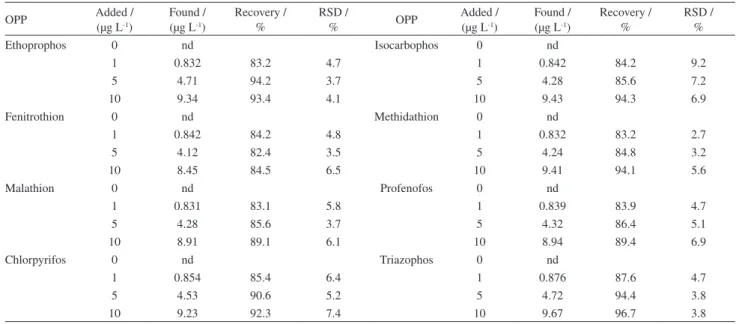
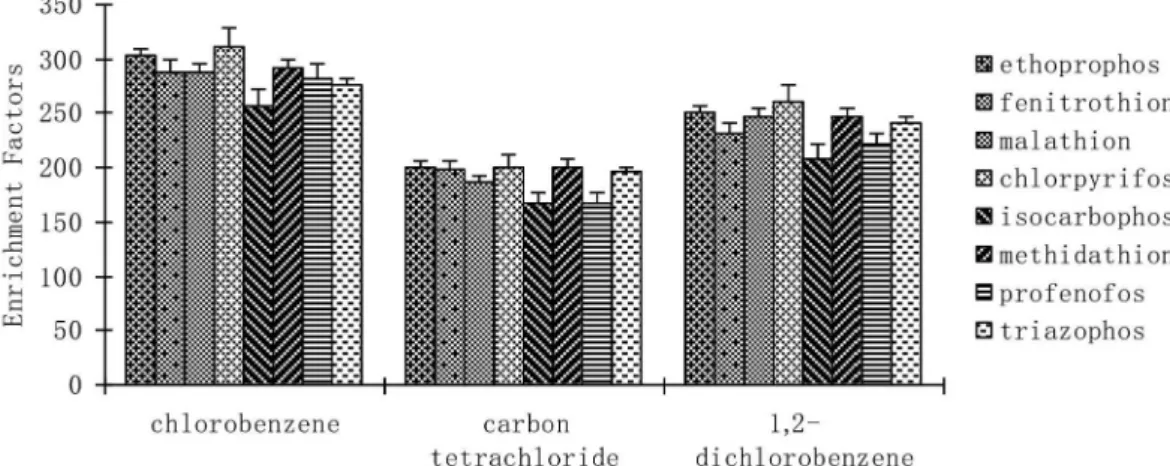
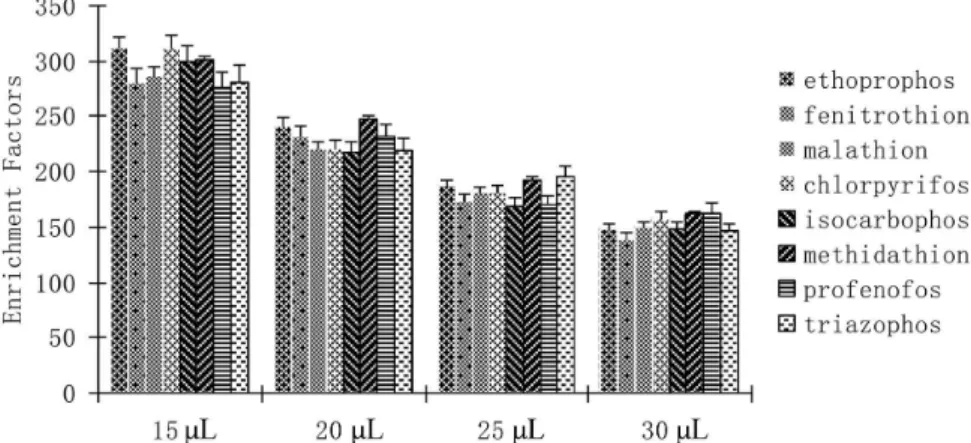
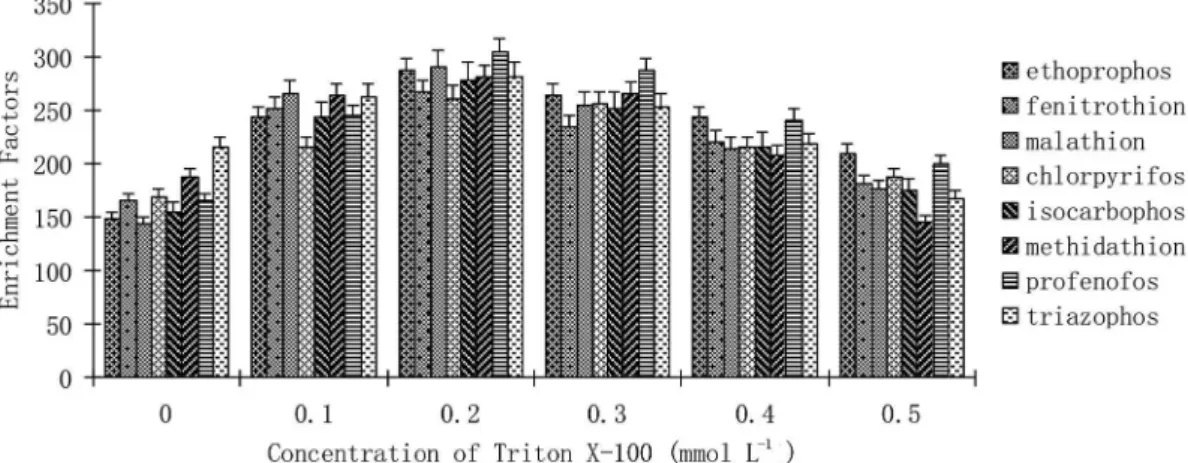
J. Braz. Chem. Soc. vol.28 número12
Texto
Imagem
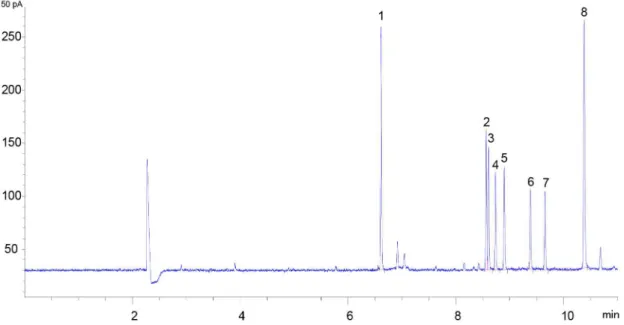
Documentos relacionados
A drawback of deterministic formulations, in the context of HEMS scheduling, is the lack of precise knowledge con- cerning demand, local micro generation or grid prices over
Feature extraction was computed with features from all four domains (statistical, time, frequency and time-frequency) whose output was used as input of the unsuper- vised
A estética do mundo cor de rosa da Barbie encontrou na técnica de design de superfície da estamparia o caminho para representação dos signos ligados à Barbie,
In particular, DPPH radical is widely used for quickly assessing the ability of antioxidants to transfer labile H atoms to radicals (Brand-Williams et al. Following a similar line
In the absence of demographic parameters that can evidence source and sink patches, spatial models that predict occurrence and mortality can provide valuable
The homogenate extraction method was compared with traditional heating extraction method, and the antioxidant activity of crude Lentinus edodes polysaccharides was investigated..
The first group of animals was treated with iodine tincture at 10% (IT), the second group was treated with sodium hypochlorite at 2.5% (SH) and the third group was used as
e entre a Escala de Atitudes face à saúde e a Escala de Comportamentos de Saúde e de Risco, não podemos esqueoer que existem algumas conelações significativas entre