Braz. Dent. J. vol.26 número6
Texto
Imagem


Documentos relacionados
Thus, the method using micro-CT images showed consistent results to measure small changes in length and could be considered to evaluate the abutment screw elongation.. Within
The present study aimed to investigate whether human gingival fibroblasts would increase their proliferation potential and extracellular matrix protein expression in the presence
The evaluated Parameters included: total pore count, total pore volume, mean pore volume, total porosity (% of pore volume in relation to total sealer volume) and mean pore
The amount of remaining filling material after using ProTaper Retreatment was similar to that achieved with rotary and reciprocating files and after PUI (p>0.05).. Rotary
The following data were collected and evaluated: tooth type; type of endodontic treatment performed (treatment or re-treatment); amount of remaining coronal dentin after
The main finding of the present study pointed out a similar shaping ability between the reciprocating systems (Reciproc and WaveOne) and the conventional multi-file rotary
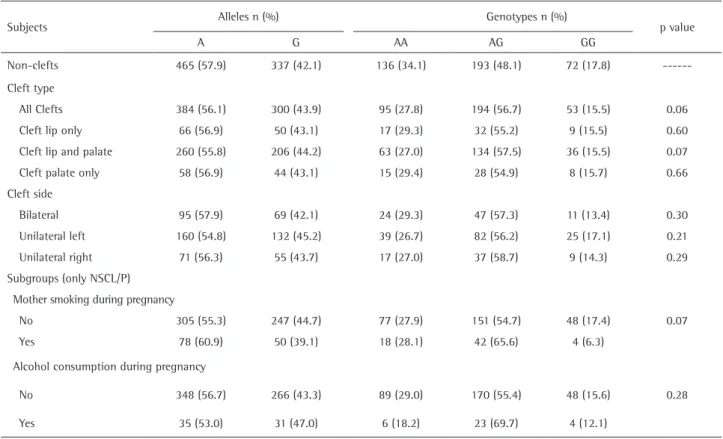
Association study of folate-related enzymes (MTHFR, MTR, MTRR) genetic variants with non-obstructive male infertility in a Polish population.. Mateusz Kurzawski 1 , Anna Wajda 1
Because of the involvement of MTHFR, MTR and MTRR genes with folate metabolism and the evidence that maternal use of folic acid in early pregnancy reduces the risk for CL/P,