,NivorozhkinA,SzabóC.Therapeuticeffectsof xanthineoxidaseinhibitors:renaissancehalfacenturyafter thediscoveryofallopurinol.PharmacolRev.2006;58:87–114.
5. SautinYY,JohnsonRJ.Uricacid:theoxidant–antioxidant paradox.NucleosidesNucleotidesNucleicAcids. 2008;27:608–19.
6. ZhangW,DohertyM,BardinT,PascualE,BarskovaV, ConaghanP,etal.EULARevidencebasedrecommendations
forgout.PartII:Management.Reportofataskforceofthe EULARStandingCommitteeforInternationalClinical StudiesIncludingTherapeutics(ESCISIT).AnnRheumDis. 2006;65:1312–24.
7.LipkowitzMS.Regulationofuricacidexcretionbythe kidney.CurrRheumatolRep.2012;14:179–88.
8.Perez-RuizF,CastilloE,ChinchillaSP,Herrero-BeitesAM. Clinicalmanifestationsanddiagnosisofgout.RheumDis ClinNorthAm.2014;40:193–206.
9.ParkJW,KoDJ,YooJJ,ChangSH,ChoHJ,KangEH,etal. Clinicalfactorsandtreatmentoutcomesassociatedwith failureinthedetectionofuratecrystalinpatientswith acutegoutyarthritis.KoreanJInternMed.2014;29: 361–9.
10.KrishnanE,SvendsenK,NeatonJD,GranditsG,KullerLH. Long-termcardiovascularmortalityamongmiddle-aged menwithgout.ArchInternMed.2008;168:1104–10.
11.StackAG,HanleyA,CasserlyLF,CroninCJ,AbdallaAA, KiernanTJ,etal.Independentandconjointassociationsof goutandhyperuricaemiawithtotalandcardiovascular mortality.QJM.2013;106:647–58.
12.ZhangW,DohertyM,PascualE,BardinT,BarskovaV, ConaghanP,etal.EULARevidencebasedrecommendations forgout.PartI:diagnosis.ReportofataskforceoftheEULAR StandingCommitteeforInternationalClinicalStudies IncludingTherapeutics(ESCISIT).AnnRheumDis. 2006;65:1301–11.
13.MaruhashiT,NakashimaA,SogaJ,FujimuraN,IdeiN, MikamiS,etal.Hyperuricemiaisindependentlyassociated withendothelialdysfunctioninpostmenopausalwomenbut notinpremenopausalwomen.BMJOpen.2013;3:e003659.
14.Duskin-BitanH,CohenE,GoldbergE,ShochatT,LeviA, GartyM,etal.Thedegreeofasymptomatichyperuricemia andtheriskofgout.Aretrospectiveanalysisofalarge cohort.ClinRheumatol.2014;33:549–53.
15.PasalicD,MarinkovicN,Feher-turkovicL.Review.Uricacid asoneoftheimportantfactorsinmultifactorialdisorders– factsandcontroversies.BiochemMed(Zagreb).
2012;22:63–75.
16.RoddyE,ChoiHK.Epidemiologyofgout.RheumDisClin NorthAm.2014;40:155–75.
17.ChoiHK,CurhanG.Independentimpactofgouton mortalityandriskforcoronaryheartdisease.Circulation. 2007;116:894–900.
18.ChoiHK,AtkinsonK,KarlsonEW,WillettW,CurhanG. Purine-richfoods,dairyandproteinintake,andtheriskof goutinmen.NEnglJMed.2004;350:1093–103.
19.ReesF,HuiM,DohertyM.Optimizingcurrenttreatmentof gout.NatRevRheumatol.2014;10:271–83.
20.CronsteinBN,SunkureddiP.Mechanisticaspectsof inflammationandclinicalmanagementofinflammationin acutegoutyarthritis.JClinRheumatol.2013;19:19–29.
21.Sicras-MainarA,Navarro-ArtiedaR,Ibá ˜nez-NollaJ.Usode recursoseimpactoeconómicodelospacientescongota: estudiomulticéntricodeámbitopoblacional.ReumatolClin. 2013;9:94–100.
22.AldermanMH.Podagra,uricacid,andcardiovascular disease.Circulation.2007;116:880–3.
23.SimkinPA.Thepathogenesisofpodagra.AnnInternMed. 1977;86:230–3.
24.LiotéF,LancrenonS,LanzS,GuggenbuhlP,LambertC, SarauxA,etal.GOSPEL:Prospectivesurveyofgoutin France.PartI:designandpatientcharacteristics(n=1003). BoneSpine.2012;79:464–70.
26.DalbethN,CollisJ,GregoryK,ClarkB,RobinsonE,Mcqueen FM.Tophaceousjointdiseasestronglypredictshand functioninpatientswithgout.Rheumatology. 2007;46:1804–7.
27.DalbethN,ClarkB,McQueenF,DoyleA,TaylorW.Validation ofaradiographicdamageindexinchronicgout.Arthritis CareRes.2007;57:1067–73.
28.Perez-RuizF,CalabozoM,PijoanJI,Herrero-BeitesAM, RuibalA.Effectofurate-loweringtherapyonthevelocityof sizereductionoftophiinchronicgout.ArthritisRheum. 2002;47:356–60.
29.MonuJUV,PopeTL.Gout:aclinicalandradiologicreview. RadiolClinNorthAm.2004;42:169–84.
30.LawrenceRC,HochbergMC,KelseyJL,McDuffieFC,Medsger TA,FeltsWR,etal.Estimatesoftheprevalenceofselected arthriticandmusculoskeletaldiseasesintheUnitedStates. JRheumatol.1989;16:427–41.
31.LawrenceRC,FelsonDT,HelmickCG,ArnoldLM,ChoiH, DeyoRA,etal.Estimatesoftheprevalenceofarthritisand otherrheumaticconditionsintheUnitedStates.PartII. ArthritisRheum.2008;58:26–35.
32.RoubenoffR,KlagMJ,MeadLA,LiangKY,SeidlerAJ, HochbergMC.Incidenceandriskfactorsforgoutinwhite men.JAMA.1991;266:3004–7.
33.PillingerMH,GoldfarbDS,KeenanRT.Goutandits comorbidities.BullNYUHospJtDis.2010;68:199–203.
34.ChoiHK,AtkinsonK,KarlsonEW,WillettW,CurhanG. Alcoholintakeandriskofincidentgoutinmen:a prospectivestudy.Lancet.2004;363:1277–81.
35.ZhangY,WoodsR,ChaissonCE,NeogiT,NiuJ,McAlindon TE,etal.Alcoholconsumptionasatriggerofrecurrentgout attacks.AmJMed.2006;119,800e13-8.
36.ZhangY,ChenC,ChoiH,ChaissonC,HunterD,NiuJ,etal. Purine-richfoodsintakeandrecurrentgoutattacks.Ann RheumDis.2012;71:1448–53.
37.KokVC,HorngJ-T,ChangW-S,HongY-F,ChangT-H. Allopurinoltherapyingoutpatientsdoesnotassociatewith beneficialcardiovascularoutcomes:apopulation-based matched-cohortstudy.PLoSOne.2014;9:e99102.
38.BholeV,DeVeraM,RahmanMM,KrishnanE,ChoiH. Epidemiologyofgoutinwomen:fifty-two-yearfollowupofa prospectivecohort.ArthritisRheum.2010;62:1069–76.
39.RichetteP,ClersonP,PérissinL,FlipoR-M,BardinT. Revisitingcomorbiditiesingout:aclusteranalysis.Ann RheumDis.2015;74:142–7.
40.LyngdohT,VuistinerP,Marques-VidalP,RoussonV,Waeber G,VollenweiderP,etal.Serumuricacidandadiposity: decipheringcausalityusingabidirectionalmendelian randomizationapproach.PLoSOne.2012;7:e39321.
41.Ishikawa-TakataK,OhtaT,MoritakiK,GotouT,InoueS. Obesity,weightchange,andrisksforhypertension,diabetes, andhypercholesterolemiainJapanesemen.EurJClinNutr. 2002;56:601–7.
42.ChoiHK.Aprescriptionforlifestylechangeinpatientswith hyperuricemiaandgout.CurrOpinRheumatol.
2010;22:165–72.
43.SureshE,DasP.Recentadvancesinmanagementofgout. QJM.2012;105:407–17.
44.StampLK,O’DonnellJL,FramptonC,DrakeJM,ZhangM, ChapmanPT.Clinicallyinsignificanteffectofsupplemental vitaminConserumurateinpatientswithgout:apilot randomizedcontrolledtrial.ArthritisRheum. 2013;65:1636–42.
45.ChoiHK,WillettW,CurhanG.Coffeeconsumptionandrisk ofincidentgoutinmenaprospectivestudy.Arthritis Rheum.2007;56:2049–55.
46.TaylorWJ,FransenJ,DalbethN,NeogiT,SchumacherHR, BrownM,etal.Performanceofclassificationcriteriaforgout
inearlyandestablisheddisease.AnnRheumDis. 2016;75:178–82.
47.KienhorstLBE,JanssensHJEM,FransenJ,JanssenM.The validationofadiagnosticruleforgoutwithoutjointfluid analysis:aprospectivestudy.Rheumatology(Oxford). 2015;54:1329–30.
48.PérezRuizF,RuizLópezJ,HerreroBeitesAM.Influenceofthe naturalhistoryofdiseaseonapreviousdiagnosisinpatients withgout.ReumatolClínica(EnglishEd).2009;5:248–51.
49.ShojiA,YamanakaH,KamataniN.Aretrospectivestudyof therelationshipbetweenserumuratelevelandrecurrent attacksofgoutyarthritis:evidenceforreductionof recurrentgoutyarthritiswithantihyperuricemictherapy. ArthritisRheum.2004;51:321–5.
50.AleleJD,KamenDL.Theimportanceofinflammationand vitaminDstatusinSLE-associatedosteoporosis.
AutoimmunRev.2010;9:137–9.
51.BeckerMA,SchumacherHR,MacDonaldPA,LloydE, LademacherC.Clinicalefficacyandsafetyofsuccessful longtermurateloweringwithfebuxostatorallopurinolin subjectswithgout.JRheumatol.2009;36:1273–82.
52.SundyJS,BarafHSB,YoodRA,EdwardsNL,Gutierrez-Urena SR,TreadwellEL,etal.Efficacyandtolerabilityofpegloticase forthetreatmentofchronicgoutinpatientsrefractoryto conventionaltreatment:tworandomizedcontrolledtrials. JAMA.2011;306:711–20.
53.PascualE,SiveraF.Timerequiredfordisappearanceofurate crystalsfromsynovialfluidaftersuccessfulhypouricaemic treatmentrelatestothedurationofgout.AnnRheumDis. 2007;66:1056–8.
54.SchlesingerN.Treatmentofacutegout.RheumDisClin NorthAm.2014;40:329–41.
55.KhannaD,FitzgeraldJD,KhannaPP,BaeS,SinghMK,Neogi T,etal.2012AmericanCollegeofRheumatologyguidelines formanagementofgout.Part1:systematic
nonpharmacologicandpharmacologictherapeutic approachestohyperuricemia.ArthritisCareRes(Hoboken). 2012;64:1431–46.
56.SchlesingerN.Managementofacuteandchronicgouty arthritis:presentstate-of-the-art.Drugs.2004;64:2399–416.
57.DalbethN,HaskardDO.Mechanismsofinflammationin gout.Rheumatology(Oxford).2005;44:1090–6.
58.BurnsCM,WortmannRL.Latestevidenceongout management:whattheclinicianneedstoknow.TherAdv ChronicDis.2012;3:271–86.
59.LiT,ChenSL,DaiQ,HanXH,LiZG,WuDH,etal.Etoricoxib versusindometacininthetreatmentofChinesepatients withacutegoutyarthritis:arandomizeddouble-blindtrial. ChinMedJ(Engl).2013;126:1867–71.
60.NationalT,CentreC.Nationalclinicalguidelineforcareand managementinadults.
61.TerkeltaubRA.Colchicineupdate:2008.SeminarsArthritis Rheum.2009;38:411–9.
62.CrittendenDB,PillingerMH.Theyearingout:2011–2012. BullNYUHospJtDis.2012;70:145–51.
63.RiccardiC,BruscoliS,MiglioratiG.Molecularmechanismsof immunomodulatoryactivityofglucocorticoids.Pharmacol Res.2002;45:361–8.
64.JanssensHJ,JanssenM,vandeLisdonkEH,vanRielPL,van WeelC.Useoforalprednisoloneornaproxenforthe treatmentofgoutarthritis:adouble-blind,randomised equivalencetrial.Lancet.2008;371:1854–60.
65.RitterJ,DubinKerrL,Valeriano-MarcetJ,SpieraH.ACTH revisited:effectivetreatmentforacutecrystalinduced synovitisinpatientswithmultiplemedicalproblems.J Rheumatol.1994;21:696–9.
clinicalevidenceandmechanismsofaction.SeminArthritis Rheum.2014;43:648–53.
67.AxelrodD,PrestonS.Comparisonofparenteral
adrenocorticotropichormonewithoralindomethacininthe treatmentofacutegout.ArthritisRheum.1988;31:803–5.
68.BussoN,EaH-K.Themechanismsofinflammationingout andpseudogout(CPP-inducedarthritis).Reumatism. 2011;63:230–7.
69.KuipersMT,AslamiH,VlaarAPJ,JuffermansNP,Tuip-de BoerAM,HegemanM,a,etal.Pre-treatmentwith
allopurinoloruricaseattenuatesbarrierdysfunctionbutnot inflammationduringmurineventilator-inducedlunginjury. PLoSOne.2012;7:e50559.
70.TerkeltaubRA,SchumacherHR,CarterJD,BarafHS,Evans RR,WangJ,etal.Rilonaceptinthetreatmentofacutegouty arthritis:arandomized,controlledclinicaltrialusing indomethacinastheactivecomparator.ArthritisResTher. 2013;15:R25.
71.RamiroS,Gaujoux-VialaC,NamJL,SmolenJS,BuchM, GossecL,etal.SafetyofsyntheticandbiologicalDMARDs:a systematicliteraturereviewinformingthe2013updateof theEULARrecommendationsformanagementof rheumatoidarthritis.AnnRheumDis.2014;73: 529–35.
72.SchlesingerN,DetryMA,HollandBK,BakerDG,BeutlerAM, RullM,etal.Localicetherapyduringboutsofacutegouty arthritis.JRheumatol.2002;29:331–4.
73.ReesF,JenkinsW,DohertyM.Patientswithgoutadhereto curativetreatmentifinformedappropriately:
proof-of-conceptobservationalstudy.AnnRheumDis. 2013;72:826–30.
74.SpencerK,CarrA,DohertyM.Patientandproviderbarriers toeffectivemanagementofgoutingeneralpractice:a qualitativestudy.AnnRheumDis.2012;71:1490–5.
75.ChaichianY,ChohanS,BeckerMA.Long-termmanagement ofgout:nonpharmacologicandpharmacologictherapies. RheumDisClinNorthAm.2014;40:357–74.
76.HunterDJ,YorkM,ChaissonCE,WoodsR,NiuJ,ZhangY. Recentdiureticuseandtheriskofrecurrentgoutattacks: theonlinecase-crossovergoutstudy.JRheumatol. 2006;33:1341–5.
77.ChoiHK,SorianoLC,ZhangY,RodríguezLAG.
Antihypertensivedrugsandriskofincidentgoutamong patientswithhypertension:populationbasedcase–control study.BMJ.2012;344:d8190.
78.VanLieshout-ZuidemaMF,BreedveldFC.Withdrawalof longtermantihyperuricemictherapyintophaceousgout.J Rheumatol.1993;20:1383–5.
79.Perez-RuizF,AtxotegiJ,HernandoI,CalabozoM,NollaJM. Usingserumuratelevelstodeterminetheperiodfreeof goutysymptomsafterwithdrawaloflong-term
urate-loweringtherapy:aprospectivestudy.ArthritisCare Res.2006;55:786–90.
80.NomuraJ,BussoN,IvesA,MatsuiC,TsujimotoS,Shirakura T,etal.Xanthineoxidaseinhibitionbyfebuxostatattenuates experimentalatherosclerosisinmice.SciRep.2014;4:4554.
81.StockerSL,McLachlanAJ,SavicRM,KirkpatrickCM,Graham GG,WilliamsKM,etal.Thepharmacokineticsofoxypurinol inpeoplewithgout.BrJClinPharmacol.2012;74:477–89.
82.WertheimerAI,DavisMW,LauterioTJ.Anewperspectiveon thepharmacoeconomicsofcolchicine.CurrMedResOpin. 2011;27:931–7.
83.SchumacherHR,BeckerMA,WortmannRL,MacDonaldPA, HuntB,StreitJ,etal.Effectsoffebuxostatversusallopurinol andplaceboinreducingserumurateinsubjectswith hyperuricemiaandgout:a28-week,phaseIII,randomized, double-blind,parallel-grouptrial.ArthritisCareRes. 2008;59:1540–8.
84.BeckerMA,SchumacherHRJr,WortmannRL,MacDonald PA,EustaceD,PaloWA,etal.Febuxostatcomparedwith allopurinolinpatientswithhyperuricemiaandgout.NEngl JMed.2005;353:2450–61.
85.KimSC,NewcombC,MargolisD,RoyJ,HennessyS.Severe cutaneousreactionsrequiringhospitalizationinallopurinol initiators:apopulation-basedcohortstudy.ArthritisCare Res.2013;65:578–84.
86.HarroldLR,MazorKM,PetersonD,NazN,FirnenoC,Yood RA.Patients’knowledgeandbeliefsconcerninggoutandits treatment:apopulationbasedstudy.BMCMusculoskelet Disord.2012;13:180.
87.GoldfarbDS,MacDonaldPA,HuntB,GunawardhanaL. Febuxostatingout:serumurateresponseinuricacid overproducersandunderexcretors.JRheumatol. 2011;38:1385–9.
88.BeckerMA,SchumacherHR,EspinozaLR,WellsAF, MacDonaldP,LloydE,etal.Theurate-loweringefficacyand safetyoffebuxostatinthetreatmentofthehyperuricemia ofgout:theCONFIRMStrial.ArthritisResTher.2010;12:R63.
89.KhannaD,FitzGeraldJD,KhannaPP,BaeS,SinghM,NeogiT, etal.2012AmericanCollegeofRheumatologyguidelinesfor mmanagementofgout.Part1:systematic
nonpharmacologicandpharmacologictherapeutic approachestohyperuricemia.ArthritisCareRes. 2012;64:1431–46.
90.StevensonM,PandorA.Febuxostateforthemanagementof hyperuricaemiainpatientswithgout:anicesingle technologyappraisal.Pharmacoeconomics.2011;29: 133–40.
91.BachMH,SimkinPA.Uricosuricdrugs:theonceandfuture therapyforhyperuricemia?CurrOpinRheumatol. 2014;26:169–75.
92.TakahashiS,MoriwakiY,YamamotoT,TsutsumiZ,KaT, FukuchiM.Effectsofcombinationtreatmentusing
anti-hyperuricaemicagentswithfenofibrateand/orlosartan onuricacidmetabolism.AnnRheumDis.2003;62:572–5.
93.ChoiHK,GaoX,CurhanG.VitaminCintakeandtheriskof goutinmen:aprospectivestudy.ArchInternMed. 2009;169:502–7.
94.WuXW,MuznyDM,LeeCC,CaskeyCT.Twoindependent mutationaleventsinthelossofurateoxidaseduring hominoidevolution.JMolEvol.1992;34:78–84.
95.ShermanMR,SaiferMGP,Perez-RuizF.PEG-uricaseinthe managementoftreatment-resistantgoutand
hyperuricemia.AdvDrugDelivRev.2008;60:59–68.
96.BarafHS,BeckerMA,Gutierrez-UrenaSR,TreadwellEL, Vazquez-MelladoJ,RehrigCD,etal.Tophusburden reductionwithpegloticase:resultsfromphase3
randomizedtrialsandopen-labelextensioninpatientswith chronicgoutrefractorytoconventionaltherapy.Arthritis ResTher.2013;15:R137.
97.GansonNJ,KellySJ,ScarlettE,SundyJS,HershfieldMS. Controlofhyperuricemiainsubjectswithrefractorygout, andinductionofantibodyagainstpoly(ethyleneglycol) (PEG),inaphaseItrialofsubcutaneousPEGylatedurate oxidase.ArthritisResTher.2006;8:R12.
98.CastrejonI,ToledanoE,RosarioMP,LozaE,Pérez-RuizF, CarmonaL.Safetyofallopurinolcomparedwithother urate-loweringdrugsinpatientswithgout:asystematic reviewandmeta-analysis.RheumatolInt.2015;35:1127–37.
99.AzevedoVF,BuiarPG,GiovanellaLH,SeveroCR,CarvalhoM. Allopurinol,benzbromarone,oracombinationintreating patientswithgout:analysisofaseriesofoutpatients.IntJ Rheumatol.2014;2014:263720.
narcaricina-12626/forum/falta-do-medicamento-no-mercado-7053.
101.vanEchteldIA,vanDurmeC,FalzonL,LandewéRB,vander HeijdeDM,AletahaD.Treatmentofgoutpatientswith impairmentofrenalfunction:asystematicliteraturereview. JRheumatolSuppl.2014;92:48–54.
102.RanuH,JiangJ,MingPS.Acaseseriesofallopurinol-induced toxicepidermalnecrolysis.IndianJDermatol.2011;56:74–6.
103.RachidA,MagalhãesFLGM,TafarelJR,SchmitzR.Óbito decorrentedasíndromedehipersensibilidadeaoalopurinol (SHA).RevBrasReumatol.2004;44:248–50.
104.FerrazMB,SatoEI,NishieIA,VisioniRA.Asurveyofcurrent prescribingpracticesingoutyarthritisandsymptomatic hyperuricemiainSanPaulo,Brazil.JRheumatol. 1994;21:374–5.