rev bras hematol hemoter. 2016;38(4):298–301
w w w . r b h h . o r g
Revista
Brasileira
de
Hematologia
e
Hemoterapia
Brazilian
Journal
of
Hematology
and
Hemotherapy
Original
article
Herpes
zoster
after
autologous
hematopoietic
stem
cell
transplantation
Kelli
Borges
dos
Santos
∗,
Rafaela
Souto
e
Souza,
Angelo
Atalla,
Abrahão
Elias
Hallack-Neto
UniversidadeFederaldeJuizdeFora(UFJF),JuizdeFora,MG,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received27July2015 Accepted18May2016 Availableonline12July2016
Keywords:
Transplantofhematopoieticstem cells
Autologous Herpeszoster Preventionandcontrol
a
b
s
t
r
a
c
t
Background:The autologoushematopoieticstem celltransplantationprocedureinvolves immunosuppressionofthepatient.Thus,thepatienthasanelevatedriskforseveral dis-eases,suchasinfectionswiththevaricella-zostervirus.Preventionprotocolshavebeen proposedbasedontheuseofacyclovirfromthefirstdayofconditioning,andmaintaining thisdrugfor30–100daysaftertheprocedureorforasmuchasoneyear.Theobjectiveof thisworkwastoevaluatetheincidenceofherpeszosterafterautologoustransplantations relatedtotheearlysuspensionofacyclovir.
Methods:Aretrospectivestudywascarriedoutbasedonthecollectionofdatafrom231 medicalrecordsoftransplantpatientsintheBoneMarrowTransplantUnitoftheteaching hospitaloftheUniversidadeFederaldeJuizdeForaintheperiodbetween2004and2014.
Results:Fourteen(6.1%)patientshadherpeszosterinthepost-transplantperiodonaverage withinsixmonthsoftheprocedure.Patientswithmultiplemyeloma(64.3%)werethemost affected.Therewasastatisticallysignificantdifferenceintheageofthepatients,witholder individualshavingagreaterchanceofdevelopingtheinfection(p-value=0.002).Therewere nosignificantdifferencesfortheothervariablesanalyzed.
Conclusion:Theearlysuspensionofacyclovircanbesafeinpatientswhoreceive autolo-goushematopoieticstemcelltransplants.Howeversomegroupsmaybenefitfromextended prophylaxiswithacyclovir,particularlyolderpatientsandpatientswithmultiplemyeloma. ©2016Associac¸ ˜aoBrasileiradeHematologia,HemoterapiaeTerapiaCelular.Published byElsevierEditoraLtda.ThisisanopenaccessarticleundertheCCBY-NC-NDlicense
(http://creativecommons.org/licenses/by-nc-nd/4.0/).
Introduction
Autologous hematopoieticstemcell transplants (HSCT)are usuallyrecommendedasarecoverytherapyforpatientswho receivemyeloablativechemotherapy.1 The
immunosuppres-sioncausedbytheconditioningleavesthepatientatahigh
∗ Correspondingautorat:RuaDeputadoLahyrTostes,365,SpinaVille,SãoPedro,36037-754JuizdeFora,MG,Brazil.
E-mailaddress:[email protected](K.B.Santos).
risk ofacquiringdifferent types ofdiseases.Infections are an important cause of morbidity in this process.2 These
patientsthereforereceiveprophylacticmedications,themost common of which are antibiotic, antiviral and antifungal agents.
Viruses,usuallyoftheherpesfamily,suchastheherpes simplexvirus(HSV),cytomegalovirus(CMV)orvaricella-zoster
http://dx.doi.org/10.1016/j.bjhh.2016.05.015
revbrashematolhemoter.2016;38(4):298–301
299
virus(VZV),aresomeofthemostcommoncausesofinfections intheperiodafterHSCT.3
Herpes zoster (HZ) is a painful vesicular eruption that istypically restricted toone or two dermatomes. Itis the result of the reactivation of latent VZV virus in nervous ganglia,usuallymanyyearsaftertheprimaryinfection.4
Infec-tionscausedbythevaricella-zostervirusarequitecommon afterHSCT, occurring inapproximately 20–30% ofpatients submitted toautologous transplants within oneyear after transplantation.4–6 Studieshaveshownthatthe occurrence
ofHZismorecommonafterthethirdmonthaftertransplant, withanincidencepeakinthefourthmonth.5
Theuseofacyclovirasprophylaxisagainstthereactivation ofherpesisconsideredstandardcareduringneutropeniain autologousHSCTpatients.7 Different protocols includethe
prophylacticuseofacyclovironthefirstdayof chemother-apy(conditioning),maintainingitsuseuntilDay30–100after HSCTintheabsenceofimmunosuppression.8
In the Bone Marrow Transplant Unit of the Universi-dadeFederaldeJuizdeFora(UFJF),thestartofprophylaxis withacycloviroccursonthefirstdayofchemotherapy,and is suspended when the neutrophil count is greater than 500cells/mm3,i.e.whenengraftmentoccurs,resultinginless
timeofusethanrecommended.Thepresentstudytherefore aimstoevaluatetheincidenceofinfectionbytheherpesvirus inpatientssubmittedtoautologousHSCTinrespecttothe earlysuspensionofacyclovir.
Methods
This retrospective study compared the incidence ofHZ in patientssubmittedtoautologousHSCTwithearly interrup-tionofprophylacticacyclovircomparedtotheusualscheme ofoneyearreportedintheliterature.
Themedicalrecordsof221patientssubmittedtoHSCTin theperiodbetween2004and2014attheBoneMarrow Trans-plantationUnitoftheUFJF,wereanalyzedretrospectively.Of these,ninepatientsunderwenttwotransplants,totaling230 procedures.
Datacollectionandvariables
Thedatawerecollectedfrommedicalrecords.Datacollection occurredintheperiodfromMarchtoSeptember2014.Forthe characterizationofthepopulation,datawascollectedrelated togender,age,diagnosis,presenceorabsenceofdiabetes,in additiontotheoccurrenceofdeath.Additionally,the occur-renceofherpesbeforeandafterHSCTandothervariablesof interestwerestudiedincludingthetimeofuseofacyclovir, durationofneutropenia,lengthofhospitalization,useof cor-ticosteroidsorthalidomideafteranoutbreakofherpes,and thetimeofonsetofthediseaseafterthetransplant.Although somerecordspresentedpriorserologyforHZ,manypatients werenottestedandthustheanalysisofthisvariablewasnot performed.
AllpatientssubmittedtoautologousBMT,independentof thebaselinedisease,receivedprophylacticacycloviratadose of500mg/m2/daydividedin2–4dosesperdayaccordingto
theperiodinwhichtheprocedurewasperformed.Prophylaxis
wasstartingonthefirstdayofconditioningandsuspended whentheneutrophilcountwasgreaterthan500cells/mm3.
Afterhospitaldischarge,thepatientsweremonitoredon anoutpatientbasisforaperiodof24monthsinthetransplant serviceoftheUFJF.
Data collection was only started after approval by
the Research Ethics Committee of the UFJF (# CAAE
25735614.3.0000.5133).
Statistical
analysis
ThedatawereanalyzedusingtheStatisticalPackageforthe SocialSciencessoftware(version19.0forWindows).The Chi-squaretestwasusedforcategoricalvariablesandStudent’s
t-testfornumericalvariables,meansandmedians.Ap-value <0.05wasconsideredstatisticallysignificant.
Results
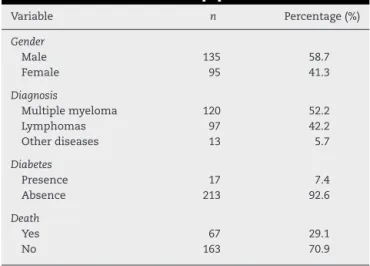
Twohundredandthirtymedicalrecordswereanalyzed.The mean age was 48.73years (range: 4–79) and mostpatients were male(58.7%).Themostcommonlyfounddiagnosisin thetransplantservicewasmultiplemyeloma,totaling52.2%of hospitaladmissions.Theaveragelengthofhospitalstayofthe patientswas20.78days.Thecharacteristicsofthepopulation aredescribedinTable1.
Fewpatientshadsometypeofassociatedcomorbidity,with diabetesmellitusbeingthemostcommon,occurringin7.4% (n=17)ofthepatients.
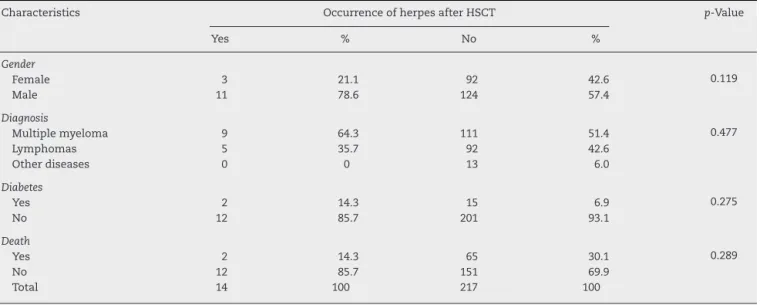
Eightpatients(3.5%)hadhadHZbeforehospitalizationto performtheHSCT,onlyoneofwhom(0.43%)alsopresentedHZ aftertheHSCT.Fourteen(6.1%)patientspresentedHZafterthe HSCT;therewerenosignificantdifferencesintheevaluated variablesbetweenpatientswho hadHZaftertheHSCTand thosethatdidnot(Table2).Theaveragetimeofonsetofthe HZoutbreakwas164.6daysafterthetransplant(median:144; range:49–330days).
Ofthe14patientswhohadHZinthepost-HSCTperiod, onlyonewastakingcorticosteroids/thalidomideduringthe onsetoftheinfection(7.21%).Furthermore,onlyonepatient
Table1–Characteristicsofthepopulation.
Variable n Percentage(%)
Gender
Male 135 58.7
Female 95 41.3
Diagnosis
Multiplemyeloma 120 52.2
Lymphomas 97 42.2
Otherdiseases 13 5.7
Diabetes
Presence 17 7.4
Absence 213 92.6
Death
Yes 67 29.1
300
revbrashematolhemoter.2016;38(4):298–301Table2–Occurrenceofherpesaftertheautologoushematopoieticstemcelltransplantandcharacteristicsofthe population.
Characteristics OccurrenceofherpesafterHSCT p-Value
Yes % No %
Gender
Female 3 21.1 92 42.6 0.119
Male 11 78.6 124 57.4
Diagnosis
Multiplemyeloma 9 64.3 111 51.4 0.477
Lymphomas 5 35.7 92 42.6
Otherdiseases 0 0 13 6.0
Diabetes
Yes 2 14.3 15 6.9 0.275
No 12 85.7 201 93.1
Death
Yes 2 14.3 65 30.1 0.289
No 12 85.7 151 69.9
Total 14 100 217 100
Table3–Occurrenceofherpesaftertheautologous hematopoieticstemcelltransplantregardingage, durationofacyclovirtreatmentandneutropenia.
Characteristics Mean p-Value
Age
Herpesyes 57.14 0.001
Herpesno 48.19
Prophylacticuseofacyclovirindays
Herpesyes 18.57 0.690
Herpesno 19.13
Durationofneutropeniaindays
Herpesyes 8.82 0.350
Herpesno 9.15
(7.21%)hada relapseofthe disease.Moreover,onepatient incurred the infection during mobilization, prior to HSCT. MostpatientswhodevelopedHZwerepatientswith multi-plemyeloma(64.3%).Table3listssomecharacteristicsrelated totheincidenceofHZ.
Astatisticallysignificantdifferencewasobservedregarding theageofthepatientswhohadherpesandthosewhodidnot developit,witholderpatientshavingahigherlikelihoodof developingHZ.Consideringonlythepatientswithlymphoma ormyelomatogetherwithHZ,thedifferenceinagewasnot significant;thefirstgrouphadameanageof52.40yearsand thesecond,59.78years(p-value=0.233).
Discussion
TheprophylacticuseofacyclovirintheBoneMarrow trans-plantUnitoftheUFJFstartsonthefirstdayofchemotherapy andendswhenengraftmentoccurs.Fourteenpatients(6.1%) hadoutbreaksofHZafterHSCT.Differentprotocolsprovide for the prophylactic use of acyclovir from the first day of chemotherapy(conditioning)toDay30orDay100afterHSCT intheabsenceofimmunosuppression,andpossiblyaslong asoneyearofprophylaxis.7,8
However,thereisinsufficientevidencetostronglysupport theprolongeduseofacyclovirinautologousHSCT,orto sug-gest that its effectivenessoutweighs the potential adverse consequences.5,9 Studies havereportedthatthecontinuous
useof400mg/dayofacycloviruntiltheendof immunosup-pressive therapymay notsuppressthe reactivationofVZV afterthediscontinuationofacyclovir.6Theprophylactic
bene-fitsshouldthereforebeweighedagainstthetoxicity,costand riskofinducingresistance.Intheanalysisofallogeneic trans-plantations,however,theuseofacyclovirforuptooneyear provedtobeeffectiveinthepreventionofthereactivationof VZV.6,8,10Intransplantsperformedusingumbilicalcordstem
cells,anincreasedincidenceofVZVwasalsodemonstrated (46%)therebyjustifyingextendedprophylaxis.11
Data fromtheliteraturedemonstratedtheprobabilityof reactivationofVZVin8.2%ofthepatientswhoreceivedlow prophylactic doses of acyclovir for one year.12 In patients
who didnot receive acyclovir or who tookit forashorter period(untiltheendofneutropenia),theratesincreasedto 21–25%.3,12 Inthe current study,14 patientshad outbreaks
ofHZafterHSCT,withanincidenceof6.1%,thatis,alower valuethanthatfoundintheliterature.Thisfactorindicates thatearlysuspensionofacyclovirmaybeconsideredin sta-blepatients.However,thepresentstudyhasthelimitationof beingretrospective.
Onestudyshowedthattheunderlyingdiseasemightbea riskfactorforthedevelopmentofHZ.Inastudyconducted bySchuchteretal.,3theinfectionoccurredmainlyinpatients
withHodgkin’sornon-Hodgkinlymphoma(46%),compared withleukemia(20%)andsolidtumors(9%),differingfromthis study,inwhichthegreatestnumberofcases,albeit insignif-icant,occurredwithpatientswithmultiplemyeloma(64.3%), followedbylymphoma(35.7%).
revbrashematolhemoter.2016;38(4):298–301
301
Theuseofthalidomidehasapotential immunosuppress-iveeffect.Onecasestudyreportedadisseminatedinfection causedbyherpessimplexandaninfectionwithVZVfiveyears afterautologousHSCTinapatienttakingthalidomide.14In
thisstudy,however,onlyoneofthepatientswhohadHZtook thismedication.
Althoughnoserologyisperformedbeforethetransplant, apriorhistoryofHZhasastrongrelationshipwiththe posi-tivityoftheserologictesting.15Inthisspecificcase,onlyone
oftheeightpatientswhohadhadHZpriortothetransplant sufferedarecurrenceofthedisease.Thisleadsustobelieve thatperformingserologyforHZbeforetheHSCTmaynotbe necessary,asthiswouldindicatetheextendeduseofacyclovir inpatientswithpositiveresults.
Newmeasures,suchasvaccinationagainstHZ,arebeing implemented.Previousstudies haveshownthatthe results ofthis measuremaybenegligible,sincethevaccine isnot recommendedwithinthefirstyearaftertransplant(live atten-uated vaccine) and the incidenceof VZV israre after this period.15Morerecentstudies,however,usingimmunization
twomonthsafterHSCTwithanadjuvantsubunitof varicella-zostervirusglycoproteinE,haveshownittobeareasonable strategytoreduceHZinautologousHSCTpatients.4
Eventhoughitwasnotinthescopeofthisarticletoassess thesurvivalrateofpatients,themortalityrate,regardlessofits cause,wasnotinfluencedbytheoccurrenceofHZinfection.
Althoughthis work evaluated the records of230 trans-plants, it was necessary to compare the data found with studiescarriedout inother institutionsbecausethiswas a singlecenterstudy.Weconcludethattheearlysuspensionof acyclovircanbesafeinlow-riskpatientssubmittedto autolo-gousHSCT.Thismakesitpossibletoreducenotonlytherisk ofresistancetomedication,butthetreatmentcosts.However, somegroupsmaybenefitfromextendedprophylaxiswith acy-clovir,particularlyolderpatientsandpatientswithmultiple myeloma.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
r
e
f
e
r
e
n
c
e
s
1. MendesED.PerfilCli ´nicoeEpidemiolo ´gicodospacientes
submetidosaTransplantedeCélulasHematopoiéticasno
Servic¸odeHematologiadoHospitaldasCli ´nicas/FMUSPde
2001a2009.TrabalhodeConclusãodeCurso:Programade
residênciamédicaeminfectologiadaUSP;2010.
2. SrinivasanA,McLaughlinL,WangC,SrivastavaDK,Shook
DR,LeungW,etal.Earlyinfectionsafterautologous
hematopoieticstemcelltransplantationinchildrenand
adolescents:theSt.Judeexperience.TransplInfectDis.
2014;16(1):90–7.
3.SchuchterLM,WingardJR,PiantadosiS,BurnsWH,Santos
GW,SaralR.Herpeszosterinfectionafterautologousbone
marrowtransplantation.Blood.1989;74(4):1424–7.
4.StadtmauerEA,SullivanKM,MartyFM,DadwalSS,
PapanicolaouGA,SheaTC,etal.Aphase
½
studyofanadjuvantedvaricella-zostervirussubunitvaccinein
autologoushematopoieticcelltransplantrecipients.Blood.
2014;124(19):2921–9.
5.NucciM,MaiolinoA.Infecc¸õesemtransplantedemedula
óssea.Medicina(RibeirãoPreto).2000;33(3):278–93.
6.Asano-MoriY,KandaY,OshimaK,KakoS,ShinoharaA,
NakasoneH,etal.Long-termultra-low-doseacycloviragainst
varicella-zostervirusreactivationafterallogeneic
hematopoieticstemcelltransplantation.AmJHematol.
2008;83(6):472–6.
7.CDCguidelinesforpreventingopportunisticinfections
amonghematopoieticstemcelltransplantrecipients.MMWR
MorbMortalWklyRep.2000;49(RR10):1–128.
8.TomblynM,ChillerT,EinseleH,GressR,SepkowitzK,Storek
J,etal.Guidelinesforpreventinginfectiouscomplications
amonghematopoieticcelltransplantationrecipients:aglobal
perspective.BiolBloodMarrowTransplant.
2009;15(10):1143–238.
9.LeungTF,ChikKW,LiCK,LaiH,ShingMM,ChanPK,etal.
Incidence,riskfactorsandoutcomeofvaricella-zostervirus
infectioninchildrenafterhaematopoieticstemcell
transplantation.BoneMarrowTransplant.2000;25(2):167–72.
10.BoeckhM,HyungW,KimHW,FlowersME,MeyersJD,Bowden
RA.Long-termacyclovirforpreventionofvaricellazoster
virusdiseaseafterallogeneichematopoieticcell
transplantation–arandomizeddouble-blind
placebo-controlledstudy.Blood.2006;107(5):1800–5.
11.VandenboschK,OvetchkineP,ChampagneMA,HaddadE,
AlexandrovL,DuvalM.Varicella-zostervirusdiseaseismore
frequentaftercordbloodthanafterbonemarrow
transplantation.BiolBloodMarrowTransplant.
2008;14(8):867–71.
12.ErardV,GuthrieKA,VarleyC,HeugelJ,WaldA,FlowersME,
etal.One-yearacyclovirprophylaxisforpreventing
varicella-zostervirusdiseaseafterhematopoieticcell
transplantation:noevidenceofreboundvaricella-zoster
virusdiseaseafterdrugdiscontinuation.Blood.2007;110(8):
3071–7.
13.StyczynskiJ,ReusserP,EinseleH,delaCamaraR,Cordonnier
C,WardKN,etal.ManagementofHSV,VZVandEBV
infectionsinpatientswithhematologicalmalignanciesand
afterSCT:guidelinesfromtheSecondEuropeanConference
onInfectionsinLeukemia.BoneMarrowTransplant.
2009;43(10):757–70.
14.CurleyMJ,HusseinSA,HassounPM.Disseminatedherpes
simplexvirusandvaricellazosterviruscoinfectionina
patienttakingthalidomideforrelapsedmultiplemyeloma.J
ClinMicrobiol.2002;40(6):2302–4.
15.GarnicaM,MachadoC,CappellanoP,CarvalhoVV,NicolatoA,
CunhaCA,etal.Recomendac¸õesnomanejodascomplicac¸ões
infecciosasnotransplantedecélulas-troncohematopoéticas.