www.rpped.com.br
REVISTA
PAULISTA
DE
PEDIATRIA
ORIGINAL
ARTICLE
Heparin
for
clearance
of
peripherally
inserted
central
venous
catheter
in
newborns:
an
in
vitro
study
Talita
Balaminut
a,
Danielle
Venturini
a,
Valéria
Costa
Evangelista
da
Silva
b,
Edilaine
Giovanini
Rossetto
a,
Adriana
Valongo
Zani
a,∗aUniversidadeEstadualdeLondrina(UEL),Londrina,PR,Brazil bHospitalUniversitáriodeLondrina(HUL),Londrina,PR,Brazil
Received26July2014;accepted23January2015 Availableonline29June2015
KEYWORDS
Centralvenous catheterization; Catheterobstruction; Heparin;
Newborn
Abstract
Objective: Tocomparethe efficacyoftwo concentrations ofheparin toclearthelumenof
invitroclottedneonatalperipherallyinsertedcentralcatheters(PICCs).
Methods: Thisisaninvitro,experimentalquantitativestudyof76neonatal2.0-FrPICCs coag-ulatedinvitro.Thecathetersweredividedintotwogroupsof38PICCseach.Inbothgroupsan infusionoflowmolecularweightheparinwasadministeredwithadoseof25IU/mLforGroup1 and50IU/mLforGroup2.Thenegativepressuretechniquewasappliedtothecathetersofboth groupsat5,15and30minandat4htotesttheirpermeability.Kaplan---Meiersurvivalanalysis wasusedtoverifytheoutcomeofthegroupsaccordingtotimeintervals.
Results: Thecomparisonbetweenbothgroupsinthefirst5minshowedthatmorecatheters fromGroup2wereclearedcomparedtoGroup1(57.9vs.21.1%,respectively).Kaplan---Meier survivalanalysisshowedthatlesstimewasneededtoclearcatheterstreatedwith50IU/mLof heparin(p<0.001).
Conclusions: Theuseoflowmolecularweightheparinataconcentrationof50IU/mLwasmore effectiveinrestoringthepermeabilityofneonatalPICCsoccludedinvitrobyaclot,andthe useofthisconcentrationiswithinthesafetymarginindicatedbyscientificliterature. © 2015Sociedadede Pediatria de SãoPaulo. Published by Elsevier Editora Ltda.All rights reserved.
DOIoforiginalarticle:http://dx.doi.org/10.1016/j.rpped.2015.01.009 ∗Correspondingauthor.
E-mail:[email protected](A.V.Zani).
PALAVRAS-CHAVE
Cateterismovenoso central;
Obstruc¸ãodocateter; Heparina;
Recém-nascido
Heparinaparadesobstruc¸ãodecatetervenosocentraldeinserc¸ãoperiféricano
recém-nascido:estudoinvitro
Resumo
Objetivo: Comparar aeficácia deduas concentrac¸ões de heparinapara adesobstruc¸ão por coágulodocatetervenosocentraldeinserc¸ãoperiférica(CCIP)neonatalinvitro.
Métodos: Estudoexperimentalinvitroquantitativoqueusou76CCIPsneonataisdetamanho 2Frenchcoaguladosinvitro.Oscateteresforamdivididosemdoisgruposcom38CCIPscada. Ambososgruposreceberaminfusãodeheparinadebaixopesomolecular,comdosede25UI/mL no Grupo Ie de 50UI/mLno Grupo II.Os cateteresde ambos os grupos foramsubmetidos à técnicade pressãonegativa comcinco,15 e 30 minutose comquatro horase testou-se suapermeabilidade.Usou-seaanálisedesobrevivênciaparaverificarodesfechodosgrupos conformeosintervalosdetempo.
Resultados: Acomparac¸ãodosdoisgruposnointervalodetempodecincominutosmostrouum númeromaiordedesobstruc¸ãodecateteresnoGrupoII(57,9%)emrelac¸ãoaogrupo1(21,1%). A análise deKaplan Meier indicou menor tempopara desobstruc¸ão doscateteres quandoa heparinaemmaiorconcentrac¸ão(50UI/mL)foiusada(p<0,001).
Conclusões: Ousodeheparinadebaixopesomolecularnaconcentrac¸ãode50UI/mLfoimais eficaz narestaurac¸ãodapermeabilidadedeCCIPsneonataisocluídos invitroporcoáguloe situou-setalconcentrac¸ãodentrodamargemdeseguranc¸aindicadanaliteraturacientífica. © 2015Sociedadede Pediatriade SãoPaulo. Publicado porElsevier Editora Ltda.Todosos direitosreservados.
Introduction
Theperipherallyinsertedcentralcatheter(PICC)hasbeen shown to be safe for intravenous infusion of solutions in neonates.1,2Ithasalowerincidenceofcomplicationswhen
comparedtoothercentralvenouscatheters,supportingthe
thesisthatitisasafeandusefuldevicetobeusedin
situa-tionstobeusedwhenvenousaccessislimitedanddifficult.3
Eventually, complications can occur, anticipating the
unscheduled removal of the catheter.2 Among the main
complications are obstructions, with rates that can vary
from 11% to 50%, and catheter rupture.2---8 Obstruction
maybecausedbythrombusformation,apoorlypositioned
cathetertipordrugprecipitation.9,10
These complications can be prevented and minimized
through specific interventions. There are many practices
relatedtomaintainingthePICCpermeability,althoughthere
islittlescientificevidenceonthebestthrombolyticagent,
aswellasitssafeandeffectiveconcentrationthatcan
sup-portasinglepractice.11---13
Although heparin is almost universally used in clinical
practice,itsbenefitshave not beenfirmly established,as
well as the effective and safe dose of this substance for
arterialandvenouscatheters inneonatology.12,13 Thelack
ofscientificevidenceandstandardizationthroughprotocols
leads totheuse of severalheparin concentrations,which
can often be abusive as well asbe as underdoses,which
can result in unknown side effects or failure in catheter
clearance.14
Inthiscontext,theaimofthisstudywastocomparetwo
different concentrations of sodium low molecular weight
heparin(SLMWH)regardingitsefficacytorestore the
per-meability of neonatal PICC obstructed by a clot in the
laboratory.
Method
This is an experimental, in vitro study of quantitative approach, developed in the Analysis Laboratory of a uni-versityhospitalin Londrina, stateof Paraná,fromJuly to December2013.Thestudysampleconsistedof76PICCused innewbornsadmittedattheNeonatalIntensiveCareUnit. Inthisunit, theindicationfor PICC useis the administra-tionofantibioticsforlongerthan7days,startofvasoactive drugs, need for glucose infusion rate (GIR) >7, and par-enteralnutrition;thus,allthecathetersusedinthisstudy receivedallorsomeofthesetherapies.Thecatheterswere obtainedfromtheNeonatalIntensiveCareUnit(NICU)ofthe above-mentioned hospital, after being removed from the newbornsbecauseoftheendofthetreatment.Afterthey wereremoved,thecatheterswereimmediatelywashedwith a10mLsyringe,filledwithdistilledwater, at leasttwice, untiltheywereclean,andtestedforintegrityand perme-ability,afterwhichtheywerestoredintheoriginalcatheter plasticpackaging.Thesecatheterswerestoredinacabinet awayfromlightormoistureforapproximately6months,the necessarytimetoobtainasufficientnumberofcathetersto startthestudy.
Samplecalculationwasobtainedthroughafile contain-ingtherecordofallcathetersusedinnewbornsintheNICU, whichhadthepatient’sname,catheterbrand,batch, cal-iber, reason for catheter installation, length of catheter introducedandreasonfor removal.Thedataspannedthe last6monthsof theyear priortocollection, i.e.,July to December2012.A totalof 68PICCswere removedduring thisperiodbecauseoftheendoftreatmentand/or obstruc-tions.Consideringasamplingerrorof5%,confidencelevel of95%and clearingdifferencebetweenGroupsIandII of 50%,thenecessityofhaving36cathetersineachgroupwas verified.
In most newborns, the length of the introduced PICC rangesfrom8to28cm,dependingonthenewborn’slength andthepuncturelocation.Consequently,forthestudy,the PICCswerecutwithascalpelataspecificmarkingoneach catheterthatindicated 11cm. Subsequently,the 76PICCs weredividedintotwogroupsof38catheterseach,randomly distributed,regardlessofstoragetime,astheyhadalready beentestedforpermeability,andwereproperlyidentified asGroupsIandII,followedbythenumericsequence1---38. The randomization was carried out throughdrawing lots. Forthatpurpose,theresearcherplaced76cathetersonthe workbenchandaskedthebiochemisttochoose38catheters, whichwerecalledGroupI,andtheother38cathetersleft ontheworkbench werecalledGroup II.These PICCswere coagulatedinvitrousinghumanbloodobtainedby venipunc-turefromtwohealthyemployees,whoacceptedtodonate thebloodandgavewrittenconsent,bothhavingbloodtype ORh+,hematocrit39.2%(donor1)and42.2%(donor2)and platelets410,000(donor1)and400,000(donor2), respec-tively---thatis,withinthenormalrange.Immediatelyafter collection,madewitha23Gscalpveinneedleanda10mL syringe,bloodwastransferredto1mLsyringes,andavolume of0.04mLwasintroducedintoeachofthecatheters,which werearrangedonasurgical field.This volumewas neces-sarybecausea10cmcatheterobstructionwasused,which correspondstoapproximately0.04mLoftheinternalvolume capacity(priming).Catheterobstructionwasperformedby twoprofessionalswithprovencapacityanddexterity,who werepreviously trainedto preventclotting of blood out-sidethecatheter.Themeantimebetweenbloodcollection andtheobstructionofeachcatheterwas2min.Afterfilling thecatheterswithblood,thedistalendwasconnectedto athree-way tap,which wasproperlyclosed, whereasthe proximalendwasoccluded withaKelly clamp toprevent the escape of blood or air-drying. The technique used in thisresearchtoperforminvitrocoagulationandthe perme-abilitytestingofthecatheterswasbasedonanotherstudy, whichaimedtotestanewtechniquetorestorepermeability ofPICCsoccludedbyclotsinthelaboratory.15
Then, each catheter wastransferred toand immersed
inacontainerwith100mLofsalineheatedto37◦Cinorder
tosimulatethebodytemperatureoftheneonate.Each
con-tainerhad14cathetersfromthesamegroup.Thecontainers
with the catheters were placed in a water bath (Quimis
AparelhosCientíficosLtda.),keepingthesolutionbetween
36.5and37◦C,withstricttemperaturecontrolthrough
ther-mometers immersed in the solution. The catheters were
immersed for 6h and then removed and cut at the 10cm
markingtoeliminateanydamagecausedtothecatheterby
thepreliminaryclamping.Subsequently,theirpermeability
wastestedthrougha1mLinfusionofsalinesolution,
consid-ering ascompletelyobstructedthosecathetersintowhich
it wasnot possible toinfuse the salinesolution andfrom
whichthebloodcouldnotbedrawnbythesyringe.Then,
thecatheterswereagainplacedinthewaterbathdevicefor
thestartoftheinfusionofbothheparinconcentrations.The
laboratoryroomwasusedexclusivelyfortheprocedure,and
noprofessionalsotherthantheresearcherswereallowedto
enter.The roomtemperaturewasmaintainedbetween 22
and24◦C,andtheperiodoftimeduringwhichthecatheters
remainedinthesalinesolutionwaschosensoastonotaffect
theresults.
The SLMWH (5000IU/mL) was used due to its
mecha-nismof action,it beingthemost oftenusedheparintype
in neonatal intensive care units, and having the lowest
cost. SLMWH exerts anticoagulant action by activation of
antithrombin III, which has its activity accelerated up to
1000 times in orderto inhibit coagulationfactors IIa and
Xaand,toalesserextent,IXa,XIaandXIIa.Heparin
bind-ingtoantithrombinIIIdependsonthepresenceofasingle
pentasaccharidesequencecontainedinapproximately
one-thirdofheparinmolecules.Theremainingtwo-thirdshave
minimal anticoagulant activity at usual therapeutic
con-centrations. This pentasaccharide sequence confers high
affinity of the SLMWH to the antithrombin III. Any
hep-arinorSLMWHmoleculecontainingthepentasaccharidecan
inhibit Xaaction simplyby activating AT III.To inactivate
thrombin (IIa), theSLMWH must bind totheantithrombin
IIIandthefactorIIasimultaneously,formingaternary
com-plexthat onlyoccurs withlongerchains,withat least 18
saccharides. In additionto decreasing the anti-IIa effect,
SLMWH has other pharmacokinetic advantages, such as
decreasedbindingtoplasmaproteinsandacutephase
pro-teins, decreased binding tomacrophages and endothelial
cells, decreased binding to platelets and platelet factor
4 (PF4), and decreased binding toosteoblasts. These
dif-ferences result in therapeutic advantages, such as more
predictableanticoagulantresponse,longerplasmahalf-life,
increased bioavailability, reducedheparin-induced
throm-bocytopeniaandreducedosteopenia.16
GroupIreceivedaninfusionofSLMWHataconcentration
of 25IU/mL,dilutedin saline solution,in a 10mLsyringe;
Group II received the same SLMWH at a concentration
of 50IU/mL, also diluted in 10mL saline solution. The
researcher was blinded to the choice of group to which
thecatheterbelonged.Twonursesparticipatedatthetime
of the experiment, one of whom wasresponsible for the
clearing of Group I catheters, whereas the other nurse
wasresponsible for Group II. Bothnurses weretrainedto
perform the experiment, and they had a time period of
3mintoperformthetechniqueforeachcatheter,whichwas
controlledbythebiochemistusingadigitaltimer.The
bio-chemistwasresponsiblefordeliveringthetwosyringes,one
withSLMWHwith25IU/mLandtheotherwith50IU/mL,and
onlyhehadknowledgeoftheconcentrationsusedineach
catheter. According to the literature, the recommended
intermittentdosesofheparinare50---100IU/kg/doseevery
4h, without causing significant changes in coagulation,17
which was not exceeded in this study, as the maximum
concentration of heparin was 50IU/mL every 4h in the
groupwiththehighest concentration.Thechoice ofusing
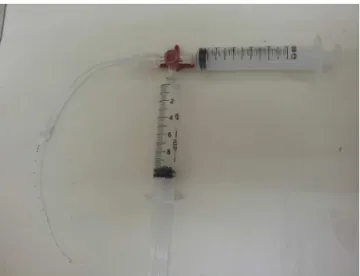
Figure1 Negativepressuretechnique.
literaturewasmade tomaintain asafedose andtoallow lowerdosestobeusedintheneonatalpopulation.
Thenegativepressuretechniquewasusedforthe clear-anceattempts,18asshowninFig.1.Thetechniqueinvolves
theuseofathree-waytap,connectedtothecatheter,with
two 10mL syringes connected to the tap, one containing
10mL ofheparinsolution accordingtothe group,and the
other one empty.The contentofthe catheterwasdrawn
tothe10mLgraduationusingtheemptysyringe,forminga
vacuuminside,withthisroutebeingclosedsoonafter.The
taproute containingthesolutionwasthenopened,aiming
tofillthe catheterwithjust thevolumeaspirated by the
vacuum. The system was thus blocked, and the negative
pressuretechniquewasrepeatedfourtimes,with5,15and
30min and4h.The same heparinsolutionwasusedin the
fourintervals.Alwayspriortosolutioninfusion,anattempt
toaspirate the blood clot and evaluate catheter patency
wasmade and, when cleared, subsequent tests were not
performed.
Attheendofeachattemptinbothgroups,theheparin
solutionwastotallyaspirated,whilealsotryingtoaspirate
the clot, and the permeability wastested again by using
1mLofsalinesolutioninfusionina10mLsyringe.Catheter
permeabilitywasconsideredtoberestoredwhenclot
suc-tioncapacitywaspossible, aswell astheinfusion of 1mL
ofsaline solutionfromthe distalendtotheproximalend
of thecatheter.To ensurethat catheterpermeabilityhad
been restored, the saline solution was disposed off in a
container withanaccurate 0.1mLgraduation,sothat the
researcherscouldverifywhethertheentireinfusedvolume
hadbeenreturnedtothecontainer.Toensurethatthe
infu-sion rateamong nurses wasasaccurate aspossible, they
used a digital timer manipulated by the biochemist, who
controlledthetimeof 1min for thesalineinfusion by the
professionals.Theexaminersresponsibleforthe
permeabil-itytestwerethenursesresponsibleforgroupsIandII,who
determinedwhetherthecatheterhadrecoveredits
perme-ability,whereasthebiochemistwasthesecond toconfirm
andrecordtheresult.
Ifcatheterpermeabilitywasnotachieved,thedosesof
heparinwererepeatedfivemoretimesatthetwoassessed
concentrations, with new dilutions every 4h, due to the
PICCs excluded according to the exclusion criteria: Catheters with length <11cm (n=4);
1.9Fr caliber catheters (n=2); Catheters of other brands (n=2) Non-coagulated catheters during follow-up
(n=2) Total excluded: 10
Analyzed PICCs (n=76)
PICCs assessed regarding the inclusion criteria: Catheters with a minimum length of 11cm;
2.0Fr caliber catheters; polyurethane catheters and single brand
(n=86)
Figure2 FlowchartofneonatalPICCeligibility.
timeofheparinstability. The maximum timefor catheter clearancewouldbe24haftertheinfusionofthefirst hep-arinconcentration.Theseintervalswereusedfollowingthe informalaccountof nursesof the above-mentionedNICU, whoalreadyusedtheminaprotocolforclearingcatheters withheparinconcentrationof50IU/mL.
The results were recorded in a previously structured formand entered in a spreadsheet using Microsoft Office Excel 2007. Later, survival analysis was performed using Kaplan---Meiercurve,andthelog-rank (Mantel---Cox), Bres-low (Generalized Wilcoxon) and Tarone---Ware tests were appliedtoverifythedevelopmentofthegroupsaccordingto thetimeintervals.Theconfidenceintervalwassetat95%.
The research was carried out after approval of
the Institutional Review Board of Universidade Estad-ual de Londrina, process n. CEP/UEL: 066/2013, CAAE: 13890613.8.0000.5231,and was performed in accordance withtherequiredethicalstandards.
Results
During the data collection period, the daily removal of PICCsat the NICU was followed, aiming to identify eligi-blecatheterstobeincludedinthisstudysample,asshown
inFig. 2.Finally, 76 catheters were included in the
sam-ple.Therefore,thestudygroupsconsistedof38catheters
eachfromthe samebrand,2.0-Fr caliberand 11cmlong,
withameantimeofuseof30days.Ofthese,35(46%)were
usedexclusively inpartial parenteralnutrition (PPN),and
41(54%)wereusedfor theinfusionofantibiotics,glucose
andelectrolytesolutions.
Thebehaviorofgroupsaccordingtothetimeintervalsis
depictedinFig.3,showingasignificantdifferencebetween
thebehaviorsofdifferentconcentrations,withlesstimeof
1.0
0.8
0.6
0.4
0.2
0.0
.00 50.00 100.00 150.00 200.00
Time
Cumulative survival
Functions of survival
Group 1 Group 2 Group
250.00
Figure3 Kaplan---Meierchartfortimeofclearanceof76PICCs filledbyclotsinthelaboratory,accordingtotwoheparin con-centrations.
ofheparin.Afterperformingthetest forsurvival distribu-tionequality for the different group levels (Table 1), we
obtainedp<0.001,confirmingthatGroupII,withthehigher
concentration,allowsclearanceinlesstime.
These data are reaffirmed in Table 2, which indicates
that when comparing the results of the two groups
dur-ingthe5-mintimeinterval,agreaternumberofcatheters
wascleared inGroupII (57.9%),whencomparedtoGroup
I (21.1%), whereas in the 4-h time interval the inverse
occurred,which allows usto conclude thatthe catheters
from Group II, which received the concentration of low
molecularweightheparinof50IU/mL,showedfaster
clear-ance than those from Group I, which were treated with
25IU/mLoflowmolecularweightheparin.
Table1 Testofsurvivaldistributionequalityforthe dif-ferentgrouplevels.
Chi-square df p-value LogRank(Mantel---Cox) 13.183 1 <0.001 Breslow(GeneralizedWilcoxon) 13.483 1 <0.001 Tarone---Ware 13.591 1 <0.001
Table2 ClearanceofPICCsobstructedbyclotsinthe lab-oratoryaccordingtotwoheparinconcentrations,duringthe 4daysofthestudy.Londrina,2013.
Timeinterval GroupI(25IU/mL) GroupII(50IU/mL)
n % n %
5min 08/38 21.1 22/38 57.9 15min 07/30 23.3 06/16 37.5 30min 05/23 21.7 05/10 50 4h 18/18 100 05/05 100 Total 38/38 100 38/38 100
Discussion
Intraluminalobstructionofacentralcathetermaybemore commoninpatientswithPICCsbecause,asinsertionoccurs through peripheral vessels that have a smaller caliberat the introduction,the catheteroccupiesmost ofthe lumi-naldiameter,hinderingbloodflow,allowingclotformation and,consequently,obstruction.Anotherfactorresponsible forthrombusformationistheinappropriatecatheter posi-tioning,which canlead itstiptoleanagainst thewall of the vessel,allowing obstruction tooccur. In addition,the selectedlimbmaycontributetothiscomplication,aslimb movement cancause catheterdislocation,facilitating the formationofclotsandobstruction.19Thus,thereisanoption
toperformmaneuversforclotclearance,astheycanoccur
even with adequate catheter handling and maintenance,
unlikethe obstruction duetocrystal formation caused by
incompatibledrugswithlowsolubility,whichisconsidered
aniatrogeniceventbynursingcare.
To simulate what occurs in vivo, it was decided,
dur-ingthecathetercoagulationprocess,toimmersethemina
solutionheatedat36---37◦C,inordertomimicthebody
tem-peratureofthenewborn,stipulatinga6-hperiodoftimeto
subsequentlytesttheirpermeability.Thistimeintervalwas
determinedtakingintoaccountoneofthemostwidespread
recommendations to maintain PICC permeability: flushing
thecatheteratpre-establishedintervals,usuallyevery8h.20
Consideringthenursingcareprocess,thecatheterisusually
flushedatevery shift.Forthisreason,duringthepractice
ofnursingcare,itisanimportantsteptocleanthecatheter
beforeandafterdrugadministration,whentheintervalfor
the administration of these drugs exceeds 6---8h; that is,
the team should be able to identify and start the
clear-ingmaneuverofanobstructedcatheterasearlyaspossible
afterthestartoftheevent.
When considering that the obstruction concept also
includestheslowedflowalongthecatheter,9onequestions
whethertheclearanceprocedurebymeansofnegative
pres-sure technique withheparinconcentration should alsobe
usedinthesecases.Itisknownthatthecathetermay
rup-ture if syringes with volumes <10mL are used, since the
lowerthevolumeofthe syringe,thegreater thepressure
exertedonthecatheter.Therefore,inthisstudy,we only
used10mLsyringes withthenegativepressuretechnique,
and at the time of the procedure, all the plungers were
pulleduptothemarkof10mL.Thus,thepressurewas
con-trolledand it wasassured thatall cathetersreceived the
samepressureduringtheclearanceprocess.
The action mechanism of the heparin depends on its
binding to a plasma cofactor, the antithrombin, thereby
inactivatingthrombin(factorIIa)andtheactivated
coagula-tionfactorX,preventingtheconversionofprothrombininto
thrombin,andoffibrinogenintofibrin.21Theadvantagesof
SLMWHinclude:nointerferenceof dietormedicationson
itsactionmechanismandminimummonitoring;when
com-paredtounfractionatedheparin,itdoesnotshowincreased
risk of bleeding, andit reducesthe incidenceof
heparin-inducedthrombocytopeniawhenusedalone.22
The concentrations of heparin (25 and 50IU/mL)
cho-sen for comparison were based on clinical and scientific
practiceregardingtheirclearanceefficacyandatthe
complicationsthatmayoccurduringtheiruseinneonates.17
Amongthestudiesusingdifferentconcentrationsof
contin-uousorintermittentheparininneonates,noadverseevents
werefound.12,13,23Thisstudydecidedtoconsiderthelowest
concentrationofheparinconsideredsafe,asitwascarried
outinvitroanditwasnotpossibletoverifytheoccurrence
ofadverseeventsinnewborns.
Thisstudy,however,testedtwodifferentconcentrations
ofheparinassociatedwiththenegativepressuretechnique.
Twocatheterclearancetechniqueswerefoundinthe
liter-ature:thenegativepressuretechnique,18describedearlier,
and the standard operating procedure (SOP) technique.15
In the SOP technique, the syringeplunger is pulled back
and released at regular intervals in the catheter,sending
a shock wave that displaces the clot from the catheter
lumen, allowing the thrombus to beaspirated.15 Because
the latter technique has been tested in 3.0 and 4.0-Fr
catheters,not usedin neonatology,itwasdecidedtouse
the negative pressure technique. It is noteworthy that
only10mLsyringeswereusedinallproceduresperformed
withcatheters, according to published recommendations,
aslower-volume syringes have greater intravascular
pres-sure,increasingtheoccurrenceofadverseevents, suchas
thecatheterrupture.4,24Catheterclearanceattemptsmade
by means of positive pressure, even with the infusion of
thrombolyticagents,cancausecatheterruptureduetohigh
resistance.Therefore,thenegativepressuretechniquewas
used,whichprovidescontactofthethrombolyticagentwith
theclotwithoutcausingpositivepressure,whichfacilitates
clotremoval.13
It shouldbeemphasizedthat,inGroup II,heparinat a
higherconcentrationwasmoreeffectiveincatheter
clear-ance,asthelatteroccurredwithinashorterperiodoftime
whencomparedtoGroupI.Perhaps,theinterventions
per-formed resulted from the association of heparinwith the
negative pressure technique; however, one cannot make
thisclaim,asthe studywasnotdesignedtotestthe
neg-ativepressuretechniqueforcatheterclearance.Duringthe
experiment,however, thecriteriarelated tothenegative
pressure technique were maintained, such as using only
10mLsyringes, whichwasconsidered safer, attemptingto
removetheclotusingtheemptysyringebeforethesolution
infusion,maintainingtheobstructedcatheterimmersedin
solutionforatleast5minbeforeattemptingsuction,which
istheminimumtimeforheparintoact,anddetachingthe
clotfromthelumencatheter.Itisworthmentioningthatthe
clotisnotdilutedbytheheparinaction,butthelatterallows
theclottodetachitselffromthecatheterwall---hencethe
importanceofaspiratingittopreventfurtherobstructionor
theintroductionoftheclotintothenewborn’sbloodstream.
The fact that the clots from all catheters were
aspi-ratedintothesyringebeforethepermeabilitytestdoesnot
allowustoaffirmtheefficacyandsafetyoftheexperiment.
Thenegativepressuretechniqueassociatedwiththetested
heparin concentrations led tothe restoration of catheter
permeability and thrombus aspiration visualization,
pre-venting it from being introduced into the bloodstream,
which can lead to severe consequences for the neonate.
Althoughnoclotwasvisualizedafterthesalineinfusion
dur-ingthepermeabilitytest,itisnotpossibletoaffirmthetotal
absenceofbloodclots,asthesolutionwasnotfilteredand
analyzedmicroscopically.
Onemustrememberthatthemeasuresusedtomaintain
PICCpermeabilityareessentialforappropriateintravenous
therapy. However, when catheter obstruction eventually
occurs,manydamagestothenewbornsareidentified,such
astheneedforrepeatedpunctures,whicharepainfuland
canbedifficult,discontinuationofdrugtherapyor
contin-uous parenteral nutrition, more catheter handling, which
predisposestoinfection,andincreasedstresslevelsforthe
newbornandthestaff.
Itshouldbeemphasizedthat,althoughthestudysought
tosimulate theclinical condition ofthecatheterintothe
newborn’svessel,itwascarriedoutinalaboratory,which
bringssomelimitationsduetotheabsenceoffactorspresent
inthebloodstream,suchastheturbulenceofbloodwithin
thevessels.Anotherstudylimitationwastheimpossibility
ofverifyingmicroclotdetachmentfromthecatheterafter
thesalineinfusiontoverifythecatheter’spermeability,and
thus,other studies arenecessary toallowthis solutionto
befiltered andanalyzed microscopically, asthe presence
ofmicroclots couldtrigger newobstructions or
emboliza-tionintothebloodstream.Anotherlimitationofthestudyis
relatedtoprevioususeofthecatheters,asseveralofthem
mightbemorelikelytohaveclotsmoreorlessadheredto
them,astheywereusedindifferentneonatesforinfusion
ofPPN,antibiotics,andglucoseand electrolytesolutions,
whichwouldnotdependonthetypeofheparinused.
Forthenursingstafftoobtainsuccessfulcatheter
implan-tation,theyshouldbeawareoftherisksinvolvedinitsuse.
NursingcareisessentialtomaintainthePICC,andthe
identi-ficationofpossiblecomplicationsrelatedtoitsusebecomes
anecessityforprofessionalswhoworkdirectlywithcatheter
handling.10Thus,scientificevidenceresearchisessentialfor
the decision-making process of optimizing the PICC,
par-ticularly regarding what is the most effective solution in
preventingcatheterobstruction.25
Itcanbeconcludedthattheuseoflowmolecularweight
heparinataconcentrationof50IU/mLwasmoreeffective
inrestoringpermeabilityofneonatalPICCs(2.0Fr)occluded
invitrobyaclotthanthe25IU/mLconcentration,
empha-sizingthattheseconcentrations arestillwithinthesafety
marginindicatedintheliterature.
Funding
Thisstudydidnotreceivefunding.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
References
1.ReisAT,SantosSB,BarretoJM,daSilvaGR.Peripherally-inserted centralcatheteruseinneonatalclientsatapublicstate hospi-tal:retrospectivestudy.RevEnfermUERJ.2011;19:592---7. 2.CostaP,KimuraAF,deVizzottoMP,deCastroTE,WestA,Dorea
E.Prevalenceandreasonsfornon-electiveremovalof peripher-allyinsertedcentralcatheterinneonates.RevGauchaEnferm. 2012;33:126---33.
neonatalandpediatricintensivecareunit.RevEletronEnferm. 2012;14:883---92[serialontheInternet].Availablefrom:http:// www.revistas.ufg.br/index.php/fen/article/view/14432/13353 [accessed28.11.13].
4.DóreaE,deCastroTE,CostaP,KimuraAF,dosSantosFM. Man-agementpracticesofperipherallyinsertedcentralcatheterat aneonatalunit.RevBrasEnferm.2011;64:997---1002. 5.BaggioMA,BazziFC,BilibioCA.Peripherallyinsertedcentral
catheter:descriptionofitsuseinneonatalandpediatricICU. RevGauchaEnferm.2010;31:70---6.
6.FranceschiAT,CunhaML.Adverseeventsrelatedtotheuseof centralvenouscathetersinhospitalizednewborns.Rev Latino-AmEnferm.2010;18:57---63.
7.MottaPN,FialhoFA,DiasIM,NascimentoL.Catetercentralde inserc¸ãoperiférica:opapeldaenfermagemnasuautilizac¸ão emneonatologia.HURev.2011;37:163---8.
8.FreitasEM,NunesAB.Thenurseinthepracticeof peripher-allyinsertedcentralcatheterinneonatalcare.RevMinEnferm. 2009;13:215---24.
9.NakazawaN.Infectiousandthromboticcomplicationsofcentral venouscatheters.SeminOncolNurs.2010;26:121---31. 10.Swerts CA, Felipe AO, RochaKM, Andrade CU. Nursing care
in viewof complications regardingthe peripherallyinserted catheter in newborns. Rev Eletron Enferm. 2013;15:156---62 [serialontheInternet]. Availablefrom:http://www.fen.ufg. br/fenrevista/v15/n1/pdf/v15n1a18.pdf[accessed18.09.14]. 11.ShahPS,NgE,SinhaAK.Heparinforprolongingperipheral intra-venouscatheteruseinneonates.CochraneDatabaseSystRev. 2005;4:CD002774.
12.Shah PS, Shah VS. Continuous heparin infusion to prevent thrombosisandcatheterocclusioninneonateswith peripher-allyplacedpercutaneouscentralvenouscatheters.Cochrane DatabaseSystRev.2008;2:CD002772.
13.Araujo OR, Araujo MC, Silva JS, Barros MM. Intermittent heparinisnoteffectiveatpreventingtheocclusionof periph-erallyinsertedcentralvenouscathetersinpretermandterm neonates.RevBrasTerIntensiva.2011;23:335---40.
14.JohannDA,LazzariLS,PedroloE,MingoranceP,AlmeidaTQ, Dansski MT. Peripherally inserted central catheter care in neonates: an integrative literature review. Rev Esc Enferm USP.2012;46:1503---11[serialontheInternet].Availablefrom:
http://www.scielo.br/scielo.php?pid=S0080-623420120006000 30&script=sciarttext&tlng=en[accessed10.07.13].
15.FetzerSJ,ManningGP.SafetyandefficacyofthePOPtechnique forrestoringpatencytooccludedPICcatheters.ApplNursRes. 2004;17:297---300.
16.AroneKM,OliveiraCZ,GarbinLM,ReisPE,GalvãoGM,Silveira RC.Thromboticobstructionofthecentralvenouscatheterin patientsundergoing hematopoieticstemcelltransplantation. RevLatino-AmEnferm.2012;20:1---9.
17.Macdonald MG, Mullett MD, Seshia MM. Avery neonatologia: fisiopatologiaetratamentodorecém-nascido.6thed.Riode Janeiro:GuanabaraKoogan;2010.
18.Sharma RP, Ree CJ. New technique for declotting central venouscatheters(CVCs)byrecoveryroomnurse.IntJAngiol. 2003;12:59---61.
19.ChopraV,AnandS,HicknerA,BuistM,RogersMA,SaintS,etal. Riskofvenousthromboembolismassociatedwithperipherally inserted central catheters: a systematic review and meta-analysis.Lancet.2013;382:311---25.
20.SchallomME,PrenticeD,SonaC,MicekSt,SkrupkyLP.Heparin or0.9%sodiumchloridetomaintain centralvenouscatheter patency:arandomizedtrial.CritCareMed.2012;40:1820---6. 21.JunqueiraDR,CarvalhoMG,PeriniE.Heparin-induced
thrombo-cytopenia:areviewofconceptsregardingadangerousadverse drugreaction.RevAssocMedBras.2013;59:161---6.
22.Ignjatovic V,Najid S, Newall F, Summerhayes R, Monagle P. Dosingand monitoring of enoxaparin (low molecular weight heparin)therapyinchildren.BrJHaematol.2010;149:734---8. 23.BirchP,OgdenS,HewsonM.Arandomisedcontrolledtrialof
heparinintotalparenteralnutritiontopreventsepsis associ-atedwithneonatal long lines:theheparinin long linetotal parenteralnutrition(HILLTOP)trial.ArchDisChildFetal Neona-talEd.2010;95:F252---7.
24.BertoglioS,SolariN,MeszarosP,VassalaoF,BonventoM, Pas-torino S, et al. Efficacyof normal versus heparinized saline solutionforlockingcathetersoftotallyimplantablelong-term centralvascularaccessdevicesinadultcancerpatients.Cancer Nurs.2012;35:E35---42.