Full paper published online: August 31, 2009 ISSN 1678-9199.
PURIFICATION AND PARTIAL CHARACTERIZATION OF A COAGULANT SERINE PROTEASE FROM THE VENOM OF THE IRANIAN SNAKE Agkistrodon
halys
Ghorbanpur M (1), Zare Mirakabadi A (2), Zokaee F (1), Zolfagarrian H (2), Rabiei H (2)
(1) Chemical Engineering Department, Amirkabir University, Tehran, Iran; (2) Department of Venomous Animals and Antivenom Production, Razi Vaccine and Serum Research Institute, Karaj, Iran.
ABSTRACT: Agkistrodon halys is one of several dangerous snake species in Iran. Among the most important signs and symptoms in patients envenomated by this snake is disseminated intravascular coagulation. A thrombin-like enzyme, called AH143, was isolated from Agkistrodon halys venom by gel filtration on a Sephadex G-50 column, ion-exchange chromatography on a DEAE-Sepharose and high performance liquid chromatography (HPLC) on a C18 column. In the final stage of purification, 0.82 mg of purified enzyme was obtained from 182.5 mg of venom. The purified enzyme showed a single protein band by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE), under reducing conditions, and its molecular mass was found to be about 30 kDa. AH143 revealed clotting activity in human plasma, which was not inhibited by EDTA or heparin. This enzyme still demonstrated coagulation activity when exposed to variations in temperature and pH ranging, respectively, from 30 to 40°C and from 7.0 to 8.0. It also displayed proteolytic activities on synthetic substrate. The purified enzyme did not show any effect on casein. We concluded that the venom of the Iranian snake Agkistrodon halys contains about 0.45% single procoagulant protein which appears to be a thrombin-like enzyme.
KEY WORDS: Iranian snake, venom, Agkistrodon halys, serine protease, chromatography, coagulant activity.
CONFLICTS OF INTEREST: There is no conflict.
CORRESPONDENCE TO:
ABBAS ZARE MIRAKABADI, Department of Venomous Animals and Antivenom
Production, Razi Vaccine and Serum Research Institute, Karaj, Iran. Phone: +98 261
INTRODUCTION
Snake venoms contain numerous enzymes such as glucosidases and proteases,
with the latter group including metalloproteases and serine proteases. Several venom
serine proteases present important fibrino(geno)lytic, kininogenase and thrombin-like
activities (1, 2).
Thrombin-like enzymes (TLE) from snake venoms belong to a class of serine
proteases that can result in blood clotting in vitro, a feature exhibited by numerous
snake toxins. These enzymes render the blood uncoagulable when acting in vivo,
apparently by depleting the circulating fibrinogen (3, 4). Most of thrombin-like
enzymes cleave fibrinogen to release fibrinopeptide A and cannot activate factor XIII
(5, 6). Therefore, thrombin-like enzymes hydrolyze fibrinogen to produce
non-cross-linked fibrins that are more susceptible to the lytic action of plasmin than
thrombin-induced cross-linked clots. These clots formed by TLE action are removed from the
circulation either by fibrinolysis or by the reticuloendothelial system (5, 6).
Several venoms from the families Crotalidae and Viperidae contain proteolytic
enzymes that exercise some effect on the blood coagulation process (7, 8). Since
fibrinogen-clotting enzyme was first discovered in snake venom in the early years of
the 20th century, numerous TLE have been isolated from different snake species (5).
These enzymes are widely distributed among several pit viper genera (Agkistrodon,
Bothrops, Crotalus, Lachesis and Trimeresurus), as well as in true vipers (Bitis and
Cerastes) and colubrids(Dispholidus typus) (9). Some TLE from snake venoms with
known complete amino acid sequences are: ancrod (Calloselasma rhodostoma,
formerly Agkistrodon rhodostoma) batroxobin (Bothrops atrox moojeni), bilineobin (A.
binlineatus) and crotalase (C. adamanteus) (10-14).
Thrombin-like enzymes are medically promising and act as defibrinogenating agents,
such as ancrod and batroxobin, that have been extensively used in victims of
thrombosis, myocardial infarction, peripheral vascular disease, acute ischemia and
renal transplant rejection (15). Additionally, these enzymes are employed in deep
corneal ulcer repair and non-pregnant canine uterus (16, 17). A study on the efficacy
of fibrin glue derived from snake venom in colon anastomosis of rats was carried out
by Leite et al. (14).
To the best of our knowledge, no prior work has been published on purification of this
enzyme from A. halys venom; hence, in this study, we attempted to it and report its
MATERIALS AND METHODS
Material
Fresh crude venom of A. halys was obtained directly from local Iranian snakes,
lyophilized and stored at –20°C. DEAE-Sepharose, Sephadex G-50 and C18 columns were purchased from Pharmacia (Sweden). Bovine serum albumin BApNA
(N2-benzoyl-DL-arginine-p-nitroanilide), standard protein marker kits and other
reagents for enzymatic and biochemical assays were purchased from Sigma. All
chemicals were of analytical reagent grade.
Isolation of Thrombin-Like Enzyme
Lyophilized crude venom of A. halys (200 mg) was dissolved in 8 mL of 50 mM
ammonium acetate buffer (pH 7.4) and centrifuged at 5,000 rpm for 15 minutes at
4°C. Subsequently, the supernatant was filtered on a 0.45 microfilter to remove insoluble materials.
Clear supernatant was submitted to molecular exclusion on a Sephadex G-50
chromatographic column (150 x 3 cm), previously equilibrated with ammonium
acetate buffer (pH 7.4) and, then, eluted with the same buffer. Fractions of 9 mL/tube
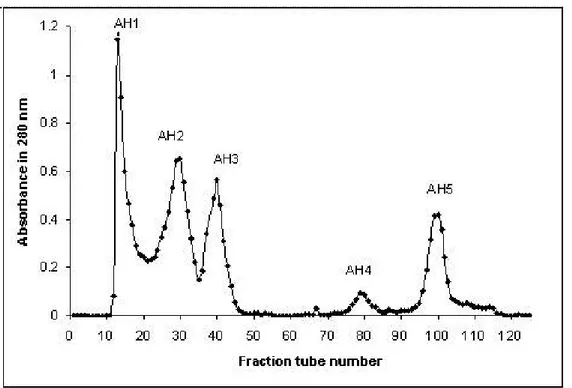
were collected at a flow rate of 60 mL/hour at 4°C. Obtained fractions were denominated consecutively AH1 to AH5 to indicate A. halys fractions 1 to 5.
Fractions revealed clotting activity during the gel chromatography step. AH1 was
pooled and dialyzed overnight at 4°C against distilled water and applied on DEAE-Sepharose column (2.5 x 20 cm), equilibrated with 20 mM Tris buffer, at pH 8.2. In
this step, proteins were eluted with a linear gradient of NaCl from 0.0 to 0.5 mM. Flow
rate was 17 mL/hour and 5-mL fractions were collected at 4°C.
The fraction that had shown clotting activity in the previous step was pooled and
dialyzed overnight at 4°C and applied on an HPLC column, C18 (H2O, 0.1% trifluoroacetic acid), and eluted with a concentration gradient of solvent B
(acetonitrile, 0.1% trifluoroacetic acid) from 0 to 30%, at a flow rate of 0.5 mL/minute
during 30 minutes. The peaks were monitored at 280 nm.
Blood Collection
Normal pooled plasma comprised samples from ten healthy donors, without history of
bleeding or thrombosis. Blood was centrifuged for 20 minutes at 2,000 rpm, and the
Determination of Molecular Mass
SDS-PAGE (12%) was performed according to Laemmli (18), utilizing a standard of
low molecular masses ranging from 6.5 to 66 kDa.
Purity Analysis
Analysis of purity was carried out by HPLC on a C18 column as previously described.
Prothrombin Time (PT) Assay
Prothrombin time reagent (200 μL) and sample aliquots (200 μL) were preincubated
for ten minutes at 37°C and mixed. Then, 100 μL of plasma was added and clotting time was recorded (19).
Amidolytic Activity
Sample proteolytic activity was assayed using the method of Laemmli (18). The
substrate solution was prepared by dissolving BApNA in 5 mL of dimethyl methyl
sulfoxide (DMSO) and adding 95 mL of 0.05 M Tris-HCl buffer (pH 8.2). Proteolytic
activity was monitored through the expression of BApNA hydrolysis by mixing 50 μL
of sample with 100 μL of the substrate solution. After ten minutes of incubation at
37°C, the absorbance was measured at 410 nm. One unit of protease activity corresponds to an increase of A410 = 0.001/min (18).
Coagulant Activity
Plasma (200 μL) and sample aliquots with different amounts of enzyme (2.5, 5.0,
10.0, 20.0 or 50.0 μg) were briefly preincubated at 37°C. After that, they were mixed and shaken and then clotting times were recorded (15, 20, 21).
Proteolytic Activity on Casein
Proteolytic activity was determined by the Sant’Ana method (22). Various amounts
(10, 20, 30, 40, 50 or 60 mg) of the enzyme were added to the buffer solution 0.1M
Tris-HCl, pH 9.0, and the final volume was adjusted to 250 μL, followed by 750 μL of
1% (m/v) casein and incubated for 15 minutes at 37°C. The reaction was stopped by addition of 1.5 mL of 30% TCA. Resulting proteolysis products in the supernatant
minutes at 280 nm. One unit of caseinolytic activity corresponds to an increase of
A280 = 0.001/min (2).
Inhibition of Enzyme Activity
The effect of ethylenediaminetetraacetic acid (EDTA) and heparin were examined by
incubation with enzymes in 180 μL of 20 mM Tris-HCI, with optimum pH at 37°C for five minutes. After mixing each of the inhibitors and 10 mL of purified enzyme, the
remaining activity was determined by measuring the hydrolysis of synthetic BApNA.
The remaining coagulating activity was calculated as percent activity in relation to the
control preparation incubated without inhibitors, which was considered 100% (18).
Effect of pH and Temperature
The effect of pH on enzyme activity was evaluated by measuring residual enzyme
activity after incubation at different pH, ranging from 4.0 to 6.0 with 0.1 M
citrate-NaOH buffer while between 7.0 and 10 with 0.1 M Tris-HCl buffer, at 37°C for ten minutes.
Additionally, enzyme activity was assayed at diverse temperatures ranging from 20 to
70°C using BApNA as a substrate. The assay was conducted at pH 7.5 with 0.05 M Tris-HCl buffer containing 0.02 M CaCl2 for five minutes in a temperature-controlled
water bath. Thereafter, heat-treated samples were rapidly cooled in an ice bath and
residual activity was measured using BApNA as substrate at pH 7.5 and 25°C for five minutes, as previously described (18).
Protein Determination
Protein concentration was measured according to Lowry et al. (23) using BSA as a
standard.
RESULTS
Isolation of the Thrombin-Like Enzyme
In the Sephadex G-50 fractionation of the crude venom, five peaks with absorbance
at 280 nm were obtained (AH1 to AH5) as shown in Figure 1. All fractions were
tested for coagulation, it was found that AH1 fraction was positive for procoagulation.
PT assay revealed that AH1 fraction had a 10 μmol/min/mg activity. The yield of
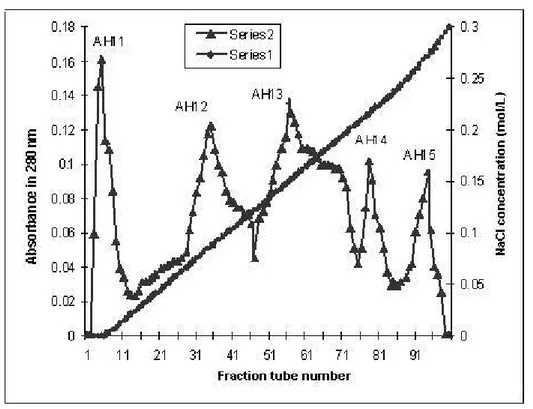
Further purification was carried out by ion exchange chromatography on
DEAE-Sepharose. In this step, five fractions were obtained (AH11 to AH15), out of which
AH14 fraction revealed procoagulant activity (Figure 2). The total yield of this fraction
was 56% that accounted for 0.98% of total venom. AH14 fraction was pooled,
dialyzed and applied to a C18 reversed-phase HPLC column. Four peaks (AH141 to
AH144) were obtained (Figure 3) and AH143 presented coagulant proteolytic activity.
The PT-test reached its elution peaks at 9 and 12 minutes (Figure 3). The purification
procedure of AH143 is summarized in Table1. Through this technique, about 0.82 mg
of purified enzyme was obtained from 182.5 mg of venom.
Table 1. Purification of a thrombin-like enzyme (AH143) from A. halys venom
Step Protein
) mg (
Specific activity*
)
μmol/min/mg (
Total amidase activity (μmol/min)
Yield (%)
Venom 182.5 10 1801.9 100
AH1 48.12 32.39 1558.6 91
AH14 1.8 622.2 1120 65
AH143 0.82 469.7 328.8 36
* Activity was determined using BApNA as substrate.
Figure 2. DEAE-Sepharose chromatography of AH1 fraction obtained from Sephadex G-50. The pooled fractions from Figure 1 were applied to DEAE-Sepharose column (2.5 x 20 cm) equilibrated with 0.05 M Tris-HCl buffer (pH 8.2). Proteins were eluted with a linear concentration gradient of NaCl from 0 to 0.5 M and 4.5-mL fractions per tube were collected.
Figure 3. HPLC chromatography of AH14 fraction obtained from DEAE-Sepharose chromatography. AH14 was applied on a HPLC C18 column equilibrated with solvent A (H2O, 0.1% TFA) and eluted with a concentration gradient of solvent B (acetonitrile, 0.1% TFA) from 0 to 30%, at a flow rate of 0.5 mL/minute during 30 minutes.
Purity and Determination of Molecular Mass
The homogeneity of purified enzymes was confirmed by SDS-PAGE and HPLC as
shown in Figures 4 and 5, respectively. Isolated AH143 revealed higher purity,
AH143 at 280 nm did not show the contaminating peptide. Furthermore, this enzyme
showed a single band by SDS-PAGE (Figure 6) whereas its molecular mass was
estimated to be about 30 kDa under reducing conditions.
Figure 4. Purity assay of AH143 by HPLC.
Figure 5. AH1 (A), AH14 (B) and AH143 (C) were analyzed by 12.5% SDS-PAGE in the presence of 1% 2-mercaptoethanol.
Caseinolytic, Clotting and Amidolytic Activity Assays
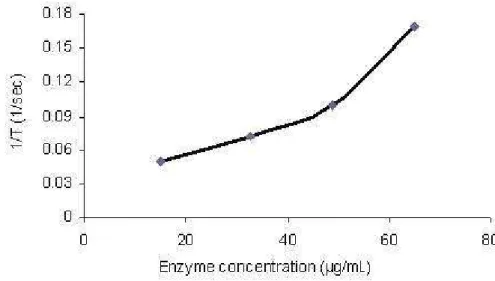
AH143 showed no proteolytic activity on casein while coagulant activity assay
demonstrated that it presented serine protease activity on BAPNA. AH143 also
concentrations (less than 15 μg/mL), this enzyme did not show any coagulant
activity. The time of plasma coagulation decreased with increased concentrations of
AH143.
Figure 6. Clotting activity of AH143 on human plasma.
Inhibition of Enzyme Activity
The amidolytic activity of AH143 was not affected by metal chelator (EDTA) or
heparin.
Effect of pH and Temperature on Activity
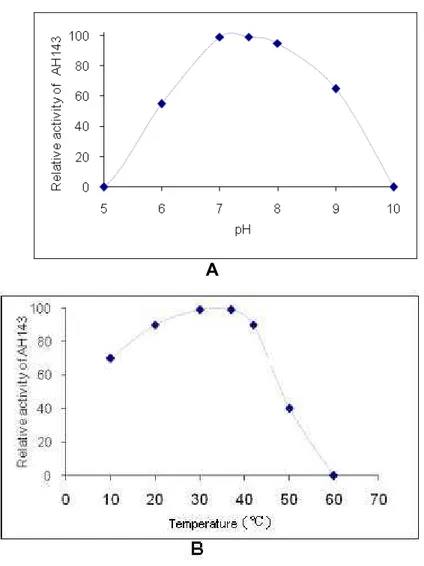
The optimum temperature for AH143 catalysis was evaluated in terms of amidolytic
activity on BApNA. AH143 activity after incubation at different pH, ranging from 7 to
8, was high (Figure 7A) and was stable at temperatures ranging from 20 to 40°C (Figure 7B). AH143 maximum activity was found to be at 7.5. At pH 6.0, AH143
activity was about 50% of the maximum; at pH 9.0, it was about 65%; while almost
95% of the activity was observed at pH 8.0.
A
Figure 7. AH143 activity depends on pH (A) and temperature (B).
DISCUSSION
Coagulant enzymes that can convert fibrinogen into fibrin gel are widely distributed in
Crotalidae snake venoms (8, 22, 24). The selected venom for this study was from the
same family group.
This article reports an efficient and relatively simple procedure for the isolation of a
highly purified thrombin-like serine protease from the venom of Iranian Agkistrodon
halys, called AH143. This enzyme was isolated and purified by a combination of gel
filtration on Sephadex G-50 (Figure 1), ion-exchange chromatography on
DEAE-Sepharose (Figure 2) and HPLC on C18 column. However, the total yield of the
enzyme accounted only for 36% (Table 1). This may be due to the partial loss of the
biological activity of the enzyme. On the other hand, HPLC was able to isolate a
unpublished) which could be the reason for reduced specific activity in the purification
final step.
In conventional methods, the thrombin-like protein represent around 1% of the
venom (2), but through the method herein described, it accounted for approximately
0.83% of the venom protein, out of which AH143 was found to be 0.45% (Table 1).
The amount of TLE in snake venoms from diverse species or in the same species
from various places can be different (6, 7). Analyses of AH143 activity in various
conditions of temperature and pH were carried out in accordance with Nikai et al.
(13). The temperature under which the purification was performed was based on the
observation that the enzyme solution was stable at 4°C (4, 7). Several reports indicate that the optimum pH for TLE activity is in the range of 7.0 to 8.5, being
practically inactive at 5 (4).
The molecular mass of AH143 is similar to other thrombin-like enzymes already
studied (6, 13). In addition, molecular mass and enzymatic activities – including
plasma clotting, amidolytic activity and not inhibition by heparin – characterize AH143
as a thrombin-like enzyme isolated from Agkistrodon halys venom (Figures 5, 6 and
7) (4, 15, 20, 21, 25, 26).
Since AH143 is not inhibited by heparin, it can be used in quantitative determination
of fibrinogen as well as in plasma of patients under heparin treatment and in the
laboratory for routine assays of coagulation factors (4).
In conclusion, the venom of Iranian snake (Agkistrodon halys) contain about 0.45%
of single procoagulant protein with molecular mass of about 30 kDa which it seems to
be a thrombin-like enzyme.
REFERENCES
1. Kini RM. Anticoagulant proteins from snake venoms: structure, function and
mechanism. Biochem J. 2006;397(3):377-87.
2. Ponce-Soto LA, Bonfim Novello VL, Navarro Oviedo R, Yarleque Chocas A,
Marangoni S. Isolation and characterization of a serine protease, Ba III-4, from
Peruvian Bothrops atrox venom. Protein J. 2007;26(6):387-94
3. Bell WR. Clinical trials with ancrod. In: Haemostasis and animal venoms. New
York: Marcel Dekker; 1988. p. 544-51.
4. Magalhães A. Purification and properties of a coagulant thrombin-like enzyme from
5. Choa SY, Hahna BS, Yangb KY, Kima YS. Purification and characterization of
calobin II, a second type of thrombin-like enzyme from Agkistrodon caliginosus
(Korean viper). Toxicon. 2001;39(4):499-506.
6. Furukawa K, Ishimara S. Use of thrombin-like snake venom enzymes in the
treatment of vascular occlusive diseases. In: Medical use of snake venom proteins.
Boca Raton: CRC Press; 1990. p. 161-99.
7. Etsuko O, Hidenobu T. Purification and characterization of a thrombin like enzyme,
elegaxobin II, with lys-bradykinin releasing activity from the venom of Trimeresurus
elegans (Sakishima-Habu). Toxicon. 2003;41(5):559-68.
8. Etsuko O, Hidenobu T. Purification and characterization of a thrombin-like enzyme,
elegaxobin, from the venom of Trimeresurus elegans (Sakishima-habu). Toxicon.
2000;38(8):1087-100.
9. Kini RM. Serine proteases affecting blood coagulation and fibrinolysis from snake
venoms. Haemost Thromb. 2005;34(4-5):200-4.
10. Burkhart W, Smith GFH, Su JL, Parikh I, Le Vine H. Amino acid sequence
determination of ancrod, the thrombin-like fibrinogenase from the venom of
Agkistrodon rhodostoma. FEBS Lett. 1992;297(3):297-301.
11. Itoh N, Tanaka N, Mihashi S, Yamashina I. Molecular cloning and sequence
analysis of cDNA for batroxobin, a thrombin-like snake venom enzyme. J Biol Chem.
1987;262(7):3132-5.
12. Au LC, Liu SB, Chou JS, Teh GW, Chang KJ, Shih CM. Molecular cloning and
sequence analysis of the cDNA for ancrod, a thrombin-like enzyme from the venom
of Calloselasma rhodostoma. Biochem J. 1993;294(Pt2):387-90.
13. Nikai T, Ohara A, Komori Y, Fox JW, Sugihara H. Primary structure of a
coagulant enzyme, bilineobin, from Agkistrodon bilineatus venom. Arch Biochem
Biophys. 1995; 318(1):89-96.
14. Leite CVS, Naresse E, Arantes HL, Lopes AF, Thomazini IA, Giannini MJS,
Mercadante MC, et al. An evaluation by rat colon anastomosis of the efficacy of fibrin
glue derived from snake venom. J Venom Anim Toxins. 2000;6(2):180-93.
15. Castro HC, Zingali RB, Albuquerque MG, Pujol-Luza M, Rodrigues CR. Review –
snake venom thrombin-like enzymes: from reptilase to now. CMLS Cell Mol Life Sci.
16. Moraes JRE, Correia PHA, Camplesi AC, Moraes FR. Experimental use of fibrin
glue derived from snake venom in non-pregnant canine uterus. J Venom Anim
Toxins incl Trop Dis. 2004;10(2):133-43.
17. Sampaio RL, Ranzani JJT, Brandão CVS, Thomazini-Santos IA, Barraviera B,
Barraviera SRCS, et al. Use of fibrin glue derived from snake venom in the repair of
deep corneal ulcers – experimental study in dogs. J Venom Anim Toxins incl Trop
Dis. 2007;13(4):857-73.
18. Laemmli UK. Cleavage of structural proteins during the assembly of the head of
bacteriophage, T4. Nature.1970;227(5259):680-5.
19. Athukorala Y, Jung WK, Vasanthan T, Jeon YJ. An anticoagulative
polysaccharide from an enzymatic hydrolysate of Ecklonia cava carbohydrate
polymers. 2006;66(2):184-91.
20. Yu X, Li Z, Xia X, Fang H, Zhou C, Chen H. Expression and purification of
ancrod, an anticoagulant drug, in Pichia pastoris. Protein Expr Purif.
2007;55(2):257-61.
21. Silva-Junior FP, Guedes HLM, Garvey LC, Bourguignon C, Di Cera Enrico,
Giovanni-De-Simone Salvatore. BJ-48, a novel thrombin-like enzyme from the
Bothrops jararacussu venom with high selectivity for Arg over Lys in P1: Role of
N-glycosylation in thermostability and active site accessibility. Toxicon.
2007;50(1):18-31.
22. Sant’Ana CD, Ticli FK, Oliveira LL. BjussuSP-I: A new thrombin-like enzyme
isolated from Bothrops jararacussu snake venom. Comp Biochem Physiol.
2008;151(3):443-54.
23. Lowry, OH, NJ Rosebrough, AL Farr, and RJ Randall. Protein measurement with
the folin phenol reagent. J. Biol. Chem. 1951;193(1):265-75.
24. Takashi S, Takashi H. Purification of two thrombin-like enzyme from the venom of
Agkistrodon caliginosus (Kankoku mamushi). Toxicon. 1984;22(1):29-38.
25. Koh YS, Chung KH, Kim DS. Biochemical characterization of a thrombin-like
enzyme and a fibrinolytic serine protease from snake (Agkistrodon saxatilis) venom,
Toxicon. 2001;39(4):555-60.
26. Matsui T, Fujimura Y, Titani K. Snake venom proteases affecting homeostasis